Abstract
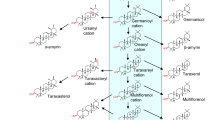
Berberine, palmatine and dehydrocoreximine are end products of protoberberine biosynthesis. These quaternary protoberberines are elicitor inducible and, like other phytoalexins, are highly oxidized. The oxidative potential of these compounds is derived from a diverse array of biosynthetic steps involving hydroxylation, intra-molecular C–C coupling, methylenedioxy bridge formation and a dehydrogenation reaction as the final step in the biosynthesis. For the berberine biosynthetic pathway, the identification of the dehydrogenase gene is the last remaining uncharacterized step in the elucidation of the biosynthesis at the gene level. An enzyme able to catalyze these reactions, (S)-tetrahydroprotoberberine oxidase (STOX, EC 1.3.3.8), was originally purified in the 1980s from suspension cells of Berberis wilsoniae and identified as a flavoprotein (Amann et al. 1984). We report enzymatic activity from recombinant STOX expressed in Spodoptera frugiperda Sf9 insect cells. The coding sequence was derived successively from peptide sequences of purified STOX protein. Furthermore, a recombinant oxidase with protoberberine dehydrogenase activity was obtained from a cDNA library of Argemone mexicana, a traditional medicinal plant that contains protoberberine alkaloids. The relationship of the two enzymes is discussed regarding their enzymatic activity, phylogeny and the alkaloid occurrence in the plants. Potential substrate binding and STOX-specific amino acid residues were identified based on sequence analysis and homology modeling.








Similar content being viewed by others
Notes
Molecular mass of coreximine used in this study is m/z 330, since only isotopic labeled coreximine was available. The label(s) are part of the isoquinoline moiety.
Abbreviations
- CDS:
-
Coding sequence
- STOX:
-
(S)-Tetrahydroprotoberberine oxidase
- BBE:
-
Berberine bridge enzyme
References
Amann M, Nagakura N, Zenk MH (1984) (S)-Tetrahydroprotoberberine oxidase, the final enzyme in protoberberine biosynthesis. Tetrahedron Lett 25:953–954
Amann M, Nagakura N, Zenk MH (1988) Purification and properties of (S)-tetrahydroprotoberberine oxidase from suspension-cultured cells of Berberis wilsoniae. Eur J Biochem 175:17–25
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Bock A, Wanner G, Zenk MH (2002) Immunocytological localization of two enzymes involved in berberine biosynthesis. Planta 216:57–63
Brochmann-Hanssen E, Cherng-Chyi F (1971) Opium alkaloids IX. Detection of coreximine in Papaver somniferum L. based on its biosynthesis from reticuline. Pharm Sci 60:873–878
Cabello-Hurtado F, Taton M, Forthoffer N, Kahn R, Bak S, Rahier A, Werck-Reichhart D (1999) Optimized expression and catalytic properties of a wheat obtusifoliol 14 alpha-demethylase (CYP51) expressed in yeast. Complementation of erg11Delta yeast mutants by plant CYP51. Eur J Biochem 262:435–446
Carter CJ, Thornburg RW (2004) Tobacco nectarin V is a flavin-containing berberine bridge enzyme-like protein with glucose oxidase activity. Plant Physiol 134:460–469
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 162:156–159
Crooks GE, Hon G, Chandonia JM, Brenner SE (2004) WebLogo: a sequence logo generator. Genome Res 14(6):1188–1190
Custers JH, Harrison SJ, Sela-Buurlage MB, van Deventer E, Lageweg W, Howe PW, van der Meijs PJ, Ponstein AS, Simons BH, Melchers LS, Stuiver MH (2004) Isolation and characterisation of a class of carbohydrate oxidases from higher plants, with a role in active defense. Plant J 39:147–160
Díaz Chávez ML, Rolf M, Gesell A, Kutchan TM (2011) Characterization of two methylenedioxy bridge-forming cytochromes P450-dependent enzymes of alkaloid formation in the Mexican prickly poppy Argemone mexicana. Arch Biochem Biophys. doi: 10.1016/j.abb.2010.11.016
Dittrich H, Kutchan TM (1991) Molecular cloning, expression, and induction of berberine bridge enzyme, an enzyme essential to the formation of benzophenanthridine alkaloids in the response of plants to pathogenic attack. Proc Natl Acad Sci USA 88:9969–9973
Frick S, Chitty JA, Kramell R, Schmidt J, Allen RS, Larkin PJ, Kutchan TM (2004) Transformation of opium poppy (Papaver somniferum L.) with antisense berberine bridge enzyme gene (anti-bbe) via somatic embryogenesis results in an altered ratio of alkaloids in latex but not in roots. Transgenic Res 13:607–613
Frick S, Kramell R, Schmidt J, Fist A, Kutchan TM (2005) Comparative qualitative and quantitative determination of alkaloids in narcotic and condiment Papaver somniferum cultivars. J Nat Prod 68:666–673
Fujii N, Inui T, Iwasa K, Morishige T, Sato F (2007) Knockdown of berberine bridge enzyme by RNAi accumulates (S)-reticuline and activates a silent pathway in cultured California poppy cells. Transgenic Res 16:363–375
Gotoh T, Miyazaki Y, Sato W, Kikuchi K, Bentley WE (2001) Proteolytic activity and recombinant protein production in virus-infected Sf9 insect cell cultures supplemented with carboxyl and cysteine protease inhibitors. J Biosci Bioeng 92:248–255
Houba-Hérin N, Pethe C, d’Alayer J, Laloue M (1999) Cytokinin oxidase from Zea mays: purification, cDNA cloning and expression in moss protoplasts. Plant J 17:615–626
Huang X, Madan A (1999) CAP3: a DNA sequence assembly program. Genome Res 9:868–877
Klahre U, Noguchi T, Fujioka S, Takatsuto S, Yokota T, Nomura T, Yoshida S, Chua NH (1998) The Arabidopsis DIMINUTO/DWARF1 gene encodes a protein involved in steroid synthesis. Plant Cell 10:1677–1690
Krauss PF, Kutchan TM (1995) Molecular cloning and heterologous expression of a cDNA encoding berbamunine synthase, a C-O phenol-coupling cytochrome P450 from the higher plant Berberis stolonifera. Proc Natl Acad Sci USA 92:2071–2075
Kutchan TM (1998) Molecular genetics of plant alkaloid biosynthesis. Alkaloids 50:257–316
Lee MH, Lai WL, Lin SF, Hsu CS, Liaw SH, Tsai YC (2005) Structural characterization of glucooligosaccharide oxidase from Acremonium strictum. Appl Environ Microbiol 71:8881–8887
Leferink NGH, Heuts DPHM, Fraaije MW, van Berkel WJH (2008) The growing VAO flavoprotein family. Arch Biochem Biophys 474:292–301
Liaw S, Lee DY, Chow LP, Lau GX, Su SN (2001) Structural characterization of the 60-kDa bermuda grass pollen isoallergens, a covalent flavoprotein. Biochem Biophys Res Commun 280:738–743
Matsuno M, Compagnon V, Schoch GA, Schmitt M, Debayle D, Bassard JE, Pollet B, Hehn A, Heintz D, Ullmann P, Lapierre C, Bernier F, Ehlting J, Werck-Reichhart D (2009) Evolution of a novel phenolic pathway for pollen development. Science 325:1688–1692
Morgenstern B (1999) DIALIGN 2: improvement of the segment-to-segment approach to multiple sequence alignment. Bioinformatics 15:211–218
Morris RO, Bilyeu KD, Laskey JG, Cheikh NN (1999) Isolation of a gene encoding a glycosylated cytokinin oxidase from maize. Biochem Biophys Res Commun 255:328–333
Naggie S, Hu YC, Pulliam-Holoman TR, Bentley WE (1997) Substrate (gelatin) gel electrophoretic method for analysis of protease activity in insect (Sf9) cells. Biotechnol Tech 11:297–300
Neelakandan AK, Song Z, Wang J, Richards MH, Wub X, Valliyodan B, Nguyen HT, Nes WD (2009) Cloning, functional expression and phylogenetic analysis of plant sterol 24C-methyltransferases involved in sitosterol biosynthesis. Phytochemistry 70:1982–1998
Nelson DR, Schuler MA, Paquette SM, Werck-Reichhart D, Bak S (2004) Comparative genomics of rice and Arabidopsis. Analysis of 727 cytochrome P450 genes and pseudogenes from a monocot and a dicot. Plant Physiol 135:756–772
Nicholas KB, Nicholas HB Jr, Deerfield DW II (1997) GeneDoc: analysis and visualization of genetic variation. EMBnet.news 4(2):1–4
Paquette S, Møller BL, Bak S (2003) On the origin of family 1 plant glycosyltransferases. Phytochemistry 62:399–413
Park SU, Yu M, Facchini PJ (2002) Antisense RNA-mediated suppression of benzophenanthridine alkaloid biosynthesis in transgenic cell cultures of California poppy. Plant Physiol 128:696–706
Park SU, Yu M, Facchini PJ (2003) Modulation of berberine bridge enzyme levels in transgenic root cultures of California poppy alters the accumulation of benzophenanthridine alkaloids. Plant Mol Biol 51:153–164
Piotrowski M, Volmer JJ (2006) Cyanide metabolism in higher plants: cyanoalanine hydratase is a NIT4 homolog. Plant Mol Biol 61:111–122
Pyle LE, Barton P, Fujiwara Y, Mitchell A, Fidge N (1995) Secretion of biologically active human proapolipoprotein A4 in a baculovirus–insect cell system: protection from degradation by protease inhibitors. J Lipid Res 36:2355–2361
Sirikantaramas S, Morimoto S, Shoyama Y, Ishikawa Y, Wada Y, Shoyama Y, Taura F (2004) The gene controlling marijuana psychoactivity: molecular cloning and heterologous expression of Delta1-tetrahydrocannabinolic acid synthase from Cannabis sativa L. J Biol Chem 279:39767–39774
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Taura F, Sirikantaramas S, Shoyama Y, Yoshikai K, Shoyama Y, Morimoto S (2007) Cannabidiolic-acid synthase, the chemotype-determining enzyme in the fiber-type Cannabis sativa. FEBS Lett 581:2929–2934
Waterhouse AM, Procter JB, Martin DMA, Clamp M, Barton GJ (2009) Jalview version 2: a multiple sequence alignment and analysis workbench. Bioinformatics 25:1189–1191
Winkler A, Hartner F, Kutchan TM, Glieder A, Macheroux P (2006) Biochemical evidence that berberine bridge enzyme belongs to a novel family of flavoproteins containing a bi-covalently attached FAD cofactor. J Biol Chem 281:21276–21285
Winkler A, Łyskowski A, Riedl S, Puhl M, Kutchan TM, Macheroux P, Gruber K (2008) A concerted mechanism for berberine bridge enzyme. Nat Chem Biol 4:739–741
Yang X, Jawdy S, Tschaplinski TJ, Tuskan GA (2009) Genome-wide identification of lineage-specific genes in Arabidopsis, Oryza and Populus. Genomics 93:473–480
Acknowledgments
The authors acknowledge Christian Böttcher (Biozentrum, Halle (Saale), GER) for technical assistance in the purification of STOX protein. We thank Prof. Dr. Jürgen Ehlting (UVIC, Victoria BC, Canada) for interesting discussions on phylogenetics and Russ Chedgy (do.) for critical reading of the manuscript. This work was supported by the Deutsche Forschungsgemeinschaft, Bonn, Fonds der Chemischen Industrie, Frankfurt, the German Academic Exchange Service (DAAD), Bonn and Consejo Nacional de Ciencia y Tecnología (CONACyT) and Programa de Mejoramiento del Profesorado/Universidad Autónoma del Estado de Morelos (PROMEP/UAEM), Mexico.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Gesell and M. L. Díaz Chávez contributed equally to the paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2011_1357_MOESM1_ESM.txt
Suppl. File S1 Translated Aquilegia coerulea contigs and Papaver somniferum Ps_cntg1 Supplementary material 1 (TXT 5 kb)
425_2011_1357_MOESM2_ESM.nwk
Suppl. File S2 Phylogenetic tree file of Fig. 6a. Abbreviations: NCBI GenBank numerical identifiers following of a letter code for taxonomy (NWK 4 kb)
Rights and permissions
About this article
Cite this article
Gesell, A., Díaz Chávez, M.L., Kramell, R. et al. Heterologous expression of two FAD-dependent oxidases with (S)-tetrahydroprotoberberine oxidase activity from Arge mone mexicana and Berberis wilsoniae in insect cells. Planta 233, 1185–1197 (2011). https://doi.org/10.1007/s00425-011-1357-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-011-1357-4