Abstract
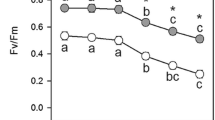
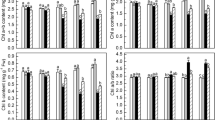
We investigated the differential expression of AOX1 multi-gene family and the regulation of alternative respiratory pathway during initial greening development in leaves of rice (Oryza sativa L.) seedlings. After exposing the dark-grown rice seedlings to continuous irradiation, total respiration (Vt), capacity of alternative pathway (Valt), and their ratio (Valt/Vt) increased with the greening of leaves. In this process, AOX1c transcript increased under constant irradiation, while AOX1a and AOX1b transcripts were hardly detected. Thus AOX1c in rice presents a similar expression pattern as AOX2 does in many dicotyledonous species during greening development. Compared with the rapid increase of cyanideresistant respiration in the presence of photon energy, CO2 fixation was not observed until 8 h after the onset of irradiation. The AOX inhibitor salicylhydroxamic acid (SHAM; 1 mM) inhibited 67.3 % of cyanide-insensitive oxygen uptake in dark-grown leaves and 69.4 % of it in leaves grown under irradiation. Dark-grown plants pre-treated with SHAM were then irradiated for 12 h. SHAM did not obviously modify photosynthetic CO2 fixation rate on a chlorophyll (Chl) content basis in both leaves and simultaneously isolated chloroplasts. Hence during initial greening steps of the plants, the induction of alternative pathway and AOX1 expression by irradiation is not directly linked with carbon assimilation of photosynthesis. The application of SHAM partially limited Chl production in rapidly greening leaves, indicating that Chl synthesis in the process of greening might be medicated to some extent by alternative respiratory pathway.
Similar content being viewed by others
References
Akerlund, P., Larsson, C., Widell, S., Møller, I.M.: NAD(P)H oxidase and peroxidase activities in purified plasma membranes from cauliflower inflorescences.-Physiol. Plant. 71: 9–19, 1987.
Arnon, D.I.: Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris.-Plant Physiol. 24: 1–15, 1949.
Azcón-Bieto, J., Lambers, H., Day, D.A.: Effect of photosynthesis and carbohydrate status on respiratory rates and the involvement of the alternative oxidase pathway in leaf respiration.-Plant Physiol. 72: 598–603, 1983.
Baker, N.R.: Development of chloroplast photochemical functions.-In: Baker, N.R., Barber, J. (ed.): Chloroplast Biogenesis. Pp. 207–251. Elsevier, Amsterdam 1984.
Bartoli, C.G., Gomez, F., Gergoff, G., Guiamét, J.J., Puntarulo, S.: Up-regulation of the mitochondrial alternative oxidase pathway enhances photosynthetic electron transport under drought conditions.-J. exp. Bot. 56: 1269–1276, 2005.
Bingham, I.J., Farrar, J.F.: Activity and capacity of respiration pathways in barley roots deprived of inorganic nutrients.-Plant Physiol. Biochem. 27: 847–854, 1989.
Bingham, I.J., Stevenson, E.A.: Causes and location of non-specific effects of SHAM on O2 uptake by wheat roots.-Physiol. Plant. 93: 427–434, 1995.
Bradbeer, J.W.: Development of photosynthetic function during chloroplast biogenesis.-In: Hatch, M.D., Boardman, N.K. (ed.): The Biochemistry of Plants. Vol. 8. Pp. 423–472. Academic Press, New York-London-Toronto-Sydney-San Francisco 1981.
Bruick, R.K., Mayfield, S.P.: Light-activated translation of chloroplast mRNAs.-Trends Plant Sci. 4: 190–195, 1999.
Chivasa, S., Carr, J.P.: Cyanide restores N gene-mediated resistance to tobacco mosaic virus in transgenic tobacco expressing salicylic acid hydroxylase.-Plant Cell 10: 1489–1498, 1998.
Chivasa, S., Murphy, A.M., Naylor, M., Carr, J.P.: Salicylic acid interferes with tobacco mosaic virus replication via a novel salicylhydroxamic acid-sensitive mechanism.-Plant Cell 9: 547–557, 1997.
Clifton, R., Lister, R., Parker, K.L., Sappl, P.G., Elhafez, D., Millar, A.H., Day, D.A., Whelan, J.: Stress-induced co-expression of alternative respiratory chain components in Arabidopsis thaliana.-Plant mol. Biol. 58: 193–212, 2005.
Considine, M.J., Holtzapffel, R.C., Day, D.A., Whelan, J., Millar, A.H.: Molecular distinction between alternative oxidase from monocots and dicots.-Plant Physiol. 129: 949–953, 2002.
Day, D.A., Wiskich, J.T.: Regulation of alternative oxidase activity in higher plants.-J. Bioenerg. Biomembr. 27: 379–385, 1995.
Diethelm, R., Miller, M.G., Shibles, R., Stewart, C.R.: Effect of salicylhydroxamic acid on respiration, photosynthesis, and peroxidase activity in various plant tissues.-Plant Cell Physiol. 31: 179–185, 1990.
Escobar, M.A., Franklin, K.A., Svensson, A.S., Salter, M.G., Whitelam, G.C., Rasmusson, A.G.: Light regulation of the Arabidopsis respiratory chain. Multiple discrete photoreceptor responses contribute to induction of type IINAD(P)H dehydrogenase genes.-Plant Physiol. 136: 2710–2721, 2004.
Finnegan, P.M., Whelan, J., Millar, A.H., Zhang, Q., Smith, M.K., Wiskich, J.T., Day, D.A.: Differential expression of the multigene family encoding the soybean mitochondrial alternative oxidase.-Plant Physiol. 114: 455–466, 1997.
Gregory P., Bradbeer, J.W.: Plastid development in primary leaves of Phaseolus vulgaris.-Biochem. J. 148: 433–438, 1975.
Gyldenholm, A.O., Whatley, F.: The onset of photophosphorylation in chloroplasts isolated from developing bean leaves.-New Phytol. 67: 461–468, 1986.
Harris, M., Mackender, R.O., Smith, D.L.: Photosynthesis of cotyledons of soybean seedlings.-New Phytol. 104: 319–329, 1986.
Hoefnagel, M.H.N., Atkin, O.K., Wiskich, J.T.: Interdependence between chloroplasts and mitochondria in the light and the dark.-Biochim. biophys. Acta 1366: 235–255, 1998.
Lennon, A.M., Pratt, J., Leach, G., Moore, A.L.: Developmental regulation of respiratory activity in pea leaves.-Plant Physiol. 107: 925–932, 1995.
Lurie, S.: Stomatal opening and photosynthesis in greening leaves of Vicia faba L.-Aust. J. Plant Physiol. 4: 69–74, 1977.
Mackenzie, S., McIntosh, L.: Higher plant mitochondria.-Plant Cell 11: 571–586, 1999.
Mahmudov, Z.M., Abdullayev, K.D.: Photoinhibition of photosystem II in vivo during greening of the wheat seedlings.-Turk. J. Bot. 29: 77–82, 2005.
McCade, T.C., Finnegan, P.M., Millar, A.H., Day, D.A., Whelan, J.: Different expression of alternative oxidase genes in soybean cotyledons during postgerminative development.-Plant Physiol. 118: 675–682, 1998.
Millenaar, F.F., Lambers, H.: The alternative oxidase: in vivo regulation and function.-Plant Biol. 5: 2–15, 2003.
Møller, I.A., Bérczi, A., van der Plas, L.H.W., Lambers, H.: Measurement of the activity and capacity of the alternative pathway in intact plant tissues: identification of problems and possible solutions.-Physiol. Plant. 72: 642–649, 1988.
Naylor, M., Murphy, A.M., Berry, J.O., Carr, J.P.: Salicylic acid can induce resistance to plant virus movement.-Mol. Plant-Microbe Interact. 11: 860–868, 1998.
Obenland, D., Diethelm, R., Shibles, R., Stewart, C.: Relationship of alternative respiratory capacity and alternative oxidase amount during soybean seedling growth.-Plant Cell Physiol. 31: 897–901, 1990.
Padmasree, K., Padmavathi, L., Raghavendra, A.S.: Essentiality of mitochondrial oxidative metabolism for photosynthesis: optimization of carbon assimilation and protection against photoinhibition.-Crit. Rev. Biochem. mol. Biol. 37: 71–119, 2002.
Padmasree, K., Raghavendra, A.S.: Response of photosynthetic carbon assimilation in mesophyll protoplasts to restriction on mitochondrial oxidative metabolism: Metabolites related to the redox status and sucrose biosynthesis.-Photosynth. Res. 62: 231–239, 1999.
Phung Nhu Hung, S., Hoarau, A., Moyse, A.: Étude de l’évolution en chloroplasts des plastes etioles d’orge. II. Photophosphorylation et photoréduction du NADP, formation de ferrédoxine, en éclairement continue et par l’action d’éclairs.-Z. Pflanzenphysiol. 62: 245–250, 1970.
Raghavendra, A.S., Padmasree, K.: Beneficial interactions of mitochondrial metabolism with photosynthetic carbon assimilation.-Trends Plant Sci. 8: 546–553, 2003.
Ribas-Carbo, M., Robinson, S.A., Gonzalez-Meler, M.A., Lennon, A.M., Giles, L., Siedow, J.N., Berry, J.A.: Effects of light on respiration and oxygen isotope fractionation in soybean cotyledons.-Plant Cell Environ. 23: 983–989, 2000.
Saika, H., Ohtsu, K., Hamanaka, S., Nakazono, M.: AOX1c, a novel rice gene for alternative oxidase, comparison with rice AOX1a and AOX1b.-Genes genet. Syst. 77: 31–38, 2002.
Saisho, D., Nakazono, M., Lee, K.H., Tsutsumi, N., Akita, S., Hirai, A.: The gene for alternative oxidase-2 (AOX-2) from Arabidopsis thaliana consists of five exons unlike other AOX genes and is transcribed at an early stage during germination.-Genes genet. Syst. 76: 89–97, 2001.
Svensson, Å.S., Rasmusson, A.G.: Light-dependent gene expression for proteins in the respiratory chain of potato leaves.-Plant J. 28: 1–73, 2001.
Takumi, S., Tomioka, M., Eto, K., Naydenow, N., Nakamura, C.: Characterization of two non-homoeologous nuclear genes encoding mitochondrial alternative oxidase in common wheat.-Genes genet. Syst. 71: 77–88, 2002.
Vanlerberghe, G.C., Alison, E., McIntosh, L.: Molecular genetic evidence of the ability of alternative oxidase to support respiratory carbon metabolism.-Plant Physiol. 113: 657–661, 1997.
Vanlerberghe, G.C., McIntosh, L.: Mitochondrial electron transport regulation of nuclear gene expression: studies with the alternative oxidase gene of tobacco.-Plant Physiol. 105: 867–874, 1994.
Vanlerberghe, G.C., McIntosh, L.: Alternative oxidase: from gene to function.-Annu. Rev. Plant Physiol. Plant mol. Biol. 48: 703–734, 1997.
Van Lis, R., Atteia, A.: Control of mitochondrial function via photosynthetic redox signals.-Photosynth. Res. 79: 133–148, 2004.
Whelan, J., Smith, M.K., Meijer, M., Yu, J.W., Badger, M.R., Price, G.D., Day, D.A.: Cloning of an additional cDNA for alternative oxidase in tobacco.-Plant Physiol. 107: 1469–1470, 1995.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s11099-007-0076-3.
Rights and permissions
About this article
Cite this article
Feng, H.Q., Li, H.Y., Zhou, G.M. et al. Influence of irradiation on cyanide-resistant respiration and AOX1 multi-gene family expression during greening of etiolated rice seedlings. Photosynthetica 45, 272–279 (2007). https://doi.org/10.1007/s11099-007-0044-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11099-007-0044-y