Abstract
Background
Venlafaxine dose regimens vary considerably between individuals, requiring personalized dosing.
Aim
This study aimed to identify dose-related influencing factors of venlafaxine through real-world data analysis and to construct a personalized dose model using advanced artificial intelligence techniques.
Method
We conducted a retrospective study on patients with depression treated with venlafaxine. Significant variables were selected through a univariate analysis. Subsequently, the predictive performance of seven models (XGBoost, LightGBM, CatBoost, GBDT, ANN, TabNet, and DT) was compared. The algorithm that demonstrated optimal performance was chosen to establish the dose prediction model. Model validation used confusion matrices and ROC analysis. Additionally, a dose subgroup analysis was conducted.
Results
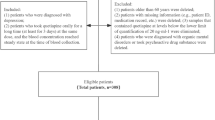
A total of 298 patients were included. TabNet was selected to establish the venlafaxine dose prediction model, which exhibited the highest performance with an accuracy of 0.80. The analysis identified seven crucial variables correlated with venlafaxine daily dose, including blood venlafaxine concentration, total protein, lymphocytes, age, globulin, cholinesterase, and blood platelet count. The area under the curve (AUC) for predicting venlafaxine doses of 75 mg, 150 mg, and 225 mg were 0.90, 0.85, and 0.90, respectively.
Conclusion
We successfully developed a TabNet model to predict venlafaxine doses using real-world data. This model demonstrated substantial predictive accuracy, offering a personalized dosing regimen for venlafaxine. These findings provide valuable guidance for the clinical use of the drug.


Similar content being viewed by others
References
Morton WA, Sonne SC, Verga MA. Venlafaxine: a structurally unique and novel antidepressant. Ann Pharmacother. 1995;29(4):387–95.
Harvey AT, Rudolph RL, Preskorn SH. Evidence of the dual mechanisms of action of venlafaxine. Arch Gen Psychiatry. 2000;57(5):503–9. https://doi.org/10.1001/archpsyc.57.5.503.
Fogelman SM, Schmider J, Venkatakrishnan K, et al. O- and N-demethylation of venlafaxine in vitro by human liver microsomes and by microsomes from cDNA-transfected cells: effect of metabolic inhibitors and SSRI antidepressants. Neuropsychopharmacology. 1999;20(5):480–90.
Ostad Haji E, Hiemke C, Pfuhlmann B. Therapeutic drug monitoring for antidepressant drug treatment. Curr Pharm Des. 2012;18(36):5818–27.
McAlpine DE, Biernacka JM, Mrazek DA, et al. Effect of cytochrome P450 enzyme polymorphisms on pharmacokinetics of venlafaxine. Ther Drug Monit. 2011;33(1):14–20.
Suwała J, Machowska M, Wiela-Hojeńska A. Venlafaxine pharmacogenetics: a comprehensive review. Pharmacogenomics. 2019;20(11):829–45.
Kobylianskii J, Wu PE. Venlafaxine-induced hypoglycemia. CMAJ. 2021;193(16):E568.
Saade YM, Nicol G, Lenze EJ, et al. Comorbid anxiety in late-life depression: Relationship with remission and suicidal ideation on venlafaxine treatment. Depress Anxiety. 2019;36(12):1125–34.
Alexander J, Nillsen A. Venlafaxine-induced delirium. Aust N Z J Psychiatry. 2011;45(7):606.
Murphy L, Rasmussen J, Murphy NG. Venlafaxine overdose treated with extracorporeal life support. CMAJ. 2021;193(5):E167–70.
Deo RC. Machine learning in medicine. Circulation. 2015;132(20):1920–30.
Blonde L, Khunti K, Harris SB, et al. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763–74.
Palacios M. The quality of research with real-world evidence. Colomb Med (Cali). 2019;50(3):140–1.
Joseph LP, Joseph EA, Prasad R. Explainable diabetes classification using hybrid Bayesian-optimized TabNet architecture. Comput Biol Med. 2022;151(Pt A):106178.
Wei X, Ouyang H, Liu M. Stock index trend prediction based on TabNet feature selection and long short-term memory. PLoS ONE. 2022;17(12):e0269195.
Yan J, Xu T, Yu Y, et al. Rainfall forecast model based on the TabNet model. Water. 2021;13(9):1272.
Lundberg S, Lee S I. A unified approach to interpreting model predictions. Adv Neural Inf Process Syst 2017;30. https://doi.org/10.48550/arXiv.1705.07874
Coutens B, Yrondi A, Rampon C, et al. Psychopharmacological properties and therapeutic profile of the antidepressant venlafaxine. Psychopharmacology. 2022;239(9):2735–52.
Debonnel G, Saint-André E, Hébert C, et al. Differential physiological effects of a low dose and high doses of venlafaxine in major depression. Int J Neuropsychopharmacol. 2007;10(1):51–61.
Furukawa TA, Cipriani A, Cowen PJ, et al. Optimal dose of selective serotonin reuptake inhibitors, venlafaxine, and mirtazapine in major depression: a systematic review and dose-response meta-analysis. Lancet Psychiatry. 2019;6(7):601–9.
Wang ZZ, Deng SH, Lu HY, et al. Effect of venlafaxine dosage, valproic acid concentration, sex, and age on steady state dose-corrected concentrations of venlafaxine and O-desmethylvenlafaxine: a retrospective analysis of therapeutic drug monitoring data in a Chinese population. Hum Psychopharmacol. 2020;35(3):e2733.
Smolarczyk-Kosowska J, Kosowski M, Kunert Ł, et al. Impact of venlafaxine on platelet count and activity-case report and narrative review. Medicina (Kaunas). 2022;58(5):626.
Gronau W, Paslakis G, Lederbogen F, et al. Increased platelet count after treatment with venlafaxine or mirtazapine in depressed patients. Pharmacopsychiatry. 2015;48(1):37–9.
Vinholt PJ, Hvas AM, Frederiksen H, et al. Platelet count is associated with cardiovascular disease, cancer and mortality: A population-based cohort study. Thromb Res. 2016;148:136–42.
Castberg I, Westin AA, Skogvoll E, et al. Effects of age and gender on the serum levels of clozapine, olanzapine, risperidone, and quetiapine. Acta Psychiatr Scand. 2017;136(5):455–64.
Fekete S, Scherf-Clavel M, Gerlach M, et al. Dose-corrected serum concentrations and metabolite to parent compound ratios of venlafaxine and risperidone from childhood to old age. Pharmacopsychiatry. 2021;54(3):117–25.
Sobieraj DM, Martinez BK, Hernandez AV, et al. Adverse effects of pharmacologic treatments of major depression in older adults. J Am Geriatr Soc. 2019;67(8):1571–81.
Strawn JR, Mills JA, Suresh V, et al. The impact of age on antidepressant response: a mega-analysis of individuals with major depressive disorder. J Psychiatr Res. 2023;159:266–73.
Yang XX, Hu ZP, Chan SY, et al. Monitoring drug-protein interaction. Clin Chim Acta. 2006;365(1–2):9–29.
Celestin MN, Musteata FM. Impact of changes in free concentrations and drug-protein binding on drug dosing regimens in special populations and disease states. J Pharm Sci. 2021;110(10):3331–44.
Berezhkovskiy LM. On the calculation of the concentration dependence of drug binding to plasma proteins with multiple binding sites of different affinities: determination of the possible variation of the unbound drug fraction and calculation of the number of binding sites of the protein. J Pharm Sci. 2007;96(2):249–57.
Berezhkovskiy LM. On the influence of protein binding on pharmacological activity of drugs. J Pharm Sci. 2010;99(4):2153–65.
Başterzi AD, Yazici K, Buturak V, et al. Effects of venlafaxine and fluoxetine on lymphocyte subsets in patients with major depressive disorder: a flow cytometric analysis. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(1):70–5.
Fazzino F, Obregón F, Morles M, et al. Taurine transporter in lymphocytes of patients with major depression treated with venlafaxine plus psychotherapy. Adv Exp Med Biol. 2009;643:217–24.
Billioti de Gage S, Collin C, Le-Tri T, et al. Antidepressants and hepatotoxicity: a cohort study among 5 million individuals registered in the French national health insurance database. CNS Drugs. 2018;32(7):673–84.
Voican CS, Corruble E, Naveau S, et al. Antidepressant-induced liver injury: a review for clinicians. Am J Psychiatry. 2014;171(4):404–15.
Takeda H, Nishikawa H, Iguchi E, et al. Impact of pretreatment serum cholinesterase level in unresectable advanced hepatocellular carcinoma patients treated with sorafenib. Mol Clin Oncol. 2013;1(2):241–8.
Arik SO, Pfister T. TabNet: Attentive Interpretable Tabular Learning, 2019, 07442. http://arxiv.org/abs/1908. Available from: https://doi.org/10.48550/arXiv.1908.07442. Accessed 16 Nov 2023.
Acknowledgements
None.
Funding
This work was supported by the Finance Department of Hebei Province in China (Grant Number ZF2023020) and the Medical science research project of Hebei Health Commission (Grant Number 20221440).
Author information
Authors and Affiliations
Author notes
Jing Yu and Chunhua Zhou are corresponding authors and contributed equally to this work.
Corresponding author
Ethics declarations
Conflicts of interest
Jinyuan Zhang and Fei Gao are employed by Beijing Medicinovo Technology Co. Ltd., China. Xin Hao is employed by Dalian Medicinovo Technology Co. Ltd., China. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Yu, Z., Ye, X. et al. Personalized venlafaxine dose prediction using artificial intelligence technology: a retrospective analysis based on real-world data. Int J Clin Pharm (2024). https://doi.org/10.1007/s11096-024-01729-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11096-024-01729-7