Abstract
Background
Venlafaxine is frequently prescribed for patients with depression. To control the concentration of venlafaxine within the therapeutic window for the best treatment effect, a model to predict venlafaxine concentration is necessary.
Aim
Our objective was to develop a prediction model for venlafaxine concentration using real-world evidence based on machine learning and deep learning techniques.
Method
Patients who underwent venlafaxine treatment between November 2019 and August 2022 were included in the study. Important variables affecting venlafaxine concentration were identified using a combination of univariate analysis, sequential forward selection, and machine learning techniques. Predictive performance of nine machine learning and deep learning algorithms were assessed, and the one with the optimal performance was selected for modeling. The final model was interpreted using SHapley Additive exPlanations.
Results
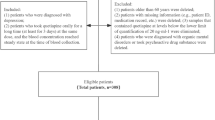
A total of 330 eligible patients were included. Five influential variables that affect venlafaxine concentration were venlafaxine daily dose, sex, age, hyperlipidemia, and adenosine deaminase. The venlafaxine concentration prediction model was developed using the eXtreme Gradient Boosting algorithm (R2 = 0.65, mean absolute error = 77.92, root mean square error = 93.58). In the testing cohort, the accuracy of the predicted concentration within ± 30% of the actual concentration was 73.49%. In the subgroup analysis, the prediction accuracy was 69.39% within the recommended therapeutic range of venlafaxine concentration within ± 30% of the actual value.
Conclusion
The XGBoost model for predicting blood concentration of venlafaxine using real-world evidence was developed, guiding the adjustment of regimen in clinical practice.




Similar content being viewed by others
References
Morton WA, Sonne SC, Verga MA. Venlafaxine: a structurally unique and novel antidepressant. Ann Pharmacother. 1995;29(4):387–95.
Harvey AT, Rudolph RL, Preskorn SH. Evidence of the dual mechanisms of action of venlafaxine. Arch Gen Psychiatry. 2000;57(5):503–9.
Fogelman SM, Schmider J, Venkatakrishnan K, et al. O- and N-demethylation of venlafaxine in vitro by human liver microsomes and by microsomes from cDNA-transfected cells: effect of metabolic inhibitors and SSRI antidepressants. Neuropsychopharmacol. 1999;20(5):480–90.
Ostad Haji E, Hiemke C, Pfuhlmann B. Therapeutic drug monitoring for antidepressant drug treatment. Curr Pharm Design. 2012;18(36):5818.
Paulzen M, Groppe S, Tauber SC, et al. Venlafaxine and O-desmethylvenlafaxine concentrations in plasma and cerebrospinal fluid. J Clin Psychiatry. 2015;76(1):25–31.
Shelton R. Serotonin and norepinephrine reuptake inhibitors. Cham: Springer; 2019. p. 145–80.
Montgomery SA, Mahe V, Haudiquet V, et al. Effectiveness of venlafaxine, extended release formulation, in the short-term and long-term treatment of generalized anxiety disorder: results of a survival analysis. J Clin Psychopharmacol. 2002;22(6):561–7.
Consensus Guidelines for Therapeutic Drug Monitoring in Neuropsychopharmacology; 2017
Hiemke C. Consensus guideline based therapeutic drug monitoring (TDM) in psychiatry and neurology. Curr Drug Deliv. 2016;13(3):353.
Schoretsanitis G, Paulzen M, Unterecker S, et al. TDM in psychiatry and neurology: A comprehensive summary of the consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology, update 2017; a tool for clinicians. World J Biol Psychiatry. 2018;19(3):162–74.
Suwała J, Machowska M, Wiela-Hojeńska A. Venlafaxine pharmacogenetics: a comprehensive review. Pharmacogenomics. 2019;20(11):829–45. https://doi.org/10.2217/pgs-2019-0031.
Kobylianskii J, Wu PE. Venlafaxine-induced hypoglycemia. CMAJ. 2021;193(16):E568. https://doi.org/10.1503/cmaj.78409.
Saade YM, Nicol G, Lenze EJ, et al. Comorbid anxiety in late-life depression: relationship with remission and suicidal ideation on venlafaxine treatment. Depress Anxiety. 2019;36(12):1125–34. https://doi.org/10.1002/da.22964.
Alexander J, Nillsen A. Venlafaxine-induced delirium. Aust N Z J Psychiatry. 2011;45(7):606. https://doi.org/10.3109/00048674.2011.567968.
Murphy L, Rasmussen J, Murphy NG. Venlafaxine overdose treated with extracorporeal life support. CMAJ. 2021;193(5):E167–70. https://doi.org/10.1503/cmaj.201318.
Palacios M. The quality of research with real-world evidence. Colomb Med (Cali). 2019;50(3):140–1.
Robson C. Real world research. 3rd ed. London: Wiley; 2011.
Obermeyer Z, Emanuel EJ. Predicting the future—big data, machine learning, and clinical medicine. N Engl J Med. 2016;375(13):1216–9.
Zheng P, Yu Z, Li L, et al. Predicting blood concentration of tacrolimus in patients with autoimmune diseases using machine learning techniques based on real-world evidence. Front Pharmacol. 2021. https://doi.org/10.3389/fphar.2021.727245.
Guo W, Yu Z, Gao Y, et al. A machine learning model to predict risperidone active moiety concentration based on initial therapeutic drug monitoring. Front Psychiatry. 2021. https://doi.org/10.3389/fpsyt.2021.711868.
Hao Y, Zhang J, Yang L, et al. A machine learning model for predicting blood concentration of quetiapine in patients with schizophrenia and depression based on real-world data. Br J Clin Pharmacol. 2023;89(9):2714–25.
Spratt DE, Tang S, Sun Y, et al. Artificial intelligence predictive model for hormone therapy use in prostate cancer. NEJM Evid. 2023;2(8):EVIDoa2300023.
Lundberg S, Lee SI. A unified approach to interpreting model Predictions//Nips.2017.
Chen T, Guestrin C. In: Proceedings of the 22nd ACM SIGKDD international conference on knowledge discovery and data mining. XGBoost: A scalable tree boosting system. San Francisco, CA: ACM; 2016. pp. 785–94.
Ke, G. et al. LightGBM: in Advances in Neural Information Processing Systems (eds.Guyon, I. et al.) http://dblp.uni-trier.de/db/conf/nips/nips2017.html (CurranAssociates, Inc., 2017).
Breiman L. Random forests. Mach Learn. 2001;45:5–32.
Zhang R, Liu Y, Cao J, et al. The incidence and risk factors analysis of acute kidney injury in hospitalized patients received diuretics: a single-center retrospective study. Front Pharmacol. 2022;13:924173.
Rodríguez-Pérez R, Bajorath J. Evolution of support vector machine and regression modeling in chemoinformatics and drug discovery. J Comput Aided Mol Des. 2022;36(5):355–62.
Renganathan V. Overview of artificial neural network models in the biomedical domain. Bratisl Lek Listy. 2019;120(7):536–40. https://doi.org/10.4149/BLL_2019_087.
Arik SO, Pfister T. TabNet: attentive interpretable tabular learning. 2020. https://doi.org/10.48550/arXiv.1908.07442.
General Chair-Krishnapuram B, General Chair-Shah M, Program Chair-Smola A, et al. Proceedings of the 22nd ACM SIGKDD international conference on knowledge discovery and data mining//Acm Sigkdd international conference on knowledge discovery & data mining. ACM; 2016.
Hansen MR, Kuhlmann IB, Pottegård A, et al. Therapeutic drug monitoring of venlafaxine in an everyday clinical setting: analysis of age, sex and dose concentration relationships. Basic Clin Pharmacol. 2017;121(4):298–302.
Richards-Belle A, Austin-Zimmerman I, Wang B, et al. Associations of antidepressants and antipsychotics with lipid parameters: Do CYP2C19/CYP2D6 genes play a role? A UK population-based study J Psychopharmacol. 2023;37(4):396–407.
Whyte EM, Romkes M, Mulsant BH, et al. CYP2D6 genotype and venlafaxine-XR concentrations in depressed elderly. Int J Geriatr Psychiatry. 2006;21(6):542–9.
Dean L. Venlafaxine therapy and CYP2D6 genotype. In: Pratt VM, Scott SA, Pirmohamed M, Esquivel B, Kattman BL, Malheiro AJ, editors. Medical genetics summaries. Bethesda: National Center for Biotechnology Information (US); 2015.
Lessard E, Yessine M, Hamelin BA, et al. Diphenhydramine alters the disposition of venlafaxine through inhibition of CYP2D6 activity in humans. J Clin Psychopharm. 2001;21(2):175–84.
Paulzen M, Schoretsanitis G, Hiemke C, et al. Reduced clearance of venlafaxine in a combined treatment with quetiapine. Prog Neuropsychopharmacol Biol Psychiatry. 2018;13(85):116–21.
Wang Z, Li L, Huang S, et al. Joint population pharmacokinetic modeling of venlafaxine and O-desmethyl venlafaxine in healthy volunteers and patients to evaluate the impact of morbidity and concomitant medication. Front Pharmacol. 2022;13:978202.
Funding
This work was supported by the Finance Department of Hebei Province in China (Grant Number ZF2023020) and the Medical science research project of the Hebei Health Commission (Grant Number 20221440).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Xin Hao is employed by Dalian Medicinovo Technology Co. Ltd., China. Jinyuan Zhang, Ze Yu and Fei Gao are employed by Beijing Medicinovo Technology Co. Ltd., China. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, L., Hao, X., Yu, J. et al. Developing a machine learning model for predicting venlafaxine active moiety concentration: a retrospective study using real-world evidence. Int J Clin Pharm (2024). https://doi.org/10.1007/s11096-024-01724-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11096-024-01724-y