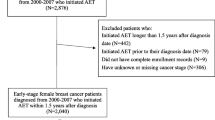
Abstract
Background
Adherence to oral endocrine therapy (OET) is crucial in ensuring its maximum benefit in the prevention and treatment of hormone receptor-positive (HR +) breast cancer (BC). Medication use behavior is suboptimal especially in racial/ethnic minorities with lower socioeconomic status (SES).
Aim
We aimed to assess the impact of the coronavirus disease 2019 (COVID-19) pandemic on OET adherence and identify demographic and/or clinical characteristics associated with nonadherence in racial/ethnic minorities with lower SES.
Method
A retrospective study was conducted at the Harris Health System in Houston, Texas. Data were collected during the 6 months before and 6 months after the start of the pandemic. The adherence was assessed using the prescription refill data using the proportion of days covered. A multivariable logistic regression model was used to identify demographic/clinical characteristics associated with nonadherence. Eighteen years or older patients on appropriate doses of OET for prevention or treatment of BC were included.
Results
In 258 patients, adherence was significantly lower during the pandemic (44%) compared to before the pandemic (57%). The demographic/clinical characteristics associated with OET nonadherence before the pandemic were Black/African American, obesity/extreme obesity, prevention setting, tamoxifen therapy, and 4 or more years on OET. During the pandemic, prevention setting and those not using home delivery were more likely to be nonadherent.
Conclusion
OET adherence was significantly reduced during the COVID-19 pandemic in racial/ethnic minority patients with low SES. Patient-centered interventions are necessary to improve OET adherence in these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Impact statements
-
The adherence to OET for breast cancer prevention or treatment was reduced in racial/ethnic minority patients with low socioeconomic status during the COVID-19 pandemic.
-
Patients taking OET for prevention have worse adherence compared to those taking it for treatment of HR+ BC; Black/African Americans had worse adherence than Hispanic patients.
-
Patients who used home delivery had better adherence during the pandemic, suggesting that this intervention may be useful even during the non-pandemic times.
Introduction
Hormone receptor-positive (HR +) breast cancer (BC) is the most common subtype found in about 80% of BC patients [1,2,3]. In HR + BC patients, better adherence to oral endocrine therapy (OET) results in superior outcomes [4,5,6,7,8,9]. Despite a 30–50% reduction in recurrence risk by 5–10 years of OET, nonadherence rate is high among the patients [10,11,12]. The Food and Drug Administration (FDA)-approved OETs used are tamoxifen and Aromatase Inhibitors (AIs) such as anastrozole, exemestane, and letrozole [13,14,15,16,17,18]. OETs are also used in the prevention setting for patients considered at higher risk of BC and have shown to reduce the risk of new BC diagnoses by ~ 40% [19]. Nonadherence to OET is more common among racial/ethnic minority groups compared to non-Hispanic whites [20,21,22,23,24,25] and in patients with financial burdens [21, 23, 26]. Higher OET nonadherence, reported in up to 80% of racial/ethnic minorities and low-income patients, likely contributes to the disparity in their outcomes [27,28,29].
Cancer health disparity has been a significant societal burden in the United States (U.S.) and is further magnified during the recent coronavirus disease 2019 (COVID-19) pandemic. Before the pandemic, high out-of-pocket medical costs and high unemployment rates due to cancer were common causes of treatment refusal or nonadherence in cancer patients [30, 31]. The COVID-19 pandemic has further increased the stress level of already vulnerable cancer patients because of the fear of contracting the virus and inadequate access to and utilization of health care, especially in those with low SES [32,33,34]. Therefore, accessible and equitable cancer care has been markedly compromised in racial/ethnic minority cancer patients with low SES during the pandemic [35, 36]. The impact of the COVID-19 pandemic and barriers to OET adherence in racial/ethnic minority patients with low SES are poorly understood despite the established clinical benefit of OET adherence.
Aim
The goal of this study was to assess the impact of the COVID-19 pandemic on OET adherence and identify patients and/or tumor characteristics associated with nonadherence in racial/ethnic minorities with lower SES.
Ethics approval
The study was approved by the institutional review board of the University of Houston (STUDY00001818, approved on Jan 30, 2020) and Harris Health System (Protocol 20-02-2286, approved on March 3, 2020) with a waiver of informed consent.
Method
Study design and data source
This retrospective, single-center study included HR + BC patients on OET at Harris Health System in Houston, Texas, which is a county hospital system serving minority patients with low SES. A list of patients of Harris Health System with at least one OET dispensed record from June 2019 through September 2020 was extracted from the EPIC Electronic Health Record (EHR) system. The dispense history from June 2019 through August 2019 was used to determine whether the patients had OET supply in and after September 2019.
Data management
We collected the following data: patient demographics, height and weight to calculate body mass index (BMI), chronic diseases and comorbidities, diagnoses date, pathological and clinical cancer stage at diagnosis, HR and HER2 status, OET name and dosing, date of first OET prescription and initiation date, OET frequency dispense data, OET dispense quantity, date of OET discontinuation, any change in OET therapy, reason for discontinuation or change, number of month’s supply that was covered using mail-order drug delivery (to patients’ homes) after March 31st, 2020, and number of telemedicine appointments after March 31st, 2020. The patients’ prescription refill data included the combination of the prescription dispensed data of any outpatient pharmacy with an integrated e-prescribing system. Thirty patients were included in piloted data collection to review any necessary updates in the data collection form and crosscheck the consistency of data collection by two investigators. Queries were resolved by licensed and board-certified oncology pharmacists (MVT and OO). Random audits were performed during and after data collection to ensure data integrity.
Study population
Inclusion criteria included patients with at least 18 years of age, seen and followed at Harris Health System, of any stage of HR + BC, and those whose OET was not discontinued due to death or progression. Patients not taking appropriate doses for OET (tamoxifen 20 mg once daily, anastrozole 1 mg once daily, letrozole 2.5 mg once daily, and exemestane 25 mg once daily) or on OET for reasons other than prevention or treatment of HR + BC were excluded.
Study variables
The primary endpoint was OET adherence during the 6 months prior to (September 2019 through February 2020) and 6 months after the start of the COVID-19 pandemic (April 2020 through September 2020). Adherence to chronic medications including OET could be assessed by calculating the medication possession ratio (MPR) and proportion of days covered (PDC) [37,38,39]. PDC is a method that avoids the limitation of overestimating adherence for patients who refill their prescriptions early with MPR. PDC was calculated by dividing the number of days patients had OET on hand by the 180 days follow-up period. Patients were considered adherent if they had 80% or more days covered for any OET during the follow-up period [40]. For example, if a patient had filled OET medication for 144 days or more out of the 180 days (follow-up days), then they were considered adherent. Independent variable selection was guided by the Andersen Behavioral Model for Healthcare Resource Use Behavior including predisposing, enabling, and need factors [41]. Predisposing factors included age, race/ethnicity, BMI, type of OET, and years on OET. Enabling factors included home delivery (via mail and free-of-charge to patients) during COVID-19 and the use of telehealth during COVID-19. Need factors included cancer stage, HER2, and comorbidities including metabolic syndrome and depression.
Statistical analysis
Adherence before and during the pandemic was compared using Student’s paired t-test or McNemar chi-square test when PDC was used as a continuous or categorical variable, respectively. Two multinomial regression models were conducted to identify the demographic/clinical characteristics associated with nonadherence to OET during each of the 6 months periods. The demographic/clinical characteristics included in the model were age, race/ethnicity, BMI, cancer stage, HER2, type of OET, years on OET, metabolic syndrome, and depression. Home delivery during COVID-19 and use of telehealth during COVID-19 were the additional characteristics included in the model during the pandemic. All the statistical analysis was done using Statistical Analysis System (SAS) version 9.4 (SAS Institute, Cary, NC) at a priori significance level of 0.05. The coefficients and resultant odds ratios (odds of an event in the adherent group divided by the odds of an event in the nonadherent group) were automatically generated by the function PROC LOGISTIC in SAS.
Results
Out of 270 patients, 12 patients were excluded for taking OET for reasons other than HR + BC. The study cohort included 258 patients.
Baseline characteristics
The baseline demographic and clinical characteristics of patients are presented in Table 1. These characteristics were representative of the patient population of Harris Health System. Among 258 patients that were included in the data analysis, 54 patients were diagnosed with non-invasive (stage 0) BC or ductal/lobular hyperplasia. Out of these 54 patients, 39 (72%) patients had ductal carcinoma in situ (DCIS), 6 (11%) patients had lobular carcinoma in situ (LCIS), 8 (15%) patients had ductal hyperplasia, 1 (2%) had lobular hyperplasia.
Impact of COVID-19 pandemic on OET adherence
The mean PDC from September 2019 to February 2020 was 0.72 and from April 2020 to September 2020 was 0.52. Of the 258 patients, 112 (43%) patients were nonadherent before the start of COVID-19 pandemic and 171 (66%) patients were nonadherent during the pandemic. There was a significant difference in the adherence before and during the pandemic when PDC was used as a continuous (p < 0.0001, Student’s paired t-test) or a categorical variable (p < 0.0001, McNemar chi-square test).
Demographic and clinical characteristics associated with nonadherence before the pandemic
An exploratory multivariable logistic regression analysis of data 6 months before the pandemic identified several factors significantly associated with OET non-adherence. Black/African American and White/Caucasian were less likely to be adherent compared to Hispanic/Latino (Black/African American: odds ratio (OR), 0.43; 95% confidence interval [CI], 0.22–0.84; White/Caucasian: OR, 0.20; 95% CI 0.05–0.73). Obese or extremely obese patients (BMI of ≥ 30) were less likely to be adherent compared to those with a BMI of < 30 (OR, 0.55; 95% CI 0.30–1.01). Patients diagnosed with invasive BC (stages 1–4) were more likely to be adherent compared to those diagnosed with non-invasive tumors or ductal/lobular hyperplasia. Patients on aromatase inhibitors were more likely to be adherent compared to patients on tamoxifen (OR, 2.60; 95% CI 1.26–5.36). Patients taking OET for four years or longer were less likely to be adherent compared to those who were on OET for less than four years (OR, 0.29; 95% CI 0.13–0.65) (Table 2). The forest plot showing the odds-ratio before COVID-19 pandemic is presented as Fig. 1.
Forest plot for predictors of OET adherence before COVID-19 pandemic. Forest plots were constructed using SAS. The codes for various covariates are as follows: Depression (0 = No, 1 = Yes), Metabolic syndrome (0 = No, 1 = Yes), Body mass index (0 = < 30 Ref, 1 = ≥ 30), Years on Oral Endocrine Therapy (0 = ≤ 1 year, 1 = 2–3 years, 2 = ≥ 4 years), Oral endocrine therapy (0 = Tamoxifen, 1 = Aromatase inhibitor), HER2 (0 = HER2-negative, 1 = HER2-positive), Stage (0 = stage 0 or Ductal/lobular hyperplasia, 1 = stage I, 2 = stage II, 3 = stage III, 4 = stage IV), Race (1 = Black/African American, 2 = Hispanic/Latino, 3 = White/Caucasian, 4 = Others), Age (0 = Below 65 years, 1 = 65 years and above)
Demographic and clinical characteristics associated with nonadherence during the pandemic
Patients who were diagnosed with invasive BC (stages 1–3) were more likely to be adherent compared to those diagnosed with non-invasive tumors or ductal/lobular hyperplasia. Patients using home delivery at least once were more likely to be adherent compared to those not using home delivery (OR, 25.42; 95% CI 7.44–86.81) (Table 3). The proportion of patients using home delivery was not significantly different between racial/ethnic groups. The forest plot showing the odds-ratio during COVID-19 pandemic is presented as Fig. 2.
Forest plot for predictors of OET adherence during COVID-19 pandemic. Forest plots were constructed using SAS. The codes for various covariates are as follows: Telemedicine (0 = 0, 1 = ≥ 1), Home delivery (0 = 0, 1 = ≥ 1), Depression (0 = No, 1 = Yes), Metabolic syndrome (0 = No, 1 = Yes), Body mass index (0 = < 30 Ref, 1 = ≥ 30), Years on Oral Endocrine Therapy (0 = ≤ 1 year, 1 = 2–3 years, 2 = ≥ 4 years), Oral endocrine therapy (0 = Tamoxifen, 1 = Aromatase inhibitor), HER2 (0 = HER2-negative, 1 = HER2-positive), Stage (0 = stage 0 or Ductal/lobular hyperplasia, 1 = stage I, 2 = stage II, 3 = stage III, 4 = stage IV), Race (1 = Black/African American, 2 = Hispanic/Latino, 3 = White/Caucasian, 4 = Others), Age (0 = Below 65 years, 1 = 65 years and above)
Discussion
Statement of key findings
This was a retrospective, single-center study evaluating the impact of the COVID-19 pandemic on OET adherence in racial/ethnic minority patients with low SES. We report that OET adherence was low and was further reduced during the pandemic in patients seen at the Harris Health System that serves the indigent population of Houston. At Harris Health, 45.9% of patients are uninsured, 21.2% are on Medicaid or Children's Health Insurance Program, 12% are on Medicare, and 19.9% are on commercial or other funding. Even before the pandemic, OET adherence was low at only 57% in patients with low SES. This was lower than the OET adherence of 82% at the Houston Methodist Hospital serving insured patients in the same geographic location of Houston before the pandemic in our previous study [42]. The pandemic further and significantly reduced OET adherence to only 44% of Harris Health patients. COVID-19 has been found to negatively impact cancer outcomes in racial/ethnic minorities and patients with low SES [43,44,45]. While access to life-saving medications such as OET was provided to Harris Health patients by introducing home delivery, it was not utilized by all patients, which significantly impacted adherence during the pandemic. Identifying prohibitive factors for using home delivery is necessary to optimize outcomes in this patient population.
Strengths and weaknesses
Strengths of our study include the significance of the research, novelty of our findings, and the study design. Harris Health System currently does not have an existing program to improve adherence. While our finding of high OET nonadherence in racial/ethnic minorities with low SES is consistent with published reports, we have several novel findings as well. For example, the impact of COVID-19 on OET adherence, although expected, has not been reported before. Also, our study has identified a major area of unmet clinical need, in the setting of improving OET adherence for BC prevention, which is a largely ignored area of research until this point. Our study highlights home delivery as a simple solution to improve OET adherence that can also be applicable to other chronic medications and relevant in future pandemic or natural disaster settings. The findings of this study can also be used to design and implement future interventions and programs customized to African American and Hispanic patients to improve their long-term adherence. Our study provides critical information for future studies conducting a real-time assessment of adherence and implementation of interventions in the ambulatory oncology clinic.
Some limitations of this study are described below. This was a retrospective study conducted in one center, which can limit the generalizability of findings to similar centers and geographic locations. This is a hypothesis generating exploratory analysis. Given the scarcity of studies specifically conducted in racial/ethnic minority patients with low SES, future more theory-driven studies that take the findings of this study into consideration are warranted. The relatively small sample size may have impacted the power to detect significant differences and lead to wide confidence intervals with some variables. Therefore, additional larger studies are needed to validate our findings and confirm the demographic and clinical characteristics associated with non-adherence in racial/ethnic minorities with low SES. Refilling the medications does not guarantee the patient actually consumes the medication as prescribed. Additionally, residual uncontrolled confounding may exist as some variables that can impact adherence were not controlled for, like educational level, income, family support, and other comorbidities. Despite these limitations, the study adds to the growing literature on low OET adherence rate among racial/ethnic minorities with low SES, underscoring the need for tailored interventions to enhance adherence in this population.
Interpretation
We found that stage was associated with OET nonadherence in our population. Patients at higher risk of invasive BC, namely those diagnosed with non-invasive BC or with ductal/lobular hyperplasia, were less likely to be adherent to OET.
Most studies to date have evaluated adjuvant OET adherence in the setting of early-stage BC treatment, not for prevention [20,21,22,23,24,25]. Our study emphasizes an important need to evaluate barriers to OET adherence in the prevention setting and for tailored interventions to improve OET adherence since it reduces the risk of new BC diagnosis.
Our results of lower OET adherence in Black/African Americans are consistent with published literature [21,22,23,24,25]. African Americans have a higher incidence of certain cancers and face greater hurdles to cancer prevention, detection, and treatment [46]. As a result, African Americans have the highest death rate and shortest survival of any racial/ethnic group for most cancers [46,47,48]. Despite slightly lower rates of breast cancer (BC) diagnosis, African American women are over 40% more likely to die from it than Caucasians in Texas and the U.S. [27, 28, 46, 47, 49]. The most common barrier to OET adherence reported by patients and physicians is adverse drug reactions [50,51,52]. African American women more often report postmenopausal symptoms and joint pain with OET than Caucasians [53, 54]. They also more often believe that their recurrence risk would not change if they stopped OET and report forgetting to take it regularly [53,54,55,56]. Therefore, patient-centered interventions addressing these key barriers to OET adherence in African American women, especially those with low SES, are urgently needed.
A large subset (62%) of patients in our study was obese or extremely obese, which was also associated with nonadherence. Higher body weight has been associated with worse BC-specific outcomes in patients, and body weight management is linked to favorable BC-specific survival [17, 57,58,59]. It is possible that patients who are more health-conscious in general are more likely to maintain their ideal body weight and stay on therapy. Therefore, adequate patient education as well as interventions to improve overall health including weight management and OET adherence are necessary to improve overall BC outcomes in HR + BC patients with low SES.
Further research
Our findings were communicated to the breast medical oncology and the cancer prevention team as well as the Pharmacy Department at the Harris Health Hospital. Department of Pharmacy had instituted the home delivery option during the pandemic. Since not using home delivery adversely affected OET adherence, encouraging patients to use it could help improve their OET adherence. The home delivery option is still available to all Harris Health patients, and its optimized use could lower the burden of the pandemic on patients with low SES. Future directions for research include developing patient-centered interventions and testing their feasibility and effectiveness to improve treatment outcomes and survival in racial and ethnic minorities with lower SES.
Conclusion
In conclusion, OET adherence is a major concern in racial/ethnic minorities with low SES with HR + BC. The COVID-19 pandemic adversely affected OET adherence. Therefore, patient-centered interventions tailored to Black/African American patients and those using OET for prevention are urgently needed to improve BC outcomes in this population.
References
Giuliano M, Schifp R, Osborne CK, et al. Biological mechanisms and clinical implications of endocrine resistance in breast cancer. Breast. 2011;20(Suppl 3):S42–9. https://doi.org/10.1016/S0960-9776(11)70293-4.
Harbeck N, Gnant M. Breast cancer. Lancet. 2017;389(10074):1134–50. https://doi.org/10.1016/S0140-6736(16)31891-8.
Clark GM, Osborne CK, McGuire WL. Correlations between estrogen receptor, progesterone receptor, and patient characteristics in human breast cancer. J Clin Oncol. 1984;2(10):1102–9. https://doi.org/10.1200/JCO.1984.2.10.1102.
Chirgwin JH, Giobbie-Hurder A, Coates AS, et al. Treatment Adherence and Its Impact on Disease-Free Survival in the Breast International Group 1–98 Trial of Tamoxifen and Letrozole, Alone and in Sequence. J Clin Oncol. 2016;34(21):2452–9. https://doi.org/10.1200/JCO.2015.63.8619.
Early Breast Cancer Trialists’ Collaborative Group. Aromatase inhibitors versus tamoxifen in premenopausal women with oestrogen receptor-positive early-stage breast cancer treated with ovarian suppression: a patient-level meta-analysis of 7030 women from four randomised trials. Lancet Oncol. 2022;23(3):382–92. https://doi.org/10.1016/S1470-2045(21)00758-0.
Early Breast Cancer Trialists’ Collaborative Group. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378(9793):771–84. https://doi.org/10.1016/S0140-6736(11)60993-8.
Font R, Buxo M, Ameijide A, et al. Using population-based data to evaluate the impact of adherence to endocrine therapy on survival in breast cancer through the web-application BreCanSurvPred. Sci Rep. 2022;12(1):8097. https://doi.org/10.1038/s41598-022-12228-y.
Ponde NF, Zardavas D, Piccart M. Progress in adjuvant systemic therapy for breast cancer. Nat Rev Clin Oncol. 2019;16(1):27–44. https://doi.org/10.1038/s41571-018-0089-9.
Waks AG, Winer EP. Breast cancer treatment: a review. JAMA. 2019;321(3):288–300. https://doi.org/10.1001/jama.2018.19323.
Murphy CC, Bartholomew LK, Carpentier MY, et al. Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat. 2012;134(2):459–78. https://doi.org/10.1007/s10549-012-2114-5.
Sood N, Liu Y, Lian M, et al. Association of endocrine therapy initiation timeliness with adherence and continuation in low-income women with breast cancer. JAMA Netw Open. 2022;5(8):e2225345. https://doi.org/10.1001/jamanetworkopen.2022.25345.
Yussof I, Mohd Tahir NA, Hatah E, et al. Factors influencing five-year adherence to adjuvant endocrine therapy in breast cancer patients: a systematic review. Breast. 2022;62:22–35. https://doi.org/10.1016/j.breast.2022.01.012.
Burstein HJ, Lacchetti C, Anderson H, et al. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: ASCO clinical practice guideline focused update. J Clin Oncol. 2019;37(5):423–38. https://doi.org/10.1200/JCO.18.01160.
Goss PE, Ingle JN, Pritchard KI, et al. Extending aromatase-inhibitor adjuvant therapy to 10 years. N Engl J Med. 2016;375(3):209–19. https://doi.org/10.1056/NEJMoa1604700.
Hong R, Xu B. Breast cancer: an up-to-date review and future perspectives. Cancer Commun (Lond). 2022. https://doi.org/10.1002/cac2.12358.
Li L, Chang B, Jiang X, et al. Clinical outcomes comparison of 10 years versus 5 years of adjuvant endocrine therapy in patients with early breast cancer. BMC Cancer. 2018;18(1):977. https://doi.org/10.1186/s12885-018-4878-4.
Ligibel JA, Basen-Engquist K, Bea JW. Weight management and physical activity for breast cancer prevention and control. Am Soc Clin Oncol Educ Book. 2019;39:e22–33. https://doi.org/10.1200/EDBK_237423.
AlFakeeh A, Brezden-Masley C. Overcoming endocrine resistance in hormone receptor-positive breast cancer. Curr Oncol. 2018;25(Suppl 1):S18–27. https://doi.org/10.3747/co.25.3752.
Chlebowski RT, Kim J, Haque R. Adherence to endocrine therapy in breast cancer adjuvant and prevention settings. Cancer Prev Res (Phila). 2014;7(4):378–87. https://doi.org/10.1158/1940-6207.CAPR-13-0389.
Biggers A, Shi Y, Charlson J, et al. Medicare D subsidies and racial disparities in persistence and adherence with hormonal therapy. J Clin Oncol. 2016;34(36):4398–404. https://doi.org/10.1200/JCO.2016.67.3350.
Farias AJ, Du XL. Racial differences in adjuvant endocrine therapy use and discontinuation in association with mortality among medicare breast cancer patients by receptor status. Cancer Epidemiol Biomarkers Prev. 2017;26(8):1266–75. https://doi.org/10.1158/1055-9965.EPI-17-0280.
Farias AJ, Wu WH, Du XL. Racial differences in long-term adjuvant endocrine therapy adherence and mortality among Medicaid-insured breast cancer patients in Texas: findings from TCR-Medicaid linked data. BMC Cancer. 2018;18(1):1214. https://doi.org/10.1186/s12885-018-5121-z.
Roberts MC, Wheeler SB, Reeder-Hayes K. Racial/Ethnic and socioeconomic disparities in endocrine therapy adherence in breast cancer: a systematic review. Am J Public Health. 2015;105(Suppl 3):e4–15. https://doi.org/10.2105/AJPH.2014.302490.
Sheppard VB, He J, Sutton A, et al. Adherence to adjuvant endocrine therapy in insured black and white breast cancer survivors: exploring adherence measures in patient data. J Manag Care Spec Pharm. 2019;25(5):578–86. https://doi.org/10.18553/jmcp.2019.25.5.578.
Sheppard VB, Sutton AL, Hurtado-de-Mendoza A, et al. Race and patient-reported symptoms in adherence to adjuvant endocrine therapy: a report from the women’s hormonal initiation and persistence study. Cancer Epidemiol Biomarkers Prev. 2021;30(4):699–709. https://doi.org/10.1158/1055-9965.EPI-20-0604.
Smith GL, Lopez-Olivo MA, Advani PG, et al. Financial burdens of cancer treatment: a systematic review of risk factors and outcomes. J Natl Compr Canc Netw. 2019;17(10):1184–92. https://doi.org/10.6004/jnccn.2019.7305.
DeSantis CE, Ma J, Goding Sauer A, et al. Breast cancer statistics, 2017, racial disparity in mortality by state. CA Cancer J Clin. 2017;67(6):439–48. https://doi.org/10.3322/caac.21412.
Ma H, Lu Y, Malone KE, et al. Mortality risk of black women and white women with invasive breast cancer by hormone receptors, HER2, and p53 status. BMC Cancer. 2013;13:225. https://doi.org/10.1186/1471-2407-13-225.
Zavala VA, Bracci PM, Carethers JM, et al. Cancer health disparities in racial/ethnic minorities in the United States. Br J Cancer. 2021;124(2):315–32. https://doi.org/10.1038/s41416-020-01038-6.
Ramsey S, Blough D, Kirchhoff A, et al. Washington State cancer patients found to be at greater risk for bankruptcy than people without a cancer diagnosis. Health Aff (Millwood). 2013;32(6):1143–52. https://doi.org/10.1377/hlthaff.2012.1263.
Ramsey SD, Bansal A, Fedorenko CR, et al. Financial insolvency as a risk factor for early mortality among patients with cancer. J Clin Oncol. 2016;34(9):980–6. https://doi.org/10.1200/JCO.2015.64.6620.
Carlos RC, Lowry KP, Sadigh G. The coronavirus disease 2019 (COVID-19) pandemic: a patient-centered model of systemic shock and cancer care adherence. J Am Coll Radiol. 2020;17(7):927–30. https://doi.org/10.1016/j.jacr.2020.05.032.
Garcia S, Albaghdadi MS, Meraj PM, et al. Reduction in ST-segment elevation cardiac catheterization laboratory activations in the united states during COVID-19 pandemic. J Am Coll Cardiol. 2020;75(22):2871–2. https://doi.org/10.1016/j.jacc.2020.04.011.
Lee CI, Raoof S, Patel SB, et al. Coronavirus disease 2019 (COVID-19) and your radiology practice: case triage, staffing strategies, and addressing revenue concerns. J Am Coll Radiol. 2020;17(6):752–4. https://doi.org/10.1016/j.jacr.2020.04.025.
Kretchy IA, Asiedu-Danso M, Kretchy JP. Medication management and adherence during the COVID-19 pandemic: Perspectives and experiences from low-and middle-income countries. Res Social Adm Pharm. 2021;17(1):2023–6. https://doi.org/10.1016/j.sapharm.2020.04.007.
Patel MI, Lopez AM, Blackstock W, et al. Cancer disparities and health equity: a policy statement from the american society of clinical oncology. J Clin Oncol. 2020;38(29):3439–48. https://doi.org/10.1200/JCO.20.00642.
Benner JS, Glynn RJ, Mogun H, et al. Long-term persistence in use of statin therapy in elderly patients. JAMA. 2002;288(4):455–61. https://doi.org/10.1001/jama.288.4.455.
Canfield SL, Zuckerman A, Anguiano RH, et al. Navigating the wild west of medication adherence reporting in specialty pharmacy. J Manag Care Spec Pharm. 2019;25(10):1073–7. https://doi.org/10.18553/jmcp.2019.25.10.1073.
Mohan A, Vadhariya A, Majd Z, et al. Impact of a motivational interviewing intervention targeting statins on adherence to concurrent hypertension or diabetes medications. Patient Educ Couns. 2021;104(7):1756–64. https://doi.org/10.1016/j.pec.2020.12.009.
Bhandari S, Ngo P, Kute B, et al. Adherence to anti-estrogen therapy in women with hormone receptor-positive breast cancer utilizing bubble packaging: a pilot study. Breast Cancer Res Treat. 2019;177(2):395–9. https://doi.org/10.1007/s10549-019-05308-7.
Andersen R, Newman JF. Societal and individual determinants of medical care utilization in the United States. Milbank Mem Fund Q Health Soc. 1973;51(1):95–124.
Hwang GS, Paranjpe R, Opsomer C, et al. Oral endocrine therapy agent, race/ethnicity, and time on therapy predict adherence in breast cancer patients in a large academic institution. Clin Breast Cancer. 2020;20(6):520–6. https://doi.org/10.1016/j.clbc.2020.06.004.
Balogun OD, Bea VJ, Phillips E. Disparities in cancer outcomes due to COVID-19-A tale of 2 cities. JAMA Oncol. 2020;6(10):1531–2. https://doi.org/10.1001/jamaoncol.2020.3327.
Cancino RS, Su Z, Mesa R, et al. The impact of COVID-19 on cancer screening: challenges and opportunities. JMIR Cancer. 2020;6(2):e21697. https://doi.org/10.2196/21697.
Newman LA, Winn RA, Carethers JM. Similarities in risk for COVID-19 and cancer disparities. Clin Cancer Res. 2021;27(1):24–7. https://doi.org/10.1158/1078-0432.CCR-20-3421.
American Cancer Society. Cancer facts & figures for African Americans. 2019–2021.
DeSantis CE, Miller KD, Goding Sauer A, et al. Cancer statistics for African Americans, 2019. CA Cancer J Clin. 2019;69(3):211–33. https://doi.org/10.3322/caac.21555.
National Cancer Institute. Seer Cancer Statistics Review, 1975–2018. Table 1.20. 2021.
DeSantis CE, Fedewa SA, Goding Sauer A, et al. Breast cancer statistics, 2015: Convergence of incidence rates between black and white women. CA Cancer J Clin. 2016;66(1):31–42. https://doi.org/10.3322/caac.21320.
Clancy C, Lynch J, O'Connor P, et al. Breast cancer patients’ experiences of adherence and persistence to oral endocrine therapy: a qualitative evidence synthesis. Eur J Oncol Nurs. 2020;44:101706. https://doi.org/10.1016/j.ejon.2019.101706.
Milata JL, Otte JL, Carpenter JS. Oral endocrine therapy nonadherence, adverse effects, decisional support, and decisional needs in women with breast cancer. Cancer Nurs. 2018;41(1):E9–18. https://doi.org/10.1097/NCC.0000000000000430.
Paranjpe R, John G, Trivedi M, et al. Identifying adherence barriers to oral endocrine therapy among breast cancer survivors. Breast Cancer Res Treat. 2019;174(2):297–305. https://doi.org/10.1007/s10549-018-05073-z.
Donevant S, Heiney SP, Wineglass C, et al. Perceptions of endocrine therapy in African-American breast cancer survivors: mixed methods study. JMIR Form Res. 2021;5(6):E23884. https://doi.org/10.2196/23884.
Wheeler SB, Spencer J, Pinheiro LC, et al. Endocrine therapy nonadherence and discontinuation in black and white women. J Natl Cancer Inst. 2019;111(5):498–508. https://doi.org/10.1093/jnci/djy136.
Brier MJ, Chambless DL, Gross R, et al. Perceived barriers to treatment predict adherence to aromatase inhibitors among breast cancer survivors. Cancer. 2017;123(1):169–76. https://doi.org/10.1002/cncr.30318.
Spencer JC, Reeve BB, Troester MA, et al. Factors associated with endocrine therapy non-adherence in breast cancer survivors. Psychooncology. 2020;29(4):647–54. https://doi.org/10.1002/pon.5289.
Blair CK, Wiggins CL, Nibbe AM, et al. Obesity and survival among a cohort of breast cancer patients is partially mediated by tumor characteristics. NPJ Breast Cancer. 2019;5:33. https://doi.org/10.1038/s41523-019-0128-4.
Chan DSM, Vieira AR, Aune D, et al. Body mass index and survival in women with breast cancer-systematic literature review and meta-analysis of 82 follow-up studies. Ann Oncol. 2014;25(10):1901–14. https://doi.org/10.1093/annonc/mdu042.
Shang L, Hattori M, Fleming G, et al. Impact of post-diagnosis weight change on survival outcomes in Black and White breast cancer patients. Breast Cancer Res. 2021;23(1):18. https://doi.org/10.1186/s13058-021-01397-9.
Acknowledgements
Not applicable.
Funding
Research reported in this publication was supported in part by the National Institute on Minority Health and Health Disparities (NIMHD) (to Drs. Ononogbu, Abughosh, and Trivedi) and the National Cancer Institute (NCI) (to Dr. Trivedi) under Award Numbers U54MD015946 and 1P20CA221729-01A1, respectively. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIMHD or NCI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no relevant conflicts of interest or financial relationships.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rahimi, S., Ononogbu, O., Mohan, A. et al. Adherence to oral endocrine therapy in racial/ethnic minority patients with low socioeconomic status before and during the COVID-19 pandemic. Int J Clin Pharm 45, 1396–1404 (2023). https://doi.org/10.1007/s11096-023-01609-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-023-01609-6