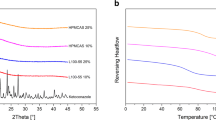
Abstract
Purpose
In amorphous solid dispersions (ASDs), the chemical potential of a drug can be reduced due to mixing with the polymer in the solid matrix, and this can lead to reduced drug release when the polymer is insoluble in the dissolution media. If both the drug and the polymer composing an ASD are ionizable, drug release from the ASD becomes pH-dependent. The goal of this study was to gain insights into the pH-dependent solubility suppression from ASD formulations.
Methods
The maximum release of clotrimazole, a weakly basic drug, from ASDs formulated with insoluble and pH-responsive polymers, was determined as a function of solution pH. Drug-polymer interactions in ASDs were probed using melting point depression, moisture sorption, and solid-state Nuclear Magnetic Resonance spectroscopy (SSNMR) measurements.
Results
The extent of solubility suppression was dependent on polymer type and drug loading. The strength of drug-polymer interactions was found to correlate well with the degree of solubility suppression. For the same ASD, the degree of solubility suppression was nearly constant across the solution pH range studied, suggesting that polymer-drug interactions in residual ASD solids was independent of solution pH. The total drug release agrees with the Henderson-Hasselbalch relationship if the suppressed amorphous solubility of the free drug is independent of solution pH.
Conclusions
The mechanism of solubility suppression at different solution pHs appeared to be drug-polymer interactions in the solid-state, where the concentration of the free drug remains the same at variable pHs and the total drug concentration follows the Henderson-Hasselbalch relationship.










Similar content being viewed by others
Abbreviations
- ASD:
-
Amorphous solid dispersion
- CA:
-
Cellulose acetate
- CTZ:
-
Clotrimazole
- CPMAS:
-
Cross polarization and magic-angle spinning
- DL:
-
Drug loading
- DSC:
-
Differential scanning calorimetry
- EC:
-
Ethylcellulose
- EUD EPO:
-
Eudragit® E PO
- EUD L100:
-
Eudragit® L100
- EUD RLPO:
-
Eudragit® RL PO
- HPLC:
-
High performance liquid chromatography
- HPMC:
-
Hydroxypropyl methylcellulose
- HPMCAS:
-
Hydroxypropyl methylcellulose acetate succinate
- HPMCP:
-
Hydroxypropyl methylcellulose phthalate
- RH:
-
Relative humidity
- SSNMR:
-
Solid-state nuclear magnetic resonance
References
Amidon G, Lennernäs H, Shah V, Crison J. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995;12(3):413–20.
Lipinski CA. Drug-like properties and the causes of poor solubility and poor permeability. J Pharmacol Toxicol. 2000;44(1):235–49.
Di L, Kerns EH, Carter GT. Drug-like property concepts in pharmaceutical design. Curr Pharm Des. 2009;15(19):2184–94.
Williams HD, Trevaskis NL, Charman SA, Shanker RM, Charman WN, Pouton CW, et al. Strategies to address low drug solubility in discovery and development. Pharmacol Rev. 2013;65(1):315–499.
Stegemann S, Leveiller F, Franchi D, de Jong H, Lindén H. When poor solubility becomes an issue: From early stage to proof of concept. Eur J Pharm Sci. 2007;31(5):249–61.
Stainmesse S, Orecchioni AM, Nakache E, Puisieux F, Fessi H. Formation and stabilization of a biodegradable polymeric colloidal suspension of nanoparticles. Colloid Polym Sci. 1995;273(5):505–11.
Raina S, Zhang GZ, Alonzo D, Wu J, Zhu D, Catron N, et al. Impact of solubilizing additives on supersaturation and membrane transport of drugs. Pharm Res. 2015:1–15.
Dahan A, Beig A, Lindley D, Miller JM. The solubility–permeability interplay and oral drug formulation design: Two heads are better than one. Adv Drug Deliver Rev. 2016;101:99–107.
Huang Y, Dai W-G. Fundamental aspects of solid dispersion technology for poorly soluble drugs. Acta Pharm Sin B. 2014;4(1):18–25.
Qian K, Stella L, Jones DS, Andrews GP, Du H, Tian Y. Drug-rich phases induced by amorphous solid dispersion: arbitrary or intentional goal in oral drug delivery? Pharmaceutics. 2021;13(6):889.
Yuan X, Xiang T-X, Anderson BD, Munson EJ. Hydrogen bonding interactions in amorphous indomethacin and its amorphous solid dispersions with poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-vinyl acetate) studied using 13C solid-state NMR. Mol Pharm. 2015;12(12):4518–28.
Aso Y, Yoshioka S. Molecular mobility of nifedipine−PVP and phenobarbital−PVP solid dispersions as measured by 13C-NMR spin-lattice relaxation time. J Pharm Sci. 2006;95(2):318–25.
Zhou D, Schmitt EA, Law D, Brackemeyer PJ, Zhang GGZ. Assessing physical stability risk using the Amorphous Classification System (ACS) based on simple thermal analysis. Mol Pharm. 2019;16(6):2742–54.
Banerjee S. Solubility of organic mixtures in water. Envir Sci Tech. 1984;18(8):587–91.
Li N, Taylor LS. Tailoring supersaturation from amorphous solid dispersions. J Control Release. 2018;279:114–25.
Huang S, Mao C, Williams RO III, Yang C-Y. Solubility advantage (and disadvantage) of pharmaceutical amorphous solid dispersions. J Pharm Sci. 2016;105(12):3549–61.
Li N, Ormes JD, Taylor LS. Leaching of lopinavir amorphous solid dispersions in acidic media. Pharm Res. 2016:1–13.
Morgen M, Bloom C, Beyerinck R, Bello A, Song W, Wilkinson K, et al. Polymeric nanoparticles for increased oral bioavailability and rapid absorption using celecoxib as a model of a low-solubility, high-permeability drug. Pharm Res. 2012;29(2):427–40.
Stephenson GA, Aburub A, Woods TA. Physical stability of salts of weak bases in the solid-state. J Pharm Sci. 2011;100(5):1607–17.
Charifson PS, Walters WP. Acidic and basic drugs in medicinal chemistry: a perspective. J Med Chem. 2014;57(23):9701–17.
Friesen DT, Shanker R, Crew M, Smithey DT, Curatolo WJ, Nightingale JAS. Hydroxypropyl methylcellulose acetate succinate-based spray-dried dispersions: an overview. Mol Pharm. 2008;5(6):1003–19.
Curatolo W, Nightingale JA, Herbig SM. Utility of hydroxypropylmethylcellulose acetate succinate (HPMCAS) for initiation and maintenance of drug supersaturation in the GI milieu. Pharm Res. 2009;26(6):1419–31.
Elkhabaz A, Sarkar S, Simpson GJ, Taylor LS. Characterization of phase transformations for amorphous solid dispersions of a weakly basic drug upon dissolution in biorelevant media. Pharm Res. 2019;36(12):174.
Maghsoodi M, Nokhodchi A, Oskuei MA, Heidari S. Formulation of cinnarizine for stabilization of its physiologically generated supersaturation. AAPS PharmSciTech. 2019;20(3):139.
Hsieh Y-L, Ilevbare GA, Van Eerdenbrugh B, Box KJ, Sanchez-Felix MV, Taylor LS. pH-Induced precipitation behavior of weakly basic compounds: determination of extent and duration of supersaturation using potentiometric titration and correlation to solid state properties. Pharm Res. 2012;29(10):2738–53.
Extrand CW. Spin coating of very thin polymer films. Polym Eng Sci. 1994;34(5):390–4.
Ashland. AquaSolveTM hydroxypropylmethylcellulose acetate succinate: Physical and chemical properties handbook [Available from: http://www.ashland.com/Ashland/Static/Documents/ASI/PC_12624_AquaSolve_AS_Handbook.pdf.
Shin-Etsu. HPMC - for pharmaceutical: Shin-Etsu cellulose [Available from: https://www.metolose.jp/en/pharmaceutical/hpmcp.html.
Jackson MJ, Kestur US, Hussain MA, Taylor LS. Characterization of supersaturated danazol solutions – impact of polymers on solution properties and phase transitions. Pharm Res. 2016;33(5):1276–88.
Sun DD, Lee PI. Probing the mechanisms of drug release from amorphous solid dispersions in medium-soluble and medium-insoluble carriers. J Control Release. 2015;211(Supplement C):85–93.
Marsac P, Shamblin S, Taylor L. Theoretical and practical approaches for prediction of drug–polymer miscibility and solubility. Pharm Res. 2006;23(10):2417–26.
Gui Y, McCann EC, Yao X, Li Y, Jones KJ, Yu L. Amorphous drug–polymer salt with high stability under tropical conditions and fast dissolution: the case of clofazimine and poly(acrylic acid). Mol Pharm. 2021;18(3):1364–72.
Yao X, Neusaenger AL, Yu L. Amorphous drug-polymer salts. Pharmaceutics. 2021;13(8):1271.
Mesallati H, Umerska A, Paluch KJ, Tajber L. Amorphous polymeric drug salts as ionic solid dispersion forms of ciprofloxacin. Mol Pharm. 2017;14(7):2209–23.
Barich DH, Gorman EM, Zell MT, Munson EJ. 3-Methylglutaric acid as a 13C solid-state NMR standard. Solid State Nucl Magn Reson. 2006;30(3):125–9.
Dixon WT, Schaefer J, Sefcik MD, Stejskal EO, McKay RA. Total suppression of sidebands in CPMAS C-13 NMR. J Magn Reson. 1982;49(2):341–5.
Fung BM, Khitrin AK, Ermolaev K. An improved broadband decoupling sequence for liquid crystals and solids. J Magn Reson. 2000;142(1):97–101.
Henderson LJ. Concerning the relationship between the strength of acids and their capacity to preserve neutrality. Am J Physiol. 1908;21(2):173–9.
Indulkar AS, Box KJ, Taylor R, Ruiz R, Taylor LS. pH-Dependent liquid–liquid phase separation of highly supersaturated solutions of weakly basic drugs. Mol Pharm. 2015;12(7):2365–77.
Fine-Shamir N, Dahan A. Methacrylate-copolymer eudragit epo as a solubility-enabling excipient for anionic drugs: investigation of drug solubility, intestinal permeability, and their interplay. Mol Pharm. 2019;16(7):2884–91.
Xie T, Gao W, Taylor LS. Impact of Eudragit EPO and hydroxypropyl methylcellulose on drug release rate, supersaturation, precipitation outcome and redissolution rate of indomethacin amorphous solid dispersions. Int J Pharm. 2017;531(1):313–23.
Onoue S, Kojo Y, Aoki Y, Kawabata Y, Yamauchi Y, Yamada S. Physicochemical and pharmacokinetic characterization of amorphous solid dispersion of tranilast with enhanced solubility in gastric fluid and improved oral bioavailability. Drug Metab Pharmacokinet. 2012;27(4):379–87.
Lin X, Su L, Li N, Hu Y, Tang G, Liu L, et al. Understanding the mechanism of dissolution enhancement for poorly water-soluble drugs by solid dispersions containing Eudragit® E PO. J Drug Delivery Sci Technol. 2018;48:328–37.
Crowley KJ, Zografi G. Water vapor absorption into amorphous hydrophobic drug/poly(vinylpyrrolidone) dispersions. J Pharm Sci. 2002;91(10):2150–65.
Li M, Xu W, Su Y. Solid-state NMR spectroscopy in pharmaceutical sciences. Trends Analyt Chem. 2021;135:116152.
Mensink MA, Nethercott MJ, Hinrichs WLJ, van der Voort MK, Frijlink HW, Munson EJ, et al. Influence of miscibility of protein-sugar lyophilizates on their storage stability. AAPS J. 2016;18(5):1225–32.
Bovey FA, Mirau PA. The solid-state NMR of polymers. In: Bovey FA, Mirau PA, editors. NMR of polymers. San Diego: Academic Press; 1996. p. 243–352.
Aso Y, Yoshioka S, Miyazaki T, Kawanishi T, Tanaka K, Kitamura S, et al. Miscibility of nifedipine and hydrophilic polymers as measured by 1H-NMR spin-lattice relaxation. Chem Pharm Bull. 2007;55(8):1227–31.
Yuan X, Sperger D, Munson EJ. Investigating miscibility and molecular mobility of nifedipine-PVP amorphous solid dispersions using solid-state NMR spectroscopy. Mol Pharm. 2014;11(1):329–37.
Ni QZ, Yang F, Can TV, Sergeyev IV, D’Addio SM, Jawla SK, et al. In situ characterization of pharmaceutical formulations by dynamic nuclear polarization enhanced mas NMR. J Phys Chem B. 2017;121(34):8132–41.
Li N, Gilpin CJ, Taylor LS. Understanding the impact of water on the miscibility and microstructure of amorphous solid dispersions: an AFM–LCR and TEM–EDX study. Mol Pharm. 2017;14(5):1691–705.
Rumondor AF, Stanford L, Taylor L. Effects of polymer type and storage relative humidity on the kinetics of felodipine crystallization from amorphous solid dispersions. Pharm Res. 2009;26(12):2599–606.
Rumondor ACF, Marsac PJ, Stanford LA, Taylor LS. Phase behavior of poly(vinylpyrrolidone) containing amorphous solid dispersions in the presence of moisture. Mol Pharm. 2009;6(5):1492–505.
DiNunzio JC, Miller DA, Yang W, McGinity JW, Williams RO. Amorphous compositions using concentration enhancing polymers for improved bioavailability of itraconazole. Mol Pharm. 2008;5(6):968–80.
Jermain SV, Lowinger MB, Ellenberger DJ, Miller DA, Su Y, Williams RO. In vitro and in vivo behaviors of kinetisol and spray-dried amorphous solid dispersions of a weakly basic drug and ionic polymer. Mol Pharm. 2020;17(8):2789–808.
Indulkar AS, Lou X, Zhang GGZ, Taylor LS. Insights into the dissolution mechanism of ritonavir–copovidone amorphous solid dispersions: importance of congruent release for enhanced performance. Mol Pharm. 2019;16(3):1327–39.
Moseson DE, Parker AS, Beaudoin SP, Taylor LS. Amorphous solid dispersions containing residual crystallinity: Influence of seed properties and polymer adsorption on dissolution performance. Eur J Pharm Sci. 2020;146:105276.
Hamed R, Awadallah A, Sunoqrot S, Tarawneh O, Nazzal S, AlBaraghthi T, et al. pH-Dependent solubility and dissolution behavior of carvedilol—case example of a weakly basic BCS class II drug. AAPS PharmSciTech. 2016;17(2):418–26.
Kataoka M, Fukahori M, Ikemura A, Kubota A, Higashino H, Sakuma S, et al. Effects of gastric pH on oral drug absorption: In vitro assessment using a dissolution/permeation system reflecting the gastric dissolution process. Eur J Pharm Biopharm. 2016;101:103–11.
Mitra A, Kesisoglou F, Beauchamp M, Zhu W, Chiti F, Wu Y. Using absorption simulation and gastric ph modulated dog model for formulation development to overcome achlorhydria effect. Mol Pharm. 2011;8(6):2216–23.
Rubbens J, Brouwers J, Tack J, Augustijns P. Gastrointestinal dissolution, supersaturation and precipitation of the weak base indinavir in healthy volunteers. Eur J Pharm Biopharm. 2016;109:122–9.
Shono Y, Jantratid E, Dressman JB. Precipitation in the small intestine may play a more important role in the in vivo performance of poorly soluble weak bases in the fasted state: Case example nelfinavir. Eur J Pharm Biopharm. 2011;79(2):349–56.
Taniguchi C, Kawabata Y, Wada K, Yamada S, Onoue S. Microenvironmental pH-modification to improve dissolution behavior and oral absorption for drugs with pH-dependent solubility. Expert Opin Drug Deliv. 2014;11(4):505–16.
Derendorf H, VanderMaelen CP, Brickl R-S, MacGregor TR, Eisert W. Dipyridamole bioavailability in subjects with reduced gastric acidity. J Clin Pharmacol. 2005;45(7):845–50.
Hens B, Tsume Y, Bermejo M, Paixao P, Koenigsknecht MJ, Baker JR, et al. Low buffer capacity and alternating motility along the human gastrointestinal tract: implications for in vivo dissolution and absorption of ionizable drugs. Mol Pharm. 2017;14(12):4281–94.
Shim JB, Lee JK, Jo H, Hwang JH, Jeong SM, Jo JI, et al. Effect of acidifier on the dissolution property of a solid dispersion of raloxifene HCl. Macromol Res. 2013;21(1):42–8.
Tran TT-D, Tran PH-L, Choi H-G, Han H-K, Lee B-J. The roles of acidifiers in solid dispersions and physical mixtures. Int J Pharm. 2010;384(1):60–6.
Espinoza R, Hong E, Villafuerte L. Influence of admixed citric acid on the release profile of pelanserin hydrochloride from HPMC matrix tablets. Int J Pharm. 2000;201(2):165–73.
Siepe S, Herrmann W, Borchert H-H, Lueckel B, Kramer A, Ries A, et al. Microenvironmental pH and microviscosity inside pH-controlled matrix tablets: An EPR imaging study. J Control Release. 2006;112(1):72–8.
Siepe S, Lueckel B, Kramer A, Ries A, Gurny R. Assessment of tailor-made HPMC-based matrix minitablets comprising a weakly basic drug compound. Drug Dev Ind Pharm. 2008;34(1):46–52.
Siepe S, Lueckel B, Kramer A, Ries A, Gurny R. Strategies for the design of hydrophilic matrix tablets with controlled microenvironmental pH. Int J Pharm. 2006;316(1):14–20.
Choi J-S, Kwon S-H, Lee S-E, Jang WS, Byeon JC, Jeong HM, et al. Use of acidifier and solubilizer in tadalafil solid dispersion to enhance the in vitro dissolution and oral bioavailability in rats. Int J Pharm. 2017;526(1):77–87.
Monschke M, Wagner KG. Amorphous solid dispersions of weak bases with pH-dependent soluble polymers to overcome limited bioavailability due to gastric pH variability – An in-vitro approach. Int J Pharm. 2019;564:162–70.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors gratefully thank Crystal Pharmatech Inc. for supporting this study. We also express thanks to the University of Connecticut for providing startup funding to support AN. The authors declare no competing financial interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editor: Vivek Gupta.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, B., Nethercott, M.J., Narula, A. et al. pH-Dependent supersaturation from amorphous solid dispersions of weakly basic drugs. Pharm Res 39, 2919–2936 (2022). https://doi.org/10.1007/s11095-021-03147-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-021-03147-0