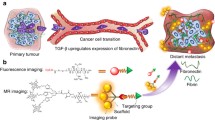
Abstract
Purpose
To investigate the effectiveness of targeted ECO/miR-200c in modulating tumor microenvironment and treating triple negative breast cancer (TNBC) using non-invasive magnetic resonance molecular imaging (MRMI) of extradomain B fibronectin (EDB-FN) with a targeted MRI contrast agent.
Methods
MDA-MB-231 and Hs578T TNBC cells were transfected with RGD-PEG-ECO/miR-200c. Invasive and migratory potential was evaluated using transwell, scratch wound, and spheroid formation assays. Athymic nude mice bearing orthotopic MDA-MB-231 and Hs578T xenografts were treated with weekly i.v. injection of RGD-PEG-ECO/miR-200c nanoparticles at 1.0 mg/kg/week RNA for 6 weeks. MRMI of EDB-FN was performed using a targeted contrast agent MT218 [ZD2-N3-Gd(DO3A)] on a 3 T MRS 3000 scanner. T1-weighted images were acquired following intravenous injection of MT218 at dose of 0.1 mmol/kg using a fast spin echo axial sequence with respiratory gating.
Results
Systemic administration of RGD-PEG-ECO/miR-200c nanoparticles in mice bearing orthotopic TNBC xenografts significantly suppressed tumor progression without toxic side-effects. MRMI with MT218 revealed that the treatment significantly suppressed tumor proliferation as compared to the control. MRMI also showed that the miR-200c treatment altered tumor microenvironment by reducing EDB-FN expression, as evidenced by decreased contrast enhancement in both MDA-MB-231 and Hs578T tumors. The reduction of EDB-FN was confirmed by immunohistochemistry.
Conclusions
Targeted delivery of miR-200c with RGD-PEG-ECO/miR-200c nanoparticles effectively modulates tumor microenvironment and suppresses TNBC proliferation in animal models. MRMI of tumor EDB-FN expression is effective to non-invasively monitor tumor response and therapeutic efficacy of RGD-PEG-ECO/miR-200c nanoparticles in TNBC.







Similar content being viewed by others
References
Gao D, Vahdat LT, Wong S, Chang JC, Mittal V. Microenvironmental regulation of epithelial-mesenchymal transitions in cancer. Cancer Res. 2012;72(19):4883–9.
Felipe Lima J, Nofech-Mozes S, Bayani J, Bartlett JM. EMT in Breast Carcinoma-A Review. J Clin Med. 2016;5(7):65.
Majumder S, Jacob ST. Emerging role of MicroRNAs in drug-resistant breast Cancer. Gene Expr. 2018;15(3):141–51.
Piasecka D, Braun M, Kordek R, Sadej R, Romanska H. MicroRNAs in regulation of triple-negative breast cancer progression. J Cancer Res Clin Oncol. 2018;144(8):1401–11.
Shimono Y, Zabala M, Cho RW, Lobo N, Dalerba P, Qian D, et al. Downregulation of miRNA-200c links breast cancer stem cells with normal stem cells. Cell. 2009;138(3):592–603.
Knezevic J, Pfefferle AD, Petrovic I, Greene SB, Perou CM, Rosen JM. Expression of miR-200c in claudin-low breast cancer alters stem cell functionality, enhances chemosensitivity and reduces metastatic potential. Oncogene. 2015;34(49):5997–6006.
Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G, et al. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol. 2008;10(5):593–601.
Li Y, VandenBoom TG, Kong D, Wang Z, Ali S, Philip PA, et al. Up-regulation of miR-200 and let-7 by natural agents leads to the reversal of epithelial-mesenchymal transition in gemcitabine-resistant pancreatic cancer cells. Cancer Res. 2009;69(16):6704–12.
Howe EN, Cochrane DR, Richer JK. Targets of miR-200c mediate suppression of cell motility and anoikis resistance. Breast Cancer Research : BCR. 2011;13(2):R45.
Mutlu M, Raza U, Saatci Ö, Eyüpoğlu E, Yurdusev E, Şahin Ö. miR-200c: a versatile watchdog in cancer progression, EMT, and drug resistance. J Mol Med. 2016;94(6):629–44.
Si W, Shen J, Zheng H, Fan W. The role and mechanisms of action of microRNAs in cancer drug resistance. Clin Epigenetics. 2019;11:25.
Kopp F, Oak PS, Wagner E, Roidl A. miR-200c Sensitizes Breast Cancer Cells to Doxorubicin Treatment by Decreasing TrkB and Bmi1 Expression. PLoS One. 2012;7(11):e50469.
Hurteau GJ, Carlson JA, Spivack SD, Brock GJ. Overexpression of the MicroRNA hsa-miR-200c leads to reduced expression of transcription factor 8 and increased expression of E-cadherin. Cancer Res. 2007;67(17):7972–6.
Cochrane DR, Howe EN, Spoelstra NS, Richer JK. Loss of miR-200c: a marker of aggressiveness and Chemoresistance in female reproductive cancers. J Oncol. 2010;2010:821717.
Cochrane DR, Spoelstra NS, Howe EN, Nordeen SK, Richer JK. MicroRNA-200c mitigates invasiveness and restores sensitivity to microtubule-targeting chemotherapeutic agents. Mol Cancer Ther. 2009;8(5):1055–66.
Jurmeister S, Baumann M, Balwierz A, Keklikoglou I, Ward A, Uhlmann S, et al. MicroRNA-200c represses migration and invasion of breast cancer cells by targeting actin-regulatory proteins FHOD1 and PPM1F. Mol Cell Biol. 2012;32(3):633–51.
Kawaguchi T, Yan L, Qi Q, Peng X, Gabriel EM, Young J, et al. Overexpression of suppressive microRNAs, miR-30a and miR-200c are associated with improved survival of breast cancer patients. Sci Rep. 2017;7(1):15945.
Xu F, He H, Huang W, Lin Y, Luo S, Du Q, et al. Decreased expression of MicroRNA-200 family in human breast cancer is associated with lymph node metastasis. Clin Transl Oncol. 2016;18(3):283–8.
Bordeleau F, Califano JP, Negron Abril YL, Mason BN, LaValley DJ, Shin SJ, et al. Tissue stiffness regulates serine/arginine-rich protein-mediated splicing of the extra domain B-fibronectin isoform in tumors. Proc Natl Acad Sci U S A. 2015;112(27):8314–9.
Nolan J, Mahdi AF, Dunne CP, Kiely PA. Collagen and fibronectin promote an aggressive cancer phenotype in breast cancer cells but drive autonomous gene expression patterns. Gene. 2020;761:145024.
Kumra H, Reinhardt DP. Fibronectin-targeted drug delivery in cancer. Adv Drug Deliv Rev. 2016;97:101–10.
Loridon-Rosa B, Vielh P, Matsuura H, Clausen H, Cuadrado C, Burtin P. Distribution of Oncofetal fibronectin in human mammary tumors: immunofluorescence study on histological sections. Cancer Res. 1990;50(5):1608–12.
Lyons AJ, Bateman AC, Spedding A, Primrose JN, Mandel U. Oncofetal fibronectin and oral squamous cell carcinoma. Br J Oral Maxillofac Surg. 2001;39(6):471–7.
Vaidya A, Wang H, Qian V, Gilmore H, Lu ZR. Overexpression of Extradomain-B Fibronectin is Associated with Invasion of Breast Cancer Cells. Cells. 2020;9(8):11127–43
Han Z, Wu X, Roelle S, Chen C, Schiemann WP, Lu Z-R. Targeted gadofullerene for sensitive magnetic resonance imaging and risk-stratification of breast cancer. Nat Commun. 2017;8(1):692.
Han Z, Zhou Z, Shi X, Wang J, Wu X, Sun D, et al. EDB fibronectin specific peptide for prostate Cancer targeting. Bioconjug Chem. 2015;26(5):830–8.
Han Z, Cheng H, Parvani JG, Zhou Z, Lu Z-R. Magnetic resonance molecular imaging of metastatic breast cancer by targeting extradomain-B fibronectin in the tumor microenvironment. Magn Reson Med. 2018;79(6):3135–43.
Ayat NR, Vaidya A, Yeung GA, Buford MN, Hall RC, Qiao PL, et al. Effective MR molecular imaging of triple negative breast Cancer with an EDB-fibronectin-specific contrast agent at reduced doses. Front Oncol. 2019;9:1351.
Vaidya A, Ayat N, Buford M, Wang H, Shankardass A, Zhao Y, et al. Noninvasive assessment and therapeutic monitoring of drug-resistant colorectal cancer by MR molecular imaging of extradomain-B fibronectin. Theranostics. 2020;10(24):11127–43.
Parvani JG, Gujrati MD, Mack MA, Schiemann WP, Lu Z-R. Silencing β3 integrin by targeted ECO/siRNA nanoparticles inhibits EMT and metastasis of triple negative breast Cancer. Cancer Res. 2015;75(11):2316–25.
Vaidya AM, Sun Z, Ayat N, Schilb A, Liu X, Jiang H, et al. Systemic delivery of tumor-targeting siRNA nanoparticles against an oncogenic LncRNA facilitates effective triple-negative breast Cancer therapy. Bioconjug Chem. 2019;30(3):907–19.
Ayat NR, Sun Z, Sun D, Yin M, Hall RC, Vaidya AM, et al. Formulation of biocompatible targeted ECO/siRNA nanoparticles with long-term stability for clinical translation of RNAi. Nucleic Acid Ther. 2019;29(4):195–207.
Malamas AS, Gujrati M, Kummitha CM, Xu R, Lu Z-R. Design and evaluation of new pH-sensitive amphiphilic cationic lipids for siRNA delivery. J Control Release. 2013;171(3):296–307.
Sun D, Sahu B, Gao S, Schur RM, Vaidya AM, Maeda A, et al. Targeted multifunctional lipid ECO plasmid DNA nanoparticles as efficient non-viral gene therapy for Leber’s congenital Amaurosis. Molecular Therapy - Nucleic Acids. 2017;7:42–52.
Kaspar M, Zardi L, Neri D. Fibronectin as target for tumor therapy. Int J Cancer. 2006;118(6):1331–9.
Lin TC, Yang CH, Cheng LH, Chang WT, Lin YR, Cheng HC. Fibronectin in Cancer: Friend or Foe. Cells. 2019;9(1):27.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schilb, A.L., Ayat, N.R., Vaidya, A.M. et al. Efficacy of Targeted ECO/miR-200c Nanoparticles for Modulating Tumor Microenvironment and Treating Triple Negative Breast Cancer as Non-invasively Monitored by MR Molecular Imaging. Pharm Res 38, 1405–1418 (2021). https://doi.org/10.1007/s11095-021-03083-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-021-03083-z