Abstract
Purpose
Available dry powder inhalers (DPIs) have very poor lung delivery efficiencies in children. The objective of this study was to advance and experimentally test a positive-pressure air-jet DPI for children based on the use of a vertical aerosolization chamber and new patient interfaces that contain a three-dimensional (3D) rod array structure.
Methods
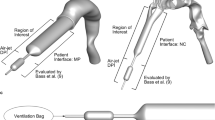
Aerosolization performance of different air-jet DPI designs was first evaluated based on a 10 mg powder fill mass of a spray-dried excipient enhanced growth (EEG) formulation. Devices were actuated with positive pressure using flow rate (10–20 L/min) and inhaled volume (750 ml) conditions consistent with a 5-year-old child. Devices with best performance were connected to different mouthpiece designs to determine the effect on aerosolization and tested for aerosol penetration through a realistic pediatric in vitro mouth-throat model.
Results
Use of the new vertical aerosolization chamber resulted in high quality aerosol formation. Inclusion of a 3D rod array structure in the mouthpiece further reduced aerosol size by approximately 20% compared to conditions without a rod array, and effectively dissipated the turbulent jet leaving the device. Best case device and mouthpiece combinations produced < 2% mouth-throat depositional loss and > 70% lung delivery efficiency based on loaded dose.
Conclusions
In conclusion, use of a 3D rod array in the MP of a positive-pressure air-jet DPI was found to reduce aerosol size by 20%, not significantly increase MP depositional loss, reduce mouth-throat deposition by 6.4-fold and enable lung delivery efficiency as high as 73.4% of loaded dose based on pediatric test conditions.







Similar content being viewed by others
Abbreviations
- 3D:
-
Three dimensional
- AAV:
-
Actuation air volume
- AS:
-
Albuterol sulfate
- CFD:
-
Computational fluid dynamics
- DPI:
-
Dry powder inhaler
- ED:
-
Emitted dose
- EEG:
-
Excipient enhanced growth
- FPF:
-
Fine particle fraction
- HPLC:
-
High performance liquid chromatography
- LPM:
-
Liters per minute
- MMAD:
-
Mass median aerodynamic diameter
- MP:
-
Mouthpiece
- MT:
-
Mouth-throat
- NGI:
-
Next Generation Impactor
- SD:
-
Standard deviation
References
Moon C, Smyth HD, Watts AB, Williams RO. Delivery technologies for orally inhaled products: an update. AAPS PharmSciTech. 2019;20:117.
De Boer A, Hagedoorn P, Hoppentocht M, Buttini F, Grasmeijer F, Frijlink H. Dry powder inhalation: past, present and future. Expert Opin Drug Deliv. 2017;14:499–512.
Weers JG, Son Y-J, Glusker M, Haynes A, Huang D, Kadrichu N, Le J, Li X, Malcolmson R, Miller DP, Tarara TE, Ung K, Clark A. Idealhalers versus realhalers: is it possible to bypass deposition in the upper respiratory tract? J Aerosol Med Pulm Drug Deliv. 2019;32:55–69.
Newman S. Respiratory drug delivery: essential theory and practice. Richmond: RDD Online; 2009.
Below A, Bickmann D, Breitkreutz J. Assessing the performance of two dry powder inhalers in preschool children using an idealized pediatric upper airway model. Int J Pharm. 2013;444:169–74.
Devadason S, Everard M, MacEarlan C, Roller C, Summers Q, Swift P, Borgstrom L, Le Souëf P. Lung deposition from the Turbuhaler in children with cystic fibrosis. Eur Respir J. 1997;10:2023–8.
Lindert S, Below A, Breitkreutz J. Performance of dry powder inhalers with single dosed capsules in preschool children and adults using improved upper airway models. Pharmaceutics. 2014;6:36–51.
Ruzycki CA, Golshahi L, Vehring R, Finlay WH. Comparison of in vitro deposition of pharmaceutical aerosols in an idealized child throat with in vivo deposition in the upper respiratory tract of children. Pharm Res. 2014;31:1525–35.
Pedersen S. How to use a rotahaler. Arch Dis Child. 1986;61:11–4.
Pedersen S, Hansen O, Fuglsang G. Influence of inspiratory flow rate upon the effect of a Turbuhaler. Arch Dis Child. 1990;65:308–10.
Lexmond AJ, Kruizinga TJ, Hagedoorn P, Rottier BL, Frijlink HW, De Boer AH. Effect of inhaler design variables on paediatric use of dry powder inhalers. PLoS One. 2014;9:e99304.
Farkas D, Hindle M, Bonasera S, Bass K, Longest W. Development of an inline dry powder inhaler for oral or trans-nasal aerosol administration to children. J Aerosol Med Pulm Drug Deliv. 2019; https://doi.org/10.1089/jamp.2019.1540.
Farkas D, Hindle M, Longest PW. Development of an inline dry powder inhaler that requires low air volume. J Aerosol Med Pulm Drug Deliv. 2018;31:255–65.
Farkas D, Hindle M, Longest PW. Application of an inline dry powder inhaler to deliver high dose pharmaceutical aerosols during low flow nasal cannula therapy. Int J Pharm. 2018;546:1–9.
Farkas D, Hindle M, Longest PW. Efficient nose-to-lung aerosol delivery with an inline DPI requiring low actuation air volume. Pharm Res. 2018;35:194.
Everard ML, Stammers J, Hardy JG, Milner AD. New aerosol delivery system for neonatal ventilator circuits. Arch Dis Child. 1992;67:826–30.
Behara SRB, Longest PW, Farkas DR, Hindle M. Development of high efficiency ventilation bag actuated dry powder inhalers. Int J Pharm. 2014;465:52–62.
Feng B, Tang P, Leung SSY, Dhanani J, Chan H-K. A novel in-line delivery system to administer dry powder mannitol to mechanically ventilated patients. J Aerosol Med Pulm Drug Deliv. 2017;30:100–7.
Okuda T, Tang P, Yu J, Finlay WH, Chan H-K. Powder aerosol delivery through nasal high-flow system: in vitro feasibility and influence of process conditions. Int J Pharm. 2017;533:187–97.
Boc ST, Farkas DR, Longest PW, Hindle M. Aerosolization of spray dried pulmonary surfactant powder using a novel low air volume actuated dry powder inhaler. Respir Drug Deliv 2018. 2018;2:639–42.
Longest W, Farkas D. Development of a new inhaler for high-efficiency dispersion of spray-dried powders using computational fluid dynamics (CFD) modeling. AAPS J. 2019;21:25.
Son Y-J, Longest PW, Hindle M. Aerosolization characteristics of dry powder inhaler formulations for the excipient enhanced growth (EEG) application: effect of spray drying process conditions on aerosol performance. Int J Pharm. 2013;443:137–45.
Longest W, Farkas D, Bass K, Hindle M. Use of computational fluid dynamics (CFD) dispersion parameters in the development of a new DPI actuated with low air volumes. Pharm Res. 2019;36:110.
Bass K, Farkas D, Longest W. Optimizing aerosolization using computational fluid dynamics in a pediatric air-jet dry powder inhaler. AAPS PharmSciTech. 2019;20:329.
Longest PW, Hindle M, Das Choudhuri S, Xi J. Comparison of ambient and spray aerosol deposition in a standard induction port and more realistic mouth-throat geometry. J Aerosol Sci. 2008;39:572–91.
Longest PW, Tian G, Walenga RL, Hindle M. Comparing MDI and DPI aerosol deposition using in vitro experiments and a new stochastic individual path (SIP) model of the conducting airways. Pharm Res. 2012;29:1670–88.
Tian G, Longest PW, Su G, Walenga RL, Hindle M. Development of a stochastic individual path (SIP) model for predicting the tracheobronchial deposition of pharmaceutical aerosols: effects of transient inhalation and sampling the airways. J Aerosol Sci. 2011;42:781–99.
DeHaan WH, Finlay WH. In vitro monodisperse aerosol deposition in a mouth and throat with six different inhalation devices. J Aerosol Med. 2001;14:361–7.
DeHaan WH, Finlay WH. Predicting extrathoracic deposition from dry powder inhalers. J Aerosol Sci. 2004;35:309–31.
Ilie M, Matida EA, Finlay WH. Asymmetrical aerosol deposition in an idealized mouth with a DPI mouthpiece inlet. Aerosol Sci Technol. 2008;42:10–7.
Matida EA, DeHaan WH, Finlay WH, Lange CF. Simulation of particle deposition in an idealized mouth with different small diameter inlets. Aerosol Sci Technol. 2003;37:924–32.
Bass K, Longest PW. Development of DPI patient interfaces for improved aerosol delivery to children. AAPS PharmSciTech. 2020; https://doi.org/10.1208/s12249-020-01667-3.
Longest PW, Son Y-J, Holbrook LT, Hindle M. Aerodynamic factors responsible for the deaggregation of carrier-free drug powders to form micrometer and submicrometer aerosols. Pharm Res. 2013;30:1608–27.
Phalen RF, Oldham MJ, Beaucage CB, Crocker TT, Mortensen JD. Postnatal enlargement of human tracheobronchial airways and implications for particle deposition. Anat Rec. 1985;212:368–80.
Golshahi L, Finlay WH. An idealized child throat that mimics average pediatric oropharyngeal deposition. Aerosol Sci Technol. 2012;46:i–iv.
Wachtel H, Bickmann D, Breitkreutz J, Langguth P. Can pediatric throat models and air flow profiles improve our dose finding strategy. Respir Drug Deliv. 2010;2010:195–204.
Cheng YS. Aerosol deposition in the extrathoracic region. Aerosol Sci Technol. 2003;37:659–71.
Cheng YS, Zhou Y, Chen BT. Particle deposition in a cast of human oral airways. Aerosol Sci Technol. 1999;31:286–300.
Xi J, Longest PW. Transport and deposition of micro-aerosols in realistic and simplified models of the oral airway. Ann Biomed Eng. 2007;35:560–81.
ICRP. Human respiratory tract model for radiological protection. New York: Elsevier Science Ltd.; 1994.
Farkas D, Hindle M, Bonasera S, Bass K, Longest W. Development of an inline dry powder inhaler for oral or trans-nasal aerosol administration to children. J Aerosol Med Pulm Drug Deliv. 2019;
Behara SRB, Farkas DR, Hindle M, Longest PW. Development of a high efficiency dry powder inhaler: effects of capsule chamber design and inhaler surface modifications. Pharm Res. 2014;31:360–72.
Son Y-J, Longest PW, Tian G, Hindle M. Evaluation and modification of commercial dry powder inhalers for the aerosolization of submicrometer excipient enhanced growth (EEG) formulation. Eur J Pharm Sci. 2013;49:390–9.
Bass K, Boc S, Hindle M, Dodson K, Longest PW. High-efficiency nose-to-lung aerosol delivery in an infant: development of a validated computational fluid dynamics method. J Aerosol Med Pulm Drug Deliv. 2019; https://doi.org/10.1089/jamp.2018.1490.
Bass K, Longest PW. Recommendations for simulating microparticle deposition at conditions similar to the upper airways with two-equation turbulence models. J Aerosol Sci. 2018;119:31–50.
Longest PW, Xi J. Effectiveness of direct Lagrangian tracking models for simulating nanoparticle deposition in the upper airways. Aerosol Sci Technol. 2007;41:380–97.
Matida EA, Finlay WH, Grgic LB. Improved numerical simulation of aerosol deposition in an idealized mouth-throat. J Aerosol Sci. 2004;35:1–19.
Wang Y, James PW. On the effect of anisotropy on the turbulent dispersion and deposition of small particles. Int J Multiphase Flow. 1999;22:551–8.
Roache PJ. Perspective: a method for uniform reporting of grid refinement studies. J Fluid Eng-T Asme. 1994;116:405–13.
Longest PW, Hindle M, Das Choudhuri S, Byron PR. Numerical simulations of capillary aerosol generation: CFD model development and comparisons with experimental data. Aerosol Sci Technol. 2007;41:952–73.
Longest PW, Vinchurkar S, Martonen TB. Transport and deposition of respiratory aerosols in models of childhood asthma. J Aerosol Sci. 2006;37:1234–57.
Farkas DR, Hindle M, Longest PW. Characterization of a new high-dose dry powder inhaler (DPI) based on a fluidized bed design. Ann Biomed Eng. 2015;43:2804–15.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Farkas, D., Bonasera, S., Bass, K. et al. Advancement of a Positive-Pressure Dry Powder Inhaler for Children: Use of a Vertical Aerosolization Chamber and Three-Dimensional Rod Array Interface. Pharm Res 37, 177 (2020). https://doi.org/10.1007/s11095-020-02889-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-020-02889-7