Abstract
Purpose
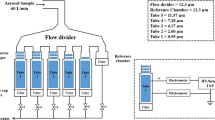
To measure the charge to mass (Q/M) ratios of the impactor stage masses (ISM) from commercial Flixotide™ 250 μg Evohaler, containing fluticasone propionate (FP), Serevent™ 25 μg Evohaler, containing salmeterol xinafoate (SX), and a combination Seretide™ 250/25 μg (FP/SX) Evohaler metered dose inhalers (MDIs). Measurements were performed with a purpose built bipolar charge measurement apparatus (bp-NGI) based on an electrostatic precipitator, which was directly connected below Stage 2 of a Next Generation Impactor (NGI).
Methods
Five successive shots of the respective MDIs were actuated through the bp-NGI. The whole ISM doses were electrostatically precipitated to determine their negative, positive and net Q/m ratios.
Results
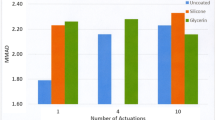
The ISM doses collected in the bp-NGI were shown to be equivalent to those collected in a standard NGI. FP particles, actuated from Flixotide™ and Seretide™ MDIs, exhibited greater quantities of negatively charged particles than positive. However, the Q/m ratios of the positively charged particles were greater in magnitude. SX particles from Serevent™ exhibited a greater quantity of positively charged particles whereas SX aerosol particles from Seretide™ exhibited a greater quantity of negatively charged particles. The Q/m ratio of the negatively charged SX particles in Serevent™ was greater in magnitude than the positively charged particles.
Conclusions
The bp-NGI was used to quantify the bipolar Q/m ratios of aerosol particles collected from the ISMs of commercial MDI products. The positive charge recorded for each of the three MDIs may have been enhanced by the presence of charged ice crystals formed from the propellant during the aerosolisation process.











Similar content being viewed by others
Abbreviations
- API:
-
Active pharmaceutical ingredient
- BNC:
-
Bayonet Neill-Concelman
- BOLAR:
-
Bipolar charge analyser
- bp-NGI:
-
Bipolar next generation impactor
- DC:
-
Direct current
- ED:
-
Emitted dose
- ELPI:
-
Electrical low pressure impactor
- ESPART:
-
Electrical single particle aerodynamic relaxation time
- FP:
-
Fluticasone propionate
- HFA:
-
Hydrofluroalkane
- HPLC:
-
High performance liquid chromatography
- ISM:
-
Impactor stage mass
- NGI:
-
Next generation impactor
- pMDI:
-
Pressurized metered dose inhaler
- RD:
-
Recovered dose
- SX:
-
Salmeterol xinafoate
References
Murata S, Izumi T, Ito H. Effect of the moisture content in aerosol on the spray performance of Stmerin ® D hydrofluoroalkane preparations (2). Chem Pharm Bull (Tokyo). 2012;60(5):593–7.
James J, Davies M, Toon R, Jinks P, Roberts CJ. Particulate drug interactions with polymeric and elastomeric valve components in suspension formulations for metered dose inhalers. Int J Pharm. 2009;366(1–2):124–32.
Kwetkus BA. Particle Triboelectrification and its use in the electrostatic separation process. Part Sci Technol. 1998;16(1):55–68.
Lowell J. The role of material transfer in contact electrification. J Phys D. 1977;10:L233–L5.
Matsusaka S, Maruyama H, Matsuyama T, Ghadiri M. Triboelectric charging of powders: a review. Chem Eng Sci. 2010;65(22):5781–807.
Kulphaisal P Peart J, Byron P, editors. (2002) Influence of water on electrical properties in hydrofluoroalkane based metered dose inhalers. Respiratory Drug Delivery VIII; Raleigh, NC: Davis Horwood International.
Ali M, Mazumder MK, Martonen TB. Measurements of electrodynamic effects on the deposition of MDI and DPI aerosols in a replica cast of human Oral-pharyngeal-laryngeal airways. Journal of Aerosol Medicine and Pulmonary Drug Delivery. 2009;22(1):35–44.
Keskinen J, Pietarinen K, Lehtimaki M. Electrical low pressure impactor. J Aerosol Sci. 1992;23(4):353–60.
Glover W, Chan H-K. Electrostatic charge characterization of pharmaceutical aerosols using electrical low-pressure impaction (ELPI). J Aerosol Sci. 2004;35(6):755–64.
Kwok P, Glover W, Chan HK. Electrostatic charge characteristics of aerosols produced from metered dose inhalers. J Pharm Sci. 2005;94(12):2789–99.
Hoe S, Young P, Chan H-K, Traini D. Introduction of the electrical next generation impactor (eNGI) and investigation of its capabilities for the study of pressurized metered dose inhalers. Pharm Res. 2009;26(2):431–7.
Hoe S. The influence of flow rate on the aerosol deposition profile and ElectrostaticCharge of single and combination metered dose inhalers. Pharm Res. 2009;26(12):2639–46.
Mizuno A. Electrostatic precipitation. IEEE Trans Dielectr Electr Insul. 2000;7(5):615–24.
O'Leary M, Balachandran W, Rogueda P, Chambers F. The bipolar nature of charge resident on supposedly unipolar aerosols. J Phys Conf Ser. 2008;142:012022.
Kulon J, Balachandran W. The measurement of bipolar charge on aerosols. J Electrost. 2001;51-52(0):552–7.
Yli-Ojanpera J, Ukkonen A, Jarvinen A, Layzell S, Niemela V, Keskinen J. Bipolar charge analyzer (BOLAR): a new aerosol instrument for bipolar charge measurements. J Aerosol Sci. 2014;77:16–30.
Wong J, Lin Y-W, Kwok PCL, Niemela V, Crapper J, Chan H-K. Measuring bipolar charge and mass distributions of powder aerosols by a novel tool (BOLAR). Mol Pharm. 2015;12(9):3433–40.
Wong J, Kwok PCL, Niemela V, Heng D, Crapper J, Chan H-K. Bipolar electrostatic charge and mass distributions of powder aerosols - effects of inhaler design and inhaler material. J Aerosol Sci. 2016;95:104–17.
Leung SSY, Chiow ACM, Ukkonen A, Chan H-K. Applicability of bipolar charge analyzer (BOLAR) in characterizing the bipolar electrostatic charge profile of commercial metered dose inhalers (MDIs). Pharm Res. 2016;33(2):283–91.
Leung SSY, Chiow ACM, Kwok PCL, Ukkonen A, Chan H-K. Effect of spacers on the bipolar electrostatic charge properties of metered dose inhaler aerosols-a case study with Tilade. J Pharm Sci. 2017;106(6):1553–9.
Marple VA, Roberts DL, Romay FJ, Miller NC, Truman KG, Van Oort M, et al. Next generation pharmaceutical impactor (a new impactor for pharmaceutical inhaler testing). Part I: design. Journal of Aerosol Medicine. 2003;16(3):283–99.
Kulon J, Hrabar S, Machowski W, Balachandran W. A bipolar charge measurement system for aerosol characterization. IEEE Trans Ind Appl. 2001;37(2):472–9.
Murnane D, Martin GP, Marriott C. Validation of a reverse-phase high performance liquid chromatographic method for concurrent assay of a weak base (salmeterol xinafoate) and a pharmacologically active steroid (fluticasone propionate). J Pharm Biomed Anal. 2006;40:1149–54.
Chow KT, Zhu K, Tan RBH, Heng PWS. Investigation of electrostatic behavior of a lactose carrier for dry powder inhalers. Pharm Res. 2008;25(12):2822–34.
Peart J, Kulphaisal P, Orban JC. Relevance of electrostatics in respiratory drug delivery. Business Briefing: Pharmagenerics. 2003:1–4.
Kwok P, Noakes T, Chan H-K. Effect of moisture on the electrostatic charge properties of metered dose inhaler aerosols. J Aerosol Sci. 2008;39(3):211–26.
Theophilus A, Moore A, Prime D, Rossomanno S, Whitcher B, Chrystyn H. Co-deposition of salmeterol and fluticasone propionate by a combination inhaler. Int J Pharm. 2006;313(1–2):14–22.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rowland, M., Cavecchi, A., Thielmann, F. et al. Measuring The Bipolar Charge Distributions of Fine Particle Aerosol Clouds of Commercial PMDI Suspensions Using a Bipolar Next Generation Impactor (bp-NGI). Pharm Res 36, 15 (2019). https://doi.org/10.1007/s11095-018-2544-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-018-2544-9