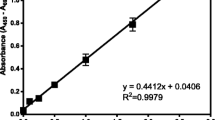
Abstract
Purpose
Tungsten and tungsten oxide leachates found in glass pre-filled syringes were identified to initiate protein precipitation and aggregation. Here, we tested the possibility of tungsten and tungsten oxide to induce the chemical degradation of proteins via reaction with hydrogen peroxide, a possible impurity present in protein formulations, to yield peroxotungstate.
Methods
A monoclonal antibody (mAb) was incubated with various concentrations of peroxotungstate and the reaction mixtures analyzed by SDS-PAGE and mass spectrometry.
Results
Exposure of a mAb to 1.07–1070 ppm peroxotungstate (based on tungsten content) at temperatures of 4°C and 22°C (pH 5–7) induced protein fragmentation. The extent of fragmentation increased with higher temperatures, lower pH and higher peroxotungstate concentrations. The mAb fragments were identified to contain different combinations of heavy chains (H) and light chains (L). Analogous mAb fragments were generated when the protein was exposed to H2O2 and orthotungstate at levels as low as 5 ppm. In addition, extracts from tungsten pins used to manufacture glass pre-filled syringes, in combination with H2O2 caused comparable fragmentation of the mAb. Mass spectrometric identification of the fragments suggests fragment generation by oxidative disulfide bond cleavage between the heavy and light chains, confirmed by mass spectrometry data on product formation. The mechanism of oxidative fragmentation was separately confirmed with insulin.
Conclusion
Fragmentation of the mAb by peroxotungstate is proposed to occur through inter-chain disulfide bond oxidation to form thiosulfinate (CyS(═O)SCy) and thiosulfonate [CyS(═O)2SCy], followed by hydrolysis.













Similar content being viewed by others
Notes
The actual value given in the paper is ca. 250 mEq, but was expressed per kg of neat PS80 by multiplication by 5 (35). Therefore, the peroxide levels in 20% PS80 can be computed to be equivalent to ca. 50 mEq.
Abbreviations
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- mAb:
-
Monoclonal antibody
- MS:
-
Mass spectrometry
- SDS:
-
Sodium dodecyl sulfate
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- UPLC:
-
Ultra performance liquid chromatography
- VHP:
-
Vaporized hydrogen peroxide
- WO4 2− :
-
Orthotungstate
References
Visiongain. Global biologics market, industry and R&D: Forecasts 2015–2025. Available from: https://www.visiongain.com/Report/1485/Global-Biologics-Market-Industry-and-R-D-Forecasts-2015-2025.
Adler M. INJECTABLES-challenges in the development of pre-filled syringes for biologics from a formulation Scientist's point of view. Am Pharm Rev. 2012;15(1):96.
Jiang Y, Nashed-Samuel Y, Li C, Liu W, Pollastrini J, Mallard D, et al. Tungsten-induced protein aggregation: solution behavior. J Pharm Sci. 2009;98(12):4695–710.
Bee JS, Nelson SA, Freund E, Carpenter JF, Randolph TW. Precipitation of a monoclonal antibody by soluble tungsten. J Pharm Sci. 2009;98(9):3290–301.
Liu W, Swift R, Torraca G, Nashed-Samuel Y, Wen ZQ, Jiang Y, et al. Root cause analysis of tungsten-induced protein aggregation in pre-filled syringes. PDA J Pharm Sci Technol. 2010;64(1):11–9.
Ronk M, Lee H, Fujimori K, Yeh P, Nashed-Samuel Y. Characterization of protein aggregating Tungstates: electrospray mass spectrometry analysis of extracts from prefilled syringes and from tungsten pins used in the manufacture of syringes. PDA J Pharm Sci Technol. 2016;70(1):51–61.
Rosenberg AS. Effects of protein aggregates: an immunologic perspective. AAPS J. 2006;8(3):E501–7.
Seidl A, Hainzl O, Richter M, Fischer R, Bohm S, Deutel B, et al. Tungsten-induced denaturation and aggregation of epoetin alfa during primary packaging as a cause of immunogenicity. Pharm Res. 2012;29(6):1454–67.
Markovic I. Challenges associated with extractable and/or leachable substances in therapeutic biologic protein products. Am Pharm Rev. 2006;9(6):20–7.
Sacha G, Rogers JA, Miller RL. Pre-filled syringes: a review of the history, manufacturing and challenges. Pharm Dev Technol. 2015;20(1):1–11.
Kishore RS, Pappenberger A, Dauphin IB, Ross A, Buergi B, Staempfli A, et al. Degradation of polysorbates 20 and 80: studies on thermal autoxidation and hydrolysis. J Pharm Sci. 2011;100(2):721–31.
Agalloco JP, Akers JE. Overcoming limitations of vaporized hydrogen peroxide. Pharm Technol. 2013;37(9):46–56.
Ishimoto R, Kamata K, Mizuno N. A highly active protonated tetranuclear peroxotungstate for oxidation with hydrogen peroxide. Angew Chem Int Ed Engl. 2012;51(19):4662–5.
Kudo T, Okamoto H, Matsumoto K, Sasaki Y. Peroxopolytungstic acids synthesized by direct reaction of tungsten or tungsten carbide with hydrogen peroxide. Inorg Chim Acta. 1986;111(2):L27–8.
Nakajima H, Kudo T, Mizuno N. Reaction of metal, carbide, and nitride of tungsten with hydrogen peroxide characterized by 183W nuclear magnetic resonance and raman spectroscopy. Chem Mater. 1999;11(3):691–7.
Okamoto A, Sugizaki K, Nakamura A, Yanagisawa H, Ikeda S. 5-Hydroxymethylcytosine-selective oxidation with peroxotungstate. Chem Commun. 2011;47(40):11231–3.
Shi X-Y, Wei J-F. Selective oxidation of sulfide catalyzed by peroxotungstate immobilized on ionic liquid-modified silica with aqueous hydrogen peroxide. J Mol Catal A Chem. 2008;280(1–2):142–7.
Usui Y, Sato K. A green method of adipic acid synthesis: organic solvent-and halide-free oxidation of cycloalkanones with 30% hydrogen peroxide. Green Chem. 2003;5(4):373–5.
Howarth OW. Oxygen-17 NMR study of aqueous peroxotungstates. Dalton Trans. 2004;(3):476–81.
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680–5.
Lassner E, Schubert W-D. Properties, chemistry, Technology of the Element, alloys, and chemical compounds. Vienna University of Technology, Vienna, Austria, Kluwer 1999:124–125.
Scientific T-F. NanoDrop 2000/2000c Protein Assay Guide (T032 - Technical bulletin. Available from: https://assets.thermofisher.com/TFS-Assets/CAD/manuals/NanoDrop-2000-User-Manual-EN.pdf.
Shevchenko A, Tomas H, Havli J, Olsen JV, Mann M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat Protoc. 2006;1(6):2856.
Saphire EO, Parren PW, Pantophlet R, Zwick MB, Morris GM, Rudd PM, et al. Crystal structure of a neutralizing human IGG against HIV-1: a template for vaccine design. Science. 2001;293(5532):1155–9.
Campbell NJ, Dengel AC, Edwards CJ, Griffith WP. Studies on transition-metal Peroxo complexes .8. The nature of Peroxomolybdates and Peroxotungstates in aqueous-solution. J Chem Soc Dalton Trans. 1989;(6):1203–8.
Kamata K, Yamaguchi K, Mizuno N. Highly selective, recyclable epoxidation of allylic alcohols with hydrogen peroxide in water catalyzed by dinuclear peroxotungstate. Chem Eur J. 2004;10(19):4728–34.
Kanner GS, Butt DP. Raman and electrochemical probes of the dissolution kinetics of tungsten in hydrogen peroxide. J Phys Chem A. 1998;102(47):9501–7.
Ross-Medgaarden EI, Wachs IE. Structural determination of bulk and surface tungsten oxides with UV-vis diffuse reflectance spectroscopy and Raman spectroscopy. J Phys Chem C. 2007;111(41):15089–99.
George B, Aruldhas G, Botto I. Vibrational spectra of sodium paratungstate 26 hydrate, Na 10 (H 2 W 12 O 42)·26H 2 O. J Mater Sci Lett. 1992;11(21):1421–3.
Zhu ZC, Chen Y, Ackerman MS, Wang B, Wu W, Li B, et al. Investigation of monoclonal antibody fragmentation artifacts in non-reducing SDS-PAGE. J Pharm Biomed Anal. 2013;83:89–95.
Fradkin AH, Mozziconacci O, Schoneich C, Carpenter JF, Randolph TW. UV photodegradation of murine growth hormone: chemical analysis and immunogenicity consequences. Eur J Pharm Biopharm. 2014;87(2):395–402.
Sreedhara A, Lau K, Li C, Hosken B, Macchi F, Zhan D, et al. Role of surface exposed tryptophan as substrate generators for the antibody catalyzed water oxidation pathway. Mol Pharm. 2013;10(1):278–88.
Ghiron AF, Thompson RC. Kinetic study of the oxygen-transfer reactions from the oxo diperoxo complexes of molybdenum (VI) and tungsten (VI) to (thiolato)-and (sulfenato) cobalt (III) complexes. Inorg Chem. 1988;27(26):4766–71.
Nagy P, Ashby MT. Reactive sulfur species: kinetics and mechanism of the hydrolysis of cysteine thiosulfinate ester. Chem Res Toxicol. 2007;20(9):1364–72.
Ha E, Wang W, Wang YJ. Peroxide formation in polysorbate 80 and protein stability. J Pharm Sci. 2002;91(10):2252–64.
Wasylaschuk WR, Harmon PA, Wagner G, Harman AB, Templeton AC, Xu H, et al. Evaluation of hydroperoxides in common pharmaceutical excipients. J Pharm Sci. 2007;96(1):106–16.
Lam XM, Lai WG, Chan EK, Ling V, Hsu CC. Site-specific tryptophan oxidation induced by autocatalytic reaction of polysorbate 20 in protein formulation. Pharm Res. 2011;28(10):2543–55.
Wu Y, Levons J, Narang AS, Raghavan K, Rao VM. Reactive impurities in excipients: profiling, identification and mitigation of drug-excipient incompatibility. AAPS PharmSciTech. 2011;12(4):1248–63.
Ding SL. Quantitation of Hydroperoxides in the aqueous-solutions of nonionic surfactants using Polysorbate-80 as the model surfactant. J Pharm Biomed Anal. 1993;11(2):95–101.
Skliri E, Lykakis IN, Armatas GS. Ordered mesoporous V2O5/WO3 composite catalysts for efficient oxidation of aryl alcohols. RSC Adv. 2014;4(86):46170–8.
Khaksar M, Boghaei DM, Amini M. Synthesis, structural characterization and reactivity of manganese tungstate nanoparticles in the oxidative degradation of methylene blue. Cr Chim. 2015;18(2):199–203.
ACKNOWLEDGMENTS AND DISCLOSURES
We would like to thank Genentech for support of this study, and Drs. G. Macpherson and C. Marshall at the Department of Geology, The University of Kansas, for their help with the ICP-MS measurements and Raman spectroscopy studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rathnayaka, H., Mozziconacci, O., Sreedhara, A. et al. Fragmentation of a Monoclonal Antibody by Peroxotungstate. Pharm Res 35, 219 (2018). https://doi.org/10.1007/s11095-018-2496-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-018-2496-0