Abstract
Purpose
To investigate a new microfluidic method for the continuous preparation of hollow-shell nanoparticles of a hydrophobic polymer and the simultaneous encapsulation within these of a hydrophilic active pharmaceutical ingredient.
Method
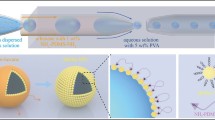
A specially designed and constructed microfluidic device which facilitates at a junction the impingement of two liquids flowing in capillaries kept 60° apart, one containing the polymer ethyl cellulose (EC) and the other active pharmaceutical ingredient amoxicillin, and a gas flowing in a capillary bisecting the two liquid flows, was used to continuously generate EC coated microbubbles at an outlet directly below the gas flow. The bubbles produce EC nanoparticles whilst encapsulating amoxicillin, and these were characterised by microscopy, zeta potential measurements, FTIR and UV spectroscopy and in vitro drug release and kinetic studies.
Results
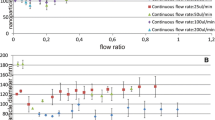
The device produced ~5 × 106 microbubbles per minute from the surface of which EC nanocarriers were released spontaneously according to an evaporation-controlled mechanism. The gas pressure was very effective in controlling the size and size distribution of the nanocarriers.
Conclusions
Nanocarriers with diameter between 10 and 800 nm were continuously produced by controlling the gas pressure between 110 and 510 kPa. Depending on their size, particles were capable of encapsulating 65–88% of amoxicillin which was released over ~12 h.












Similar content being viewed by others
References
Dendukuri D, Doyle PS. The synthesis and assembly of polymeric microparticles using microfluidics. Adv Mater. 2009;21:1–16.
Zhang C, Xu J, Ma W, Zheng W. PCR microfluidic devices for DNA amplification. Biotech Adv. 2006;24:243–84.
Zheng B, Tice JD, Roach LS, Ismagilov RF. A droplet-based, composite PDMS/glass capillary microfluidic system for evaluating protein crystallization conditions by microbatch and vapor-diffusion methods with on-chip x-ray diffraction. Angew Chem Int Ed. 2004;43:2508–11.
Fang A, Cathala B. Smart swelling biopolymer microparticles by a microfluidic approach: synthesis, in situ encapsulation and controlled release. Coll Surf B: Biointerfaces. 2011;82:81–6.
Hung L-H, Teh S-Y, Jester J, Lee AP. PLGA micro/nanosphere synthesis by droplet microfluidic solvent evaporation and extraction approaches. Lab Chip. 2010;10:1820–5.
Barratt G. Colloidal drug carriers: achievements and perspectives. Cell Mol Life Sci. 2003;60:21–37.
Reverchon E. Supercritical antisolvent precipitation of micro- and nano- particles. J Supercrit Fluids. 1999;15:1–21.
Lui L, Yang J-P, Ju X-J, Xie R, Yang L, Liang B, et al. Microfluidic preparation of monodisperse ethyl cellulose hollow microcapsules with non-toxic solvent. J Coll Interf Sci. 2009;336:100–6.
Song M, Li N, Sun S, Tiedt LR, Liebenberg W, de Villiers MM. Effect of viscosity and concentration of wall former, emulsifier and pore-inducer on the properties of amoxicillin microcapsules prepared by emulsion solvent evaporation. Il Farmaco. 2005;60:261–7.
Siepmann J, Peppas NA. Higuchi equation: derivation, applications, use and misuse. Int J Pharm. 2011;418:6–12.
Higuchi T. Mechanism of sustained- action medication Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52:1145–9.
Parikh NH, Porter SC, Rohera BD. Aqueous ethylcellulose dispersion of ethylcellulose. I. Evaluation of coating process variables. Pharmaceut Res. 1993;10:525–34.
Lin W-J, Wu T-L. Modification of the initial release of a highly water-soluble drug from ethyl cellulose microspheres. J Microencapsul. 1999;16:639–46.
Crowley MM, Schroeder B, Fredersdorf A, Obara S, Talarico M, Kucera S, et al. Physcochemical properties and mechanism of drug release from ethyl cellulose matrix tablets prepared by direct compression and hot-melt extrusion. Int J Pharm. 2004;269:509–22.
Bruno L, Kasapis S, Chaudhary V, Chow KT, Heng PWS, Leong LP. Temperature and time effects on the structural properties of a non-aqueous ethyl cellulose drug delivery system. Carb Poly. 2011;86:644–51.
Appel LE, Zentner GM. Use of modified ethycellulose lattices for microporous coating of osmotic tablets. Pharmaceut Res. 1991;8:600–4.
Kang MK, Kim J-C. pH-dependent release from ethylcellulose microparticles containing alginate and calcium carbonate. Coll Poly Sci. 2010;288:265–270.
Murtaza G. Ethylcellulose microparticles: a review. Acta Poloniae Pharm Drug Res. 2012;69:11–22.
Montes A, Gordillo MD, Pereyra EJ, de la Ossa M. Co-precipitation of amoxicillin and ethyl cellulose microparticles by supercritical antisolvent process. J Supercrit Fluids. 2011;60:75–80.
Choy YB, Choi H, Kim K. Uniform ethyl cellulose microspheres of controlled sizes and polymer viscosities and their drug-release profiles. J Appl Polym Sci. 2009;112:850–7.
Khan SA, Ahmad M, Murtaza G, Aamir MN, Madni A, Kousar R, Minhas U. Development of a single combined microencapsulated formulation of allopurinol and nimesulide and investigation of their release behaviours. Ars Pharm. 2010;51:105–16.
Hu L, Liu W, Li L, Zhao J, Yang X. Preparation and in vitro, in vivo evaluation of clarithromycin microcapsules. J Basic Clin Pharm. 2011;2:1–9.
Pancholi K, Stride E, Edirisinghe M. Dynamics of bubble formation in highly viscous liquids. Langmuir. 2008;24:4388–93.
Gunduz O, Ahmad Z, Stride E, Tamerler C, Edirisinghe M. Bioinspired bubble design for particle generation. J Roy Soc Interface. 2012;9:389–95.
Gunduz O, Ahmad Z, Stride E, Edirisinghe M. A device for the fabrication of multifunctional particles from microbubble suspensions. J Mat Sci Eng C. 2012;32:1005–10.
Stride E, Pancholi K, Edirisinghe M, Samarasinghe S. Increasing the non-linear character of microbubble oscillations at low acoustic pressures. J Roy Soc Interface. 2008;5:807–11.
Huang L-Y, Yu D-G, Branford-White C, Zhu L-M. Sustained release of ethyl cellulose micro-particle drug delivery systems prepared using electrospraying. J Mater Sci. 2012;47:1372–7.
Kalogiannis CG, Michailof CM, Panayiotou CG. Microencapsulation of amoxicillin in poly(L-lactic acid) by supercritical antisolvent precipitation. Ind Eng Chem Res. 2006;45:8738–43.
Xu Q, Hashimoto M, Dang TT, Hoare T, Kohane DS, Whitesides GM, et al. Preparation of monodisperse biodegradable polymer microparticles using a microfluidic flow-focusing device for controlled drug delivery. Small. 2009;5:1575–81.
Moine P, Mazoit JX. Streptococcus pneumoniae pneumonia in mice: Optimal amoxicillin dosing predicted from a pharmacokinetic-pharmacodynamic model. J Pharmacol Exp Ther. 1999;291:1086–92.
Wagh SC, Kumar JS, Banerjee S. Development and evaluation of a novel extended release venlafaxine hydrochloride matrix tablets. J Pharm Res. 2012;5:2184–90.
Pahwa R, Chhabra L, Lamba AK, Jindal S, Rathour A. Formulation and in-vitro evaluation of effervescent floating tablets of an antiulcer agent. J Chem Pharm Res. 2012;4:1066–73.
Bohr A, Kristensen J, Dyas M, Edirisinghe M, Stride E. Release profile and characteristics of electrosprayed particles for oral delivery of a practically insoluble drug. J R Soc Interface. 2012. doi:10.1098/rsif.2012.0166.
Acknowledgments and Disclosures
The authors wish to thank the Islamic Development Bank for supporting the doctoral research programme of Oguzhan Gunduz. They would also like to thank Kevin Reeves for the help with the scanning electron microscopes in the Archaeology Department at UCL and Dr Suguo Huo of the London Centre for Nanotechnology for the use of FIB.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gunduz, O., Ahmad, Z., Stride, E. et al. Continuous Generation of Ethyl Cellulose Drug Delivery Nanocarriers from Microbubbles. Pharm Res 30, 225–237 (2013). https://doi.org/10.1007/s11095-012-0865-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-012-0865-7