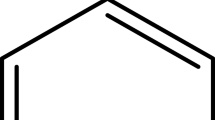
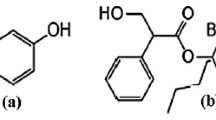
Solriamfetol is a dopamine and norepinephrine reuptake inhibitor used to treat excessive daytime sleep in patients with narcolepsy. The prime intention of this study was to develop a simple, accurate, precise and robust RP-HPLC method for the estimation of solriamfetol in pure and marketed dosage forms. The chromatographic analysis was performed on a Waters symmetry C18 column (150 × 4.6 mm, 5 μ) using a mobile phase containing a mixture of 0.1% formic acid: acetonitrile at 30:70, v/v. A flow rate of 1 mL/min was applied and detected at a wavelength of 257 nm. Solriamfetol eluted at 2.4 min over a run time of 5 min. The method showed linearity in the range of 12.5 to 75 μg/mL with a correlation coefficient of 0.9998. The sample solution was exposed to various stress conditions and shows degradation in the range of 3.4% to 27.2%. The solriamfetol peak was well resolved from the degradation peaks observed in stress degradation, which proves the specificity of the method. The developed method was found to be linear, accurate, precise, sensitive, simple and economical with a short analysis time. Thus, this method can be utilized for regular study in quality control labs for the quantification of solriamfetol in bulk and pharmaceutical dosage forms.





Similar content being viewed by others
References
M. Kiran, M. Emmanuel, P. Giuseppe, D. Yves, Clin. Sleep Med., 18(1), 289 – 304 (2022).
M. Anthony, Drugs, 79(7), 785 – 790 (2017).
Y. Junyi and G. Junfa, Clin. Pharmacol., 12(8), 723 – 728 (2019).
J. Powell, C. Piszczatoski, S. Garland, Ann. Pharmacother., 54(10),1016 – 1020 (2020).
I. Fejos, G. Toth, B. Varnai, et al., Electrophoresis, 42, 1818 – 1825 (2021).
I. Koteles, M. Foroughbakhshfasaei, M. Dobo, et al., Chromatographia, 83, 909 – 913 (2020).
ICH Harmonized Tripartite Guideline; Validation of Analytical Procedures: Text and Methodology, Q2 (R1); International Conference on Harmonization, IFPMA, Geneva, Switzerland (2005).
B. Lal, D. Kapoor, M. Jaimini, J. Drug Deliv. Ther., 9(2), 501 – 506 (2019).
S. Sharma, S. Goyal, K. Chauhan, Int. J. Appl. Pharm., 10(6), 8 – 15 (2018).
International Conference on Harmonisation of technical requirements for registration of pharmaceuticals for human use, ICH Harmonised tripartite guideline: stability testing of new drug substances and products Q1A (R2) (2003).
International Conference on Harmonisation of technical requirements for registration of pharmaceuticals for human use, ICH Harmonised tripartite guideline: Stability testing: photostability testing of new drug substances and products Q1B (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gollu, G., Gummadi, S. & Gurijala, L. Stability Indicating RP-HPLC-PDA Method for the Quantification of Solriamfetol in Bulk and Marketed Dosage Form. Pharm Chem J 57, 1111–1117 (2023). https://doi.org/10.1007/s11094-023-02991-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-023-02991-1