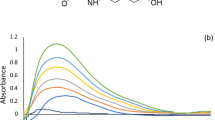
A simple, reproducible, and swift inhibitory kinetic approach for the N-acetylcysteine (NAC) determination has been developed and linked to NAC quantification in drug formulations. The method is based on the inhibitory feature of N-acetylcysteine. NAC forms a stable complex with Hg2+ and reduces the actual Hg2+ concentration and ultimately the rate of reaction between N-R-salt and [Ru(CN)6]4- catalyzed by Hg2+. Under the optimized experimental conditions with Temp = 45.0 ± 0.1°C, I = 0.05 mole dm-3 (KNO3), [N-R-salt] = 5.5 × 10-4 mole dm-3 [Hg+2] = 8.0 × 10-5 mole dm-3, pH = 6.0 ± 0.02, and [Ru(CN)64-] = 5.25× 10-5 mole dm-3, fixed time of 7 and 12 min was selected to compute the absorbance at 525 nm corresponding to the ultimate reaction product [Ru(CN)5 N-R-salt]3-. The inhibitory action of NAC toward cyanide imitation from [Ru(CN)6]4- by N-R-salt, catalyzed by Hg2+, has been demonstrated using a redesigned mechanistic scheme. With the proposed kinetic spectrophotometric method the micro-level quantification of NAC in distinct water samples can be done down to 1.25 × 10-6 mole dm-3. The developed procedure is highly reproducible and can be efficiently used to quantitatively estimate the NAC in the drug samples with high accuracy. The general additives present in drugs do not substantially interfere in the determination of NAC even up to 1000 times with [NAC].







Similar content being viewed by others
References
W. S. Waring, Ther. Adv. Drug Saf., 3(6), 305 – 315 (2012).
R. S. Hoffman, Clin. Med., 20, 100314 (2020).
D. N. Bateman and J. W. Dear, Toxicol. Res., 8(4), 489 – 498 (2019).
A. M. Sadowska, J. Verbraecken, K. Darquennes, and W. A. De Backer, Int. J. Chron. Obstruct. Pulmon. Dis., 1(4), 425 – 434 (2006).
S. Šalamon, B. Kramar, T. P. Marolt, et al., Antioxidants, 8(5), 111 (2019).
G. Tardiolo, P. Bramanti and E. Mazzon, Molecules, 23, 3305 (2018).
B. M. Craver, G. Ramanathan, S. Hoang, et al., Blood Adv., 4(2), 312 – 321 (2020).
K. Tang, Front. Mar. Sci., 7, 68 (2020).
C. Abadie and G. Tcherkez, Commun. Biol., 2, 379 (2019).
G. K. Kolluru, X. Shen and C. G. Kevil, Arterioscler. Thromb. Vasc. Biol., 40, 874 – 884 (2020).
J. M. Fukuto, L. J. Ignarro, P. Nagy, et al., FEBS Lett., 592, 2140 – 2152 (2018).
R. O. Omondi, O. Stephen, S. O. Ojwach, and D. Jaganyi, Inorg. Chim. Acta, 512, 119883 (2020).
R. M. Naik, A. Srivastava and A. Asthana, J. Iran. Chem. Soc., 5, 29 – 36 (2008).
T. Iioka, S. Takahashi, Y. Yoshida, et al., J. Comput. Chem., 40, 279 – 285 (2019).
R. M. Naik, A. Srivastava, A. K. Verma, et al., S. Bioinorg. Reac. Mech., 6, 185 – 192 (2007).
A. Srivastava, V. Sharma, A. Prajapati, et al., Chem. Chem. Technol., 13(3), 275 – 279 (2019).
S. Prasad, R. M. Naik and A. Srivastava, Spectrochim. Acta. A, 7, 958 – 965 (2008).
R. Rastogi, A. Srivastava and R. M. Naik, J. Disp. Sc. Tech., 41(7), 1045 – 1050 (2020).
A. Srivastava, R. M. Naik and R. Rastogi, J. Iran. Chem. Soc., 17(9), 2327 – 2333 (2020).
Y. Huang, T. Lin, L. Hou, et al., Microchem. J., 144, 190 – 194 (2019).
A. G. Dedov, D. Y. Marchenko and L. V. Zrelova, Pet. Chem., 58, 714 – 720 (2018).
A. Kostara, G. Z. Tsogas, A. G. Vlessidis, and D. L. Giokas, ACS Omega, 3(12), 16831 – 16838 (2018).
A. Raab and J. Feldmann, Anal. Chim. Acta, 1079, 20 – 29 (2019).
S. Zhand and J. Q. Jiang, Biointerface Res. Appl. Chem., 9, 4433 – 4438 (2019).
L. Ni, X. Geng, S. Li, et al., Talanta, 207, 120283 (2020).
J. Nelson, J. Assoc. Offic. Anal. Chem., 64, 1174 – 1178 (1981).
Q. Chao, H. Sheng, X. Cheng, and T. Ren, Anal. Sci., 21, 721 – 724 (2005).
S. Shoba, O. M. Bankole and A. S. Ogunlaja, Anal. Methods, 12, 1094 – 1106 (2020).
I. Nugrahani, I. M. Abotbina, C. N. Apsari, et al., Biointerface Res. Appl. Chem., 10(1), 4780 – 4785 (2019).
T. Perez-Ruiz, C. Martinez- Lozano, V. Tomas, and C. Sidrachde- Cardona, J. Pharm. Biomed. Anal., 15, 33 – 38 (1996).
G. Feng, S. Sun, M. Wang, et al., J. Water Supply Res. T, 67(5), 498 – 505 (2018).
U. Dzieko, N. Kubczak, K. P. Przybylska, et al., Molecules, 25, 1232 (2020).
L. Cao, T. Wei, Y. Shi, et al., J. Liq. Chrom. Relat. Tech., 41(2), 58 – 65 (2019).
A. Srivastava, Biointerface Res. Appl. Chem., 10(6), 7152 – 7161 (2020).
A. Agarwal, S. Prasad and R. M. Naik, Microchem. J., 128, 181 – 186 (2016).
A. Srivastava, Biointerface Res. Appl. Chem., 11(3), 10654 – 10663 (2021).
F. Athar, K. Husain, M. Abid, and A. Azam, Chem. Biodiversity, 2, 1320 – 1330 (2005).
C. M. Bastos, K. A. Gordon and T. D. Ocain, Bioorg. Med. Chem. Lett., 8, 147 – 150 (1998).
B. Yu, T. W. Rees, J. Liang, et al., Dalton Trans., 48, 3914 – 3921 (2019).
F. A. Gomes-Junior, R. S. Silva, R. G. Lima, and M. A. Vannier-Santos, FEMS Microbio. Lett., 364(9) (2017).
R. G. Kenny and C. J. Marmion, Chem. Rev., 119, 1058 – 1137 (2019).
L. Gua, X. Lia, Q. Ran, et al., Cancer Med., 5, 2850 – 2860 (2016).
K. Lin, Z. Z. Zhao, H. B. Bo, et al., Pharmacol., 9, 1323 (2018).
J. P. C. Coverdale, T. L. M. Carron and I. R. Canelon, Inorganics, 7, 31 (2019).
R. M. Naik, A. Agarwal, A. K. Verma, et al., Int. J. Chem. Kinet., 41, 215 – 226 (2009).
A. Srivastava, V. Sharma, V. K. Singh, and K. Srivastava, J. Mex. Chem. Soc., 66, 57 – 69 (2022).
A. Srivastava, Biointerface Res. Appl. Chem., 11(4), 11404 – 11417 (2021).
British Pharmacopoeia (1995 Ed.), British Pharmacopoeia Commission (London).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Srivastava, A., Srivastava, N., Srivastava, A. et al. Determination of N-Acetylcysteine in Pure and Drug Formulations Using Inhibitory Kinetic Approach. Pharm Chem J 57, 756–762 (2023). https://doi.org/10.1007/s11094-023-02949-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-023-02949-3