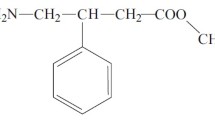
An HPLC method for quantitative determination of glutaron was developed in order to perform a pharmacokinetic analysis. The method was highly sensitive and selective and could be used for determination in biological samples. The sample preparation method was optimized and did not significantly affect the average measurement error of the HPLC quantitative determination.




Similar content being viewed by others
References
V. V. Bagmetova, Yu. V. Chernysheva, O. V. Merkushenkova, et al., Eksp. Klin. Farmakol., 76(3), 7 – 9 (2013).
A. A. Spasov, L. A. Smirnova, I. N. Iezhitsa, et al., Vopr. Med. Khim., 48(3), 233 – 258 (2002).
I. N. Tyurenkov, V. N. Perfilova, L. A. Smirnova, et al., Khim.-farm. Zh., 44(12), 68 – 70 (2010); Pharm. Chem. J., 44(12), 702 – 704 (2011).
I. N. Tyurenkov, V. N. Perfilova, L. A. Smirnova, et al., Khim.-farm. Zh., 47(3), 55 – 56 (2013); Pharm. Chem. J., 47(3), 179 – 180 (2013).
Guideline on Bioanalytical Method Validation (EMEA 2012); www.ema.europa.eu [http://www.ema.europa.eu/docs/enGB/documentlibrary/Scientificguideline/2011/08/WC500109686.pdf].
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 48, No. 6, pp. 54 – 56, June, 2014.
Rights and permissions
About this article
Cite this article
Smirnova, L.A., Ryabukha, A.F., Kuznetsov, K.A. et al. Quantative Determination of the New Glutamic Acid Derivative Glutaron in Biological Samples. Pharm Chem J 48, 414–416 (2014). https://doi.org/10.1007/s11094-014-1122-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-014-1122-7