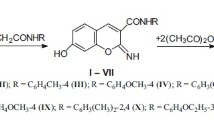
A series of new pyranochromen-3-carboxamides have been synthesized in good yield by reacting 2-phenyl-4-hydroxy-6H-1,3-thiazin-6-ones with substituted 3-formylchromones. Pharmacological screening showed an influence of the substituent in the 9-position of the pyranochromen-3-carboxamides on the acute toxicity and antifungal, analgesic, and anxiolytic activity. 9-Bromo-2H,5H-pyranochromen-3-carboxamide can be recommended for further pharmacological investigation.
Similar content being viewed by others
References
M. A. Salamaa and L. A. Almotabacanib, Phosphorus Sulfur Silicon Relat. Elem., 179(2), 305–319 (2004).
M. Koketsu, K. Tanaka, Y. Takenaka, et al., Eur. J. Pharm. Sci., 15(3), 307–310 (2002).
V. N. Kuklin, Author’s Abstract of a Doctoral Dissertation in Pharmaceutical Sciences, St. Petersburg (1996).
G. P. Yakovlev and K. F. Blinova (eds.), Medicinal Plant Raw Material. Pharmacognosy, Student Aide, Spetslit, St. Petersburg (2004), pp. 387–389.
Ya. Buresh, O. Bureshova, and J. P. Houston, Methods and Basic Experiments for Studying the Brain and Behavior [in Russian], Vysshaya Shkola, Moscow (1991).
State Pharmacopoeia of the Russian Federation, Part 1, Scientific Center for Review of Medical Agents [in Russian], Moscow (2007), pp. 150–152.
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 46, No. 1, pp. 12 – 17, January, 2012.
Rights and permissions
About this article
Cite this article
Shutov, R.V., Kuklina, E.V., Anisimova, N.A. et al. Synthesis and biological activity of new pyranochromen-3-carboxamides. Pharm Chem J 46, 10–14 (2012). https://doi.org/10.1007/s11094-012-0725-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-012-0725-0