Abstract
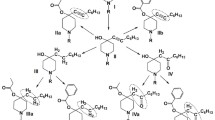
A method for the catalytic synthesis of local anesthetics — lidocaine, bipuvacaine, mepivacaine, trimecaine, and pyromecaine — by reductive acylation is described. The process takes place in mild conditions (20–50°C, hydrogen at atmospheric pressure, organic solvent) with target product yields of 54–93%.
Similar content being viewed by others
References
M. G. Abdullaev, Khim.-Farm. Zh., 35(1), 42–45 (2001).
M. G. Abdullaev, Khim.-Farm. Zh., 35(10), 30–33 (2001).
M. G. Abdullaev, Khim.-Farm. Zh., 36(1), 28–30 (2002).
M. G. Abdullaev, Khim.-Farm. Zh., 39(12), 37–39 (2005).
M. D. Mashkovskii, Medicinal Substances [in Russian], Novaya Volna, Moscow (2000), Vol. 1, pp. 294–296.
V. A. Zagorevskii, I. V. Chernyakova, T. I. Ivanov, et al., Khim.-Farm. Zh., 23(3), 292–296 (1989).
USA Patent No. 2441498 (1948).
USA Patent No. 4110331 (1977).
H. Rinderkneht, Helv. Chim. Acta, 42, 1324 (1959).
M. G. Abdullaev and M. V. Klyuev, Izv. Vuzov. Ser. Khimii Khim. Tekhnol., 48(5), 74–78 (2005).
M. G. Abdullaev and M. V. Klyuev, Izv. Vuzov. Ser. Khimii Khim. Tekhnol., 48(11), 93–97 (2005).
Kh. M. Colquhoun, D. Holton, D. Thompson, and M. Twigg, New Pathways for Organic Synthesis. Practical Applications of Transition Metals [Russian translation], Khimiya, Moscow (1989).
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 42, No. 6, pp. 46–48, June, 2008.
Rights and permissions
About this article
Cite this article
Abdullaev, M.G., Klyuev, M.V., Abdullaeva, Z.S. et al. Preparation of lidocaine, bipuvacaine, mepivacaine, trimecaine, and pyromecaine by reductive acylation on palladium catalysts. Pharm Chem J 42, 357–359 (2008). https://doi.org/10.1007/s11094-008-0126-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-008-0126-6