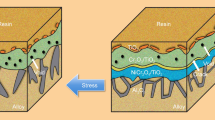
Abstract
Alloys and oxidation-resistant coatings utilized in high-temperature applications can be degraded by aerosols that deposit onto surfaces during operation. Understanding how the deposit composition influences the hot corrosion mechanisms is essential to develop more durable materials. This work advances the understanding of the effect of complex oxide and sulfate deposits on the degradation of an alumina-forming FeCrAlY alloy in comparison to reactions with single-crystal sapphire. The deposit compositions were developed to systematically understand the effect of anion makeup (mixed oxides, oxide–sulfate, and sulfates) and the effect of adding Na and K salts. CaSO4 was used as a control. The mixed oxide and oxide–sulfate deposits increased the frequency of thermally grown oxide (TGO) intrusions in FeCrAlY but did not produce a noticeable change in the sapphire. Pure CaSO4 and mixed sulfate reacted with the TGO and sapphire to form calcium aluminates and led to roughening of the specimen–reaction product interface. The primary difference between the CaSO4 and mixed sulfate deposits was the increased uniformity of the attack by the latter due to its tendency to melt and spread. Comparison between the change in the degradation features in the presence of CaSO4 and mixed sulfate deposits on both types of specimens expands the current understanding of sulfate-based hot corrosion.












Similar content being viewed by others
References
P. Marcus, Corrosion science. 36, 1994 (2155).
B. Gleeson, Shreir’s corrosion. 182, (2010)
J. A. Goebel and F. S. Pettit, Metallurgical Transactions. 1, 1970 (3421).
J. A. Goebel, F. S. Pettit, and G. W. Goward, Metall Mater Trans B. 4, 1973 (261).
G. W. Goward, Journal of Engineering for Gas Turbines and Power. 108, 1986 (421).
N. Eliaz, G. Shemesh, and R. M. Latanision, Engineering Failure Analysis. 9, 2002 (31).
J. Stringer, Annual Review of Materials Science. 7, 1977 (477).
R. A. Rapp, Corrosion Science. 44, 2002 (209).
K. L. Luthra, Reaction mechanism. Metallurgical Transactions A. 13, 1982 (1853).
K. L. Luthra and D. A. Shores, Journal of the Electrochemical Society. 127, 1980 (2202).
R. L. Jones, Cobalt oxide-SO2/SO3 reactions in cobalt-sodium mixed sulfate formation and low temperature hot corrosion. in: High temperature corrosion. Proceedings of the International Conference, San Diego, CA, March 2–6, 1981. 1983.
K. Y. Jung, F. S. Pettit, and G. H. Meier, The effect of Ca-rich deposits on the high temperature degradation of coated and uncoated superalloys. in: Materials Science Forum. Trans Tech Publ; 2008.595:805–812.
T. Gheno, G. H. Meier, and B. Gleeson, Oxidation of Metals 84, 2015 (185).
M. B. Krisak, B. I. Bentley, A. W. Phelps, and T. C. Radsick, Journal of Propulsion and Power. 33, 2017 (697).
P. Brennan, Environmental Factors Affecting CaO-and CaSO4-Induced Degradation of Second-Generation Nickel-Based Superalloys. University of Pittsburgh; 2020.
M. Kovalchuk, and B. Gleeson, Laboratory-Scale Replication of Deposit-Induced Degradation of High-Temperature Turbine Components. in: Superalloys 2020, eds. S. Tin, M. Hardy, J. Clews, et al. (Springer International Publishing; Cham, 2020). pp. 789–797. doi:https://doi.org/10.1007/978-3-030-51834-9_77.
A. S. Chikhalikar, E. P. Godbole, D. L. Poerschke Corrosion Science. 110892. (2022) doi:https://doi.org/10.1016/j.corsci.2022.110892
D. A. Shifler, The Increasing Complexity of Hot Corrosion. In: American Society of Mechanical Engineers Digital Collection; 2017. doi:https://doi.org/10.1115/GT2017-65281
D. A. Shifler, and S. R., Choi, CMAS Effects on Ship Gas-Turbine Components/Materials. In: ASME Turbo Expo 2018: Turbomachinery Technical Conference and Exposition. American Society of Mechanical Engineers Digital Collection; (2018)
D. A. Shifler, Materials at High Temperatures. 35, 2018 (225).
K. J.Meisner, E. J. Opila, Oxid Met. 2020; doi:https://doi.org/10.1007/s11085-020-09990-7.
J. L. Smialek, F. A. Archer, and R. G. Garlick, JOM 46, 1994 (39).
T. Gheno and B. Gleeson, Oxidation of Metals. 87, 2017 (249).
T. Gheno and B. Gleeson, Oxidation of Metals 86, 2016 (385).
D. L. Poerschke, R. W. Jackson, and C. G. Levi, Annual Review of Materials Research. 47, 2017 (297).
C. G. Levi, J. W. Hutchinson, M.-H. Vidal-Sétif, and C. A. Johnson, MRS bulletin. 37, 2012 (932).
A. R. Ericks, F. W. Zok, D. L. Poerschke, and C. G. Levi, Journal of the American Ceramic Society. 105, 2022 (3665).
G. H. Meier, Oxidation of Metals 98, 2022 (1).
A. S. Chikhalikar, E. P. Godbole, and D. L. Poerschke, Acta Materialia 237, 2022 (118184).
P. T. Brennan, D. Konitzer, and M. Brennan, B Gleeson, Oxidation of Metals. 1 (2022)
W. D. Summers, D. L. Poerschke, D. Park, J. H. Shaw, F. W. Zok, and C. G. Levi, Acta Materialia 160, 2018 (34).
W. D. Summers, D. L. Poerschke, A. A. Taylor, A. R. Ericks, C. G. Levi, and F. W. Zok, Journal of the American Ceramic Society 103, 2020 (2919).
D. W. Green and R. H. Perry, Perry’s chemical engineers’ handbook. (McGraw-Hill Education; 2008)
N. Kanari, N.-E. Menad, E. Ostrosi, et al., Metals. 8, 2018 (1084).
P. G. Coombs and Z. A. Munir, Journal of thermal analysis. 35, 1989 (967).
V. K. Tolpygo and D. R. Clarke, Surface and Coatings Technology. 120–121, 1999 (1).
V. K. Tolpygo and D. R. Clarke, Materials at High Temperatures. 17, 2000 (59).
E. M. Levin, C. R. Robbins and H. F. McMurdie. Phase Diagrams for Ceramists. 181, (1964)
B. Hallstedl, Journal of the American ceramic society. 73, 1990 (15).
H. Du, Journal of phase equilibria. 21, 2000 (6).
D. Freyer, W. Voigt, and K. Köhnke, European Journal of Solid State and Inorganic Chemistry. 35, 1998 (595).
M. N. Scheidema and P. Taskinen, Industrial & engineering chemistry research. 50, 2011 (9550).
A. Dimyati, H. J. Penkalla, P. Untoro, D. Naumenko, W. J. Quadakkers, and J. Mayer, International Journal of Materials Research. 94, 2022 (180).
T. Sand, A. Edgren, C. Geers, et al., Oxidation of Metals 95, 2021 (221).
B. A. Pint, Journal of the American Ceramic Society. 86, 2003 (686).
Acknowledgements
The authors are grateful to Drs. Pin Lu, Changning Niu, and Jiadong Gong (QuesTek Innovations LLC) for the insightful discussions, and to Dr. Eeshani Godbole for assistance preparing the deposit powders.
Funding
This research was supported by the Office of Naval Research (Award number N68335-20-C-0472 monitored by Dr. David Shifler) in collaboration with QuesTek Innovations LLC. Part of this work was carried out in the Characterization Facility, College of Science and Engineering, University of Minnesota, which receives support from the National Science Foundation through the MRSEC (Award Number DMR-2011401) and the NNCI (Award Number ECCS-2025124) programs.
Author information
Authors and Affiliations
Contributions
Both authors designed the study. AC performed the experiments, characterization, and initial data analysis, prepared the figures, and wrote the first draft of the manuscript. DP contributed to data interpretation and edited the figures and manuscript. Both authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chikhalikar, A.S., Poerschke, D.L. Comparative Study of FeCrAlY and Sapphire Hot Corrosion by Mixed Oxide and Sulfate Deposits. High Temperature Corrosion of mater. 100, 321–344 (2023). https://doi.org/10.1007/s11085-023-10182-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-023-10182-2