Abstract
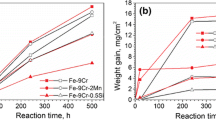
Corrosion behaviour of Fe–Cr (15, 20, 25, or 30 wt.%) alloys in wet CO2 without and with HCl (0.05 or 0.1 vol%) at 750 °C was investigated. The results are compared with those at 650 °C to reveal the temperature effect. In general, weight gain kinetics and oxide scaling rates decreased with the increased chromium levels. The effect of temperature on weight gains and oxide scaling was strongly dependent on NCr. For the 15 and 20 Cr alloys, rates increased with temperature in all conditions. However, for the 25 Cr alloy, the rate increased with temperature in HCl free gas, but decreased with temperature when HCl was present. The 30 Cr alloy experienced very small weight gains which slightly increased with T. Cross-section analysis revealed the formation of thick multi-layered oxides for Fe–15Cr and Fe–20Cr, a semi-protective duplex scale of Fe2O3 and Cr2O3 on Fe–25Cr, and a thin protective chromia scale for Fe–30Cr at both temperatures. Both oxide growth rate and oxide constitution were affected by temperature, NCr, and \({p}_{HCl}\). The correlation of these factors is discussed by considering interactions between the oxides and chlorine inside the different scale types.























Similar content being viewed by others
References
D. Mudgal, S. Singh, S. Prakash, Corrosion problems in incinerators and biomass-fuel-fired boilers, International Journal of Corrosion, 2014 (2014).
T.T. Sharobem, Mitigation of high temperature corrosion in waste-to-energy power plants, Columbia University, 2017.
D.J. Young, High temperature oxidation and corrosion of metals, Elsevier (2016).
G. Lai, A. International, Waste-to-Energy Boilers and Waste Incinerators, High-Temperature Corrosion And Materials Applications, (2007) 335–358.
L.W. McNallan MJ, Kim SH, Kang CT High Temperature Corrosion, NACE-6, Houston TX (1983) 316–321.
H. P. Nielsen, F. Frandsen, K. Dam-Johansen, and L. Baxter, The implications of chlorine-associated corrosion on the operation of biomass-fired boilers. Progress in energy and combustion science 26, 2000 (283–298).
H. Grabke, E. Reese, and M. Spiegel, The effects of chlorides, hydrogen chloride, and sulfur dioxide in the oxidation of steels below deposits. Corrosion science 37, 1995 (1023–1043).
J. Liu and M. McNallan, Effects of temperature variations on oxidation of iron–20% chromium alloys at 1200 K in Ar–20% O2-Cl2 gas mixtures. Materials and Corrosion 50, 1999 (253–260).
K. K. Aye, J. Zhang, and D. J. Young, Effect of Hydrogen Chloride on Corrosion Behaviour of Fe–Cr Alloys in Wet CO2 Gas at 650°C. Oxidation of Metals 94, 2020 (51–80).
G.H. Peter EL, Daniel GF, Thomas ED, Evan B, Physical and Chemical data, in: Chemical Engineering, The McGraw-Hill Companies, Inc., 2008.
D. Young, P. Huczkowski, T. Olszewski, T. Hüttel, L. Singheiser, and W. Quadakkers, Non-steady state carburisation of martensitic 9–12% Cr steels in CO2 rich gases at 550°C. Corrosion science 88, 2014 (161–169).
M. Taylor, J. Calvert, D. Lees, and D. Meadowcroft, The mechanism of corrosion of Fe-9% Cr alloys in carbon dioxide. Oxidation of Metals 14, 1980 (499–516).
T. Gheno, Oxidation and carburisation of model chromia-forming alloys in carbon dioxide, in, (Institute National Polytechnique of Toulouse and University of New South Wales, Ph.D diss., 2012).
T. D. Nguyen, J. Zhang, and D. J. Young, Effects of silicon on high temperature corrosion of Fe–Cr and Fe–Cr–Ni alloys in carbon dioxide. Oxidation of metals 81, 2014 (549–574).
C. Wagner, Reaktionstypen bei der Oxydation von Legierungen, Zeitschrift für Elektrochemie, Berichte der Bunsengesellschaft für physikalische. Chemie 63, 1959 (772–782).
R. Rapp, The transition from internal to external oxidation and the formation of interruption bands in silver-indium alloys. Acta Metallurgica 9, 1961 (730–741).
J. Takada and M. Adachi, Determination of diffusion coefficient of oxygen in α-iron from internal oxidation measurements in Fe-Si alloys. Journal of materials science 21, 1986 (2133–2137).
J. Takada, S. Yamamoto, S. Kikuchi, and M. Adachi, Determination of diffusion coefficient of oxygen in γ-iron from measurements of internal oxidation in Fe-Al alloys. Metallurgical Transactions A 17, 1986 (221–229).
R. Braun, M. Feller‐Kniepmeier, Diffusion of chromium in α‐iron, physica status solidi (a), 90 (1985) 553–561.
J. Swisher and E. Turkdogan, Solubility, permeability, and diffusivity of oxygen in solid iron. AIME Met Soc Trans 239, 1967 (426–431).
C. Wagner, Theoretical analysis of the diffusion processes determining the oxidation rate of alloys. Journal of the Electrochemical Society 99, 1952 (369–380).
A. Jacob, E. Povoden-Karadeniz, and E. Kozeschnik, Revised thermodynamic description of the Fe-Cr system based on an improved sublattice model of the σ phase. Calphad 60, 2018 (16–28).
F. V. F. Gesmundo, Y. Niu, and D. L. Douglass, Oxidation of Metals 40, 1993 (374–393).
H. Davies and A. Dinsdale, Theoretical study of steam grown oxides as a function of temperature, pressure and p (O2). Materials at High Temperatures 22, 2005 (15–25).
D. J. Young, T. D. Nguyen, P. Felfer, J. Zhang, and J. M. Cairney, Penetration of protective chromia scales by carbon. Scripta Materialia 77, 2014 (29–32).
I.G. Wright, R. Barry Dooley, Morphologies of oxide growth and exfoliation in superheater and reheater tubing of steam boiler, Materials at High Temperatures, 28 (2011) 40–57.
I. G. Wright, M. Schutze, P. F. Tortorelli, and R. Dooley, Towards improved prediction of scale exfoliation from steam tubes. Materials at High Temperatures 24, 2007 (265–274).
Acknowledgements
Financial support by the Australian Research Council under the Discovery Scheme is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aye, K.K., Cai, Y., Zhang, J. et al. Effect of Temperature on Corrosion Behaviour of Fe–Cr Alloys in Wet CO2 With and Without HCl Gases. Oxid Met 97, 371–400 (2022). https://doi.org/10.1007/s11085-021-10092-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-021-10092-1