Abstract
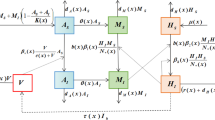
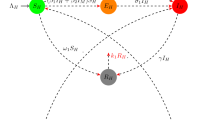
Since the outbreak in Brazil, Zika has received the worldwide attention. Zika virus is mainly transmitted via the bites of Aedes mosquito. Recently, experimental evidence demonstrates that Zika virus in contaminated aquatic environments can be transmitted to aquatic mosquitoes through breeding. To study the effects of contaminated aquatic environments on Zika transmission, we propose a new Zika model by introducing a general incidence function. For a general incidence function, we calculate the basic reproduction number \(R_0\), analyze the stability of disease-free equilibria and give general conditions with the occurrence of backward bifurcation. For the two special sublinear incidence functions, \(R_0=1\) is a sharp threshold. It means that the disease can be eradicated by reducing \(R_{0}\) below 1. For the special nonlinear incidence function, we investigate theoretically backward bifurcation and saddle-node bifurcation and simulate numerically Hopf bifurcation. Analysis results suggest that transmission force from contaminated aquatic environments to aquatic mosquitoes plays an important role in generating complex dynamics. Furthermore, our model is extended to explore the effects of temperature variability on spread of Zika disease.












Similar content being viewed by others
References
Dick, G.W.A., Kitchen, S.F., Haddow, A.J.: Zika virus (I): isolations and serological specificity. Trans. R. Soc. Trop. Med. Hyg. 46(5), 509–520 (1952)
Macnamara, F.N.: Zika virus: a report on three cases of human infection during an epidemic of jaundice in Nigeria. Trans. R. Soc. Trop. Med. Hyg. 48(2), 139–145 (1954)
Duffy, M.R., et al.: Zika virus outbreak on Yap Island, Federated States of Micronesia. New England J. Med. 360, 2536–2543 (2009)
World Health Organization (WHO). Emergency Committee on Zika virus and observed increase in neurological disorders and neonatal malformations (2016). https://www.chinadaily.com.cn/world/2016-02/02/content_23348858.htm
Rocklöv, J., Quam, M.B., Sudre, B., et al.: Assessing seasonal risks for the introduction and mosquito-borne spread of Zika virus in Europe. EBioMedicine 9, 250–256 (2016)
Watson-Brown, P., Viennet, E., Mincham, G., et al.: Epidemic potential of Zika virus in Australia: implications for blood transfusion safety. Transfusion 59(2), 648–658 (2019)
Centers for Disease Control and Prevention (CDC). Zika virus (2018). https://www.cdc.gov/zika/
Tataryn, J., Vrbova, L., Drebot, M., et al.: Emergency planning: travel-related Zika virus cases in Canada: October 2015–June 2017. Can. Commun. Dis. Rep. 44(1), 18 (2018)
Hashimoto, T., Kutsuna, S., Tajima, S., et al.: Importation of Zika virus from Vietnam to Japan, November 2016. Emerg. Infect. Dis. 23(7), 1223 (2017)
Jang, H.C., Park, W.B., Kim, U.J., et al.: First imported case of Zika virus infection into Korea. J. Korean Med. Sci. 31(7), 1173–1177 (2016)
Jia, H.M., Zhang, M., Chen, M.Y., et al.: Zika virus infection in travelers returning from countries with local transmission, Guangdong, China, 2016. Travel Med. Infect. Dis. 21, 56–61 (2018)
Singh, H., Singh, O.P., Akhtar, N., et al.: First report on the transmission of Zika virus by Aedes (Stegomyia) aegypti (L.)(Diptera: Culicidae) during the 2018 Zika outbreak in India. Acta Trop. 199, 105114 (2019)
Centers for Disease Control and Prevention (CDC). Zika travel information (2019). https://wwwnc.cdc.gov/travel/page/zika-information
Kucharski, A.J., et al.: Transmission dynamics of Zika virus in island populations: a modelling analysis of the 2013–14 French Polynesia outbreak. PloS Negl. Trop. Dis. 10(5), e0004726 (2016)
Zhang, Q., Sun, K.Y., Chinazzi, M., et al.: Spread of Zika virus in the Americas. Proc. Natl. Acad. Sci. 114(22), E4334–E4343 (2017)
Chen, J., Beier, J.C., Cantrell, R.S., et al.: Modeling the importation and local transmission of vector-borne diseases in Florida: the case of Zika outbreak in 2016. J. Theor. Biol. 455, 342–356 (2018)
Dantas, E., Tosin, M., Cunha, J.A.: Calibration of a SEIR-SEI epidemic model to describe the Zika virus outbreak in Brazil. Appl. Math. Comput. 338, 249–259 (2018)
He, D.H., Gao, D.Z., Lou, Y.J., et al.: A comparison study of Zika virus outbreaks in French Polynesia, Colombia and the State of Bahia in Brazil. Sci. Rep. 7(1), 273 (2017)
Wang, L., Zhao, H., Oliva, S.M., et al.: Modeling the transmission and control of Zika in Brazil. Sci. Rep. 7(1), 7721 (2017)
Agusto, F.B., Bewick, S., Fagan, W.F.: Mathematical model for Zika virus dynamics with sexual transmission route. Ecol. Complex. 29, 61–81 (2017)
Saad-Roy, C.M., Ma, J., van den Driessche, P.: The effect of sexual transmission on Zika virus dynamics. J. Math. Biol. 77(6–7), 1917–1941 (2018)
Zhao, H., Wang, L., Oliva, S.M., et al.: Modeling and dynamics analysis of Zika transmission with limited medical resources. Bull. Math. Biol. 82, 99 (2020)
Wang, L., Zhao, H.: Dynamics analysis of a Zika-dengue co-infection model with dengue vaccine and antibody-dependent enhancement. Phys. A Stat. Mech. Appl. 522, 248–273 (2019)
Marchette, N.J., Garcia, R., Rudnick, A.: Isolation of Zika virus from Aedes aegypti mosquitoes in Malaysia. Am. J. Trop. Med. Hyg. 18(3), 411–415 (1969)
World Health Organization (WHO). Zika virus (2018). www.who.int/mediacentre/factsheets/zika/en/
Musso, D., Roche, C., Robin, E., et al.: Potential sexual transmission of Zika virus. Emerg. Infect. Dis. 21(2), 359 (2015)
Du, S., Liu, Y., Liu, J., et al.: Aedes mosquitoes acquire and transmit Zika virus by breeding in contaminated aquatic environments. Nat. Commun. 10(1), 1324 (2019)
Diekmann, O., Heesterbeek, J.A.P., Metz, J.A.: On the definition and the computation of the basic reproduction ratio \(R_{0}\) in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28(4), 365–382 (1990)
Chitnis, N., Cushing, J.M., Hyman, J.M.: Bifurcation analysis of a mathematical model for malaria transmission. SIAM J. Appl. Math. 67(1), 24–45 (2006)
Van den Driessche, P., Watmough, J.: Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 180(1), 29–48 (2002)
Hirsch, W.M., Hanisch, H., Gabriel, J.P.: Differential equation models of some parasitic infections: methods for the study of asymptotic behavior. Commun. Pure Appl. Math. 38, 733–753 (1995)
Castillo-Chavez, C., Song, B.J.: Dynamical models of tuberculosis and their applications. Math. Biosci. Eng. 1(2), 361–404 (2004)
Zhou, X., Cui, J.: Analysis of stability and bifurcation for an SEIV epidemic model with vaccination and nonlinear incidence rate. Nonlinear Dyn. 63(4), 639–653 (2011)
Mukandavire, Z., Liao, S., Wang, J., et al.: Estimating the reproductive numbers for the 2008–2009 cholera outbreaks in Zimbabwe. Proc. Natl. Acad. Sci. 108(21), 8767–8772 (2011)
Finger, F., Genolet, T., Mari, L., et al.: Mobile phone data highlights the role of mass gatherings in the spreading of cholera outbreaks. Proc. Natl. Acad. Sci. 113(23), 6421–6426 (2016)
Tien, J.H., Earn, D.J.D.: Multiple transmission pathways and disease dynamics in a waterborne pathogen model. Bull. Math. Biol. 72(6), 1506–1533 (2010)
Misra, A.K., Singh, V.: A delay mathematical model for the spread and control of water borne diseases. J. Theor. Biol. 301, 49–56 (2012)
Eisenberg, M.C., Robertson, S.L., Tien, J.H.: Identifiability and estimation of multiple transmission pathways in cholera and waterborne disease. J. Theor. Biol. 324, 84–102 (2013)
Liu, W., Hethcote, H.W., Levin, S.A.: Dynamical behavior of epidemiological models with nonlinear incidence rates. J. Math. Biol. 25(4), 359–380 (1987)
Gilchrist, M.A., Coombs, D.: Evolution of virulence: interdependence, constraints, and selection using nested models. Theor. Popul. Biol. 69(2), 145–153 (2006)
Mordecai, E.A., Cohen, J.M., Evans, M.V., et al.: Detecting the impact of temperature on transmission of Zika, dengue, and chikungunya using mechanistic models. PLoS Negl. Trop. Dis. 11(4), e0005568 (2017)
Yang, H.M., Macoris, M.L.G., Galvani, K.C., et al.: Assessing the effects of temperature on the population of Aedes aegypti, the vector of dengue. Epidemiol. Infect. 137(8), 1188–1202 (2009)
Andraud, M., Hens, N., Marais, C., et al.: Dynamic epidemiological models for dengue transmission: a systematic review of structural approaches. PloS One 7(11), e49085 (2012)
Lambrechts, L., Paaijmans, K.P., Fansiri, T., et al.: Impact of daily temperature fluctuations on dengue virus transmission by Aedes aegypti. Proc. Natl. Acad. Sci. 108, 7460–7465 (2011)
Froeschl, G., et al.: Long-term kinetics of Zika virus RNA and antibodies in body fluids of a vasectomized traveller returning from Martinique: a case report. BMC Infect. Dis. 17(1), 55 (2017)
Brazil \(\cdot \) Population. Life expectancy at birth (2020). http://population.city/brazil/#cities
Scott, T.W., Amerasinghe, P.H., Morrison, A.C., et al.: Longitudinal studies of Aedes aegypti (Diptera: Culicidae) in Thailand and Puerto Rico: blood feeding frequency. J. Med. Entomol. 37(1), 89–101 (2000)
Althouse, B.M., Hanley, K.A., Diallo, M., et al.: Impact of climate and mosquito vector abundance on sylvatic arbovirus circulation dynamics in Senegal. Am. J. Trop. Med. Hyg. 92(1), 88–97 (2015)
Lakshmikantham, V., Leela, S., Martynyuk, A.A.: Stability Analysis of Nonlinear Systems. Dekker, New York (1989)
Zhao, X.Q.: Uniform persistence and periodic coexistence states in infinite-dimensional periodic semiflows with applications. Can. Q. Appl. Math. 3(4), 473–495 (1995)
Hale, J.K., Waltman, P.: Persistence in infinite-dimensional systems. SIAM J. Math. Anal. 20(2), 388–395 (1989)
Acknowledgements
We are very grateful to anonymous referees and editor for careful reading and helpful suggestions which led to an improvement of our original manuscript. The work is supported by the National Natural Science Foundation of China (Nos. 11971013, 11571170).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix: A Proof of Lemma 1
Proof
Denote \(\varphi (t)=(S_a(t), I_a(t), S_{m}(t), I_{m}(t), S_{h}(t), I_{h}(t), Z(t))\), and \(\varphi (t)\) satisfies system (2). It follows from system (2) and \(\varphi (0)\ge 0\) that
If \(S_{a}(0)>0,\) \(I_{a}(0)>0,\) \(S_{m}(0)>0,\) \(I_{m}(0)>0,\) \(S_{h}(0)>0,\) \(I_{h}(0)>0\) and \(Z(0)>0,\) then \(S_{a}(t)>0,\) \(I_{a}(t)>0,\) \(S_{m}(t)>0,\) \(I_{m}(t)>0,\) \(S_{h}(t)>0,\) \(I_{h}(t)>0\) and \(Z(t)>0,\) \(\forall ~t>0.\)
From system (2), we can obtain
Since \( S_a+I_a\le K,\) we have
According to the standard comparison theorem (see [49]), we can obtain
If \( S_{m}(0)+I_{m}(0)\le \frac{ \omega K}{\mu _{m}},~ S_{h}(0)+I_{h}(0)\le \frac{\varLambda }{\mu _{h}}\), then \( S_{m}(t)+I_{m}(t)\le \frac{ \omega K}{\mu _{m}},~ S_{h}(t)+I_{h}(t)\le \frac{\varLambda }{\mu _{h}},\) for \(\forall ~t\ge 0.\)
It follows from the seventh equation of system (2) that
Similarly, we have \(Z\le \frac{\sigma \varLambda }{c\mu _{h}}.\) Therefore, \(\varGamma \) is positively invariant of system (2). This completes the proof. \(\square \)
B The simulation and biological explanation of the next-generation matrix \(FV^{-1}\)
Based on the idea of P. van den Driessche and James Watmough in [30], the next-generation matrix \(FV^{-1}\) is explained below.
Let \(x=(x_1,x_2,x_3,x_4,x_5,x_6,x_7)^T,\) where \(x_1=I_a,x_2=I_m,x_3=I_h,x_4=Z,x_5=S_a,x_6=S_m,x_7=S_h.\) Let
\({\mathcal {F}}_i(x)\) represents the input rate of newly infected individuals in the ith compartment, \({\mathcal {V}}_i^{+}(x)\) represents rate of transfer of individuals into the ith compartment by all other means. \({\mathcal {V}}_i^{-}(x)\) represents the rate of transfer of individuals out of the ith compartment. So, the Zika transmission model (1) can be represented as follows
where \({\mathcal {V}}_i(x)={\mathcal {V}}_i^{-}(x)-{\mathcal {V}}_i^{+}(x).\) Let \(F =\left( \frac{\partial {\mathcal {F}}_i}{\partial x_j}(x_0) \right) \) and \(V =\left( \frac{\partial {\mathcal {V}}_i}{\partial x_j}(x_0) \right) \) with \(x_0=(0,0,0,0,A_0,M_0,S_h^0)\), \(1\le i,j\le 4.\) By simple calculation, we can get
Then, we can obtain
\(FV^{-1}\) is given by
To explain the meaning of each element of the matrix \(FV^{-1}\) and provide a meaningful definition of \(R_0\), consider the fate of an infected individual introduced into the kth compartment in a disease free population. The element (j, k) of \(V^{-1}\) is the average time that this infected individual spends in the jth compartment during its lifetime, assuming that the population remains near the DFE \(x_0\) and barring reinfection. The element (i, j) of F is the rate at which infected individuals in the jth compartment produce new infections in the ith compartment. Therefore, the element (i, k) of the product \(FV^{-1}\) is the expected number of new infections in the ith compartment produced by the infected individual originally introduced into the kth compartment. So, following Diekmann et al. [28], P. van den Driessche and James Watmough in [30] call \(FV^{-1}\) the next-generation matrix for model (1) and set
C Proof of Theorem 2
Proof
Theorem 1 shows the local stability of \(E_{01}\). Now, we just prove the global attraction of \(E_{01}\).
Denote \(f^{ \infty }\doteq \limsup \limits _{t\rightarrow +\infty } f(t)\), \(f_{ \infty }\doteq \liminf \limits _{t\rightarrow +\infty }f(t)\), \(\varphi (t)=(S_a(t), I_a(t), S_{m}(t),\) \(I_{m}(t),\) \(S_{h}(t),\) \(I_{h}(t),\) Z(t)), \(\forall ~ t\ge 0\) and \(\varphi (t)\) satisfies system (2). From Lemma 1, we know that \(\varphi (t)\) with initial value \(\varphi (0)\ge 0\) satisfies \(0\le \varphi _{ \infty }\le \varphi ^{ \infty }<+\infty \). From the fluctuation lemma [31], there exists a sequence \(\{t_n\}\) such that \(t_n\rightarrow +\infty ,~\varphi (t_n)\rightarrow \varphi ^{\infty }\) and \(\frac{\mathrm{d}\varphi (t_n)}{\mathrm{d}t}\rightarrow 0\) as \(n\rightarrow +\infty \). Substituting \(t_n\) into system (2), letting \(n\rightarrow +\infty \), and combining \(g(Z)\le g'(0)Z\), we can obtain
Let \(A^{\infty }=S_{a}^{\infty }+I_{a}^{\infty },\) \(M^{\infty }=S_{m}^{\infty }+I_{m}^{\infty }.\) From (12a) and (12b), we have
From (12c) and (12d), we have
Substituting (14) into (13), one has
So,
From (12e), we have
Substituting (12b), (12f) and (12g) into (12d), we get
If \(I_{m}^{\infty }>0\), then, from inequality (18) and (12c), we have
Since \(R_0<1\), we have \( A_{0} < S_{a}^{\infty },\) which is a contradiction since \(S_{a}^{\infty }\le A_{0}.\) Thus, \(I_{m}^{\infty }=0\). From (12f), (12g) and (12b), we have \(I_{h}^{\infty }=0\), \(Z^{\infty }=0\) and \(I_a^{\infty }=0\). So,
Using the fluctuation lemma again, one can obtain
Combining with (15), (16) and (17), we have
This completes the proof. \(\square \)
D Proof of Theorem 4
Proof
Define
First, it is easy to show that \(X_{0}\) is a positively invariant set for system (2), and \(\partial X_{0}\) is closed in \(\varGamma \). Furthermore, it follows from Lemma 1 that system (2) is point dissipative. Denote \(\varphi (0)=(S_a(0), I_a(0), S_{m}(0), I_{m}(0), S_{h}(0), I_{h}(0), Z(0))\). Let \( \varphi (t)=(S_a(t), I_a(t), S_{m}(t),I_{m}(t), S_{h}(t), I_{h}(t), Z(t))\) be a solution of system (2) with \(\varphi (0)\). Set
Next, we will show that \(G_{\partial }={\tilde{G}}_{\partial }.\) It is obvious that \({\tilde{G}} _{\partial } \subseteq G_{\partial },\) then we only need to prove \(G_{\partial } \subseteq {\tilde{G}} _{\partial }.\) Assume \(\varphi (0)\in G_{\partial }\). Then, \(\varphi (t)\in \partial X_{0}, \forall ~t\ge 0\). We will prove that \(I_{a}(t)=I_{m}(t)=I_{h}(t)=Z(t)=0, \forall ~t\ge 0.\) Suppose not. Then, there exists \(t_0\ge 0\) such that at least one of \(I_a(t_0)\), \(I_m(t_0)\), \(I_h(t_0)\) and \(Z(t_0)\) is not zero. Without loss of generality, assume that \(I_{a}(t_{0})>0\), \(I_{m}(t_{0})=I_{h}(t_{0})=Z(t_{0})=0\). Then, from system (2), we have, for \(\forall ~t>t_{0}\),
This implies that \(\varphi (t)=(S_a(t), I_a(t), S_{m}(t), I_{m}(t), S_{h}(t), I_{h}(t), Z(t))\notin \partial X_{0}\) for \(t>t_{0}\). which contradicts \(\varphi (t)\in \partial X_{0}, \forall ~t\ge 0\). So, one can obtain \(G_{\partial } \subseteq {\tilde{G}}_{\partial }.\) So \(G_{\partial }={\tilde{G}}_{\partial }.\) Therefore, it is clear that there are two equilibria \(E_{00}\) and \(E_{01}\) in \(G_{\partial }\).
Next, we will show that \(E_{00}\) and \(E_{01}\) are isolated invariant sets in \(\varGamma \). We need show that \(W^{s}(E_{00})\cap X_{0}=\emptyset \) and \(W^{s}(E_{01})\cap X_{0}=\emptyset \), where \(W^{s}(E_{00})\) and \(W^{s}(E_{01})\) are stable manifolds of \(E_{00}\) and \(E_{01}\), respectively, that is, there exist positive constants \(\delta \) and \({\bar{\delta }}\) such that for any solution \(\varPhi _{t}(\varphi (0))\) of system (2) with the initial value \(\varphi (0)\in X_{0}\), we have
where d is a distant function in \(X_{0}\). Here, we only show \(\limsup \limits _{t\rightarrow +\infty }d(\varPhi _{t}(\varphi (0)),E_{01})\) \(\ge \delta \). If not, then \(\limsup \limits _{t\rightarrow +\infty }d(\varPhi _{t}(\varphi (0)),\) \(E_{01})< \delta \), that is, there exists \(t_1>0\), such that \(A_{0}-\delta< S_{a}< A_{0}+\delta \), \(M_{0}-\delta< S_{m}< M_{0}+\delta \), \(S_h^0-\delta< S_{h}< S_h^0+\delta \), \(0< I_{a}<\delta \), \(0< I_{m}<\delta \), \(0< I_{h}<\delta \) and \(0< Z<\delta \) for \(\forall ~t>t_1\). Next, we just prove the incidence function \(g(Z)=\beta _{3}\frac{Z}{k+Z}.\) For \(g(Z)=\beta _3 Z\), its proof is similar. For the incidence function \(g(Z)=\beta _{3}\frac{Z}{k+Z},\) one can get, for \(\forall \) \(t>t_1\),
Consider an auxiliary system as follows
where \(U=(U_{1},U_{2},U_{3},U_{4})^{T}\) and
We know \(B(0)=F-V\). Note that \(B(\delta )\) is irreducible and has nonnegative off-diagonal elements. Then, \(s(B(\delta ))\) is a simple eigenvalue of \(B(\delta )\) with a positive eigenvector (see Theorem A.5 in [50]). Based on Theorem 2 in [30], we have \(s(B(0))>0\) when \(R_{0}>1.\) We know that \(B(\delta )\) is a continuous for small \(\delta \). Thus, there exists \(\delta \) small enough, such that \(s(B(\delta ))>0\). So, there exists a positive eigenvalue of \(B(\delta )\) corresponding to a positive eigenvector. Let \(U(t)=(U_{1}(t),U_{2}(t),U_{3}(t),U_{4}(t))^{T}\) be positive solution of auxiliary system (19). Then, \(U_i(t)\) is strictly increasing with \(U_i(t)\rightarrow +\infty \) as \(t\rightarrow +\infty \), \(i=1,\ldots ,4\). We know that system (19) is quasi-monotonic. Using the comparison principle, we have
This contradicts with our assumption. Then, \(E_{01}\) is an isolated invariant set in \(\varGamma \) and \(W^{s}(E_{01})\cap X_{0}=\emptyset \). Similarly, we can get that \(E_{00}\) is an isolated invariant set in \(\varGamma \) and \(W^{s}(E_{00})\cap X_{0}=\emptyset .\) Clearly, every orbit in \(G_{\partial }\) converges to either \(E_{00}\) or \(E_{01}\), and \(\{E_{00}\}\cup \{E_{01}\}\) is an acyclic covering of \(G_{\partial }\). From Theorem 4.1 in [51], system with incidence function \(g(Z)=\beta _3\frac{Z}{k+Z}\) or \(\beta _3 Z\) is uniformly persistent if \(R_{0}>1\). This completes the proof. \(\square \)
E The expressions of \(a_{i}\), \(b_{i}\) and \(c_{i}\), \(i=1, 2, 3\)
where
Rights and permissions
About this article
Cite this article
Wang, L., Zhao, H. Modeling and dynamics analysis of Zika transmission with contaminated aquatic environments. Nonlinear Dyn 104, 845–862 (2021). https://doi.org/10.1007/s11071-021-06289-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-021-06289-3
Keywords
- Zika virus
- Contaminated aquatic environment
- Stability
- Backward bifurcation
- Saddle-node bifurcation
- Hopf bifurcation