Abstract
The formation of hypoxic water zones in marine ecosystems across the world is a growing concern among marine scientists and regulatory agencies that focus on marine resource management. Nutrient-rich water with excessive nitrogen content is widely recognized as a key anthropogenic cause for the development of hypoxic zones. While the dissolved oxygen level indicates the presence of current hypoxic water conditions, the aquatic nitrogen concentration indicates the possibility of formation of hypoxic zones in future time periods. Hence, understanding the effects of both these variables on current and future fish population is important for improved water quality management and sustainability of marine resources. In this paper, we estimate the temporal effects of these two key variables on lobster harvest from three contiguous fishing zones in the Long Island Sound that vary in ambient water quality. We find there is no contemporaneous effect of these variables on harvest in both the hypoxic fishing zone and the two non-hypoxic zones. While there is some evidence of lagged effects of these variables on harvest, no systematic pattern emerges in these effects that distinguishes the hypoxic fishing zone with non-hypoxic zones.

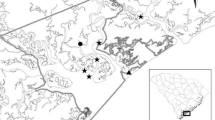
Source: Connecticut Department of Energy and Environmental Protection
Similar content being viewed by others
Notes
The map with the station locations and the data for all environmental variables were provided by Matthew Lyman at CT Department of Energy and Environmental Protection. The fisheries data were provided by Matthew Gates at CT DEEP. The authors are very grateful for the support they received from CT DEEP.
References
Balcom N, Howell P (2006) Responding to a resource disaster: American lobsters in Long Island Sound 1999–2004. CT Sea grant report
Borsuk M, Stow C, Reckhow K (2004) A Bayesian hierarchical model to predict benthic oxygen demand from organic matter loading in estuaries and coastal zones. Ecol Model 143:165–181
Breitburg DL (2002) Effects of hypoxia and the balance between hypoxia and enrichment on coastal fishes and fisheries. Estuaries 25:767–781
Breitburg DL, Hondorp DW, Davias LA, Diaz RJ (2009) Hypoxia, nitrogen, and fisheries: integrating effects across local and global landscapes. Ann Rev Mar Sci 1:329–349
Diaz RJ, Solow A (1999) Ecological and economic consequences of hypoxia. Topic 2. Gulf of Mexico hypoxia assessment. NOAA Coastal Ocean Program Decision Analysis Series. NOAA COP, Silver Springs, MD
Eby LA, Crowder LB (2002) Hypoxia-based habitat compression in the Neuse River Estuary: context-dependent shifts in behavioral avoidance thresholds. Can J Fish Aquat Sci 59:952–965
Huang L, Smith M (2011) Management of an annual fishery in the presence of ecological stress: the case of shrimp and hypoxia. Ecol Econ 70:688–697
Huang L, Nichols LAB, Craig JK, Smith MD (2012) Measuring welfare losses from hypoxia: the case of North Carolina Brown Shrimp. Mar Resour Econ 27:3–24
Miller DC, Poucher SL, Coiro L (2002) Determination of lethal dissolved oxygen levels for selected marine and estuarine fishes, crustaceans, and a bivalve. Mar Biol 140:287–296
Nixon SW, Buckley BA (2002) “A strikingly rich zone”—nutrient enrichment and secondary production in coastal marine ecosystems. Estuaries 25:782–796
Parker CA, O’Reilly JE (1991) Oxygen depletion in Long Island Sound: a historical perspective. Estuaries 14:248–254
Peterson CH, Summerson HC, Thomson E, Lenihan HS, Grabowski J, Manning L, Micheli F, Johnson G (2000) Synthesis of linkages between benthic and fish communities as a key to protecting essential fish habitat. Bull Mar Sci 66:759–774
Ribaudo MO, Heimlich R, Peters M (2005) Nitrogen sources and Gulf hypoxia: potential for environmental credit trading. Ecol Econ 52:159–168
Smith MD, Crowder LB (2005) Valuing ecosystem services with fishery rents: a lumped-parameter approach to hypoxia in the Neuse River Estuary. FEEM work paper no. 115.05. SSRN: http://ssrn.com/abstract=825587
Stierhoff KL, Targett TE, Power JH (2009) Hypoxia-induced growth limitation of juvenile fishes in an estuarine nursery: assessment of small-scale temporal dynamics using RNA:DNA. Can J Fish Aquat Sci 66:1033–1047
Taylor DL, Eggleston DB (2000) Effects of hypoxia on an estuarine predator-prey interaction: foraging behavior and mutual interference in the blue crab Callinectes sapidus and the infaunal clam prey Mya arenaria. Mar Ecol Prog Ser 196:221–237
Thomas P, Rahman S, Khan IA, Kummer JA (2007) Widespread endocrine disruption and reproductive impairment in an estuarine fish population exposed to seasonal hypoxia. Proc R Soc B 274:2693–2702
Wu J, Tanaka K (2005) Reducing nitrogen runoff from the Upper Mississippi River Basin to control hypoxia in the Gulf of Mexico: easements or Taxes? Mar Resour Econ 20:121–144
Zimmerman R, Nance J, Wiams J (1996) Trends in shrimp catch in the hypoxic area of the northern Gulf of Mexico. Galveston Laboratory, National Marine Fisheries Service. In Proceedings of the first Gulf of Mexico hypoxia management conference. EPA-55-R-97-001. Washington, DC
Acknowledgments
We are grateful to Connecticut Department of Energy and Environmental Protection for sharing the data for the empirical analysis with us. We also thank our anonymous referees for their valuable feedback.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mukherjee, Z., Dey, D.K. & Gupta, R. Time series effects of dissolved oxygen and nitrogen on Long Island Sound lobster harvest. Nat Hazards 84, 1849–1858 (2016). https://doi.org/10.1007/s11069-016-2522-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11069-016-2522-7