Abstract
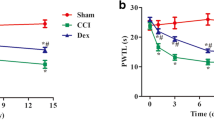
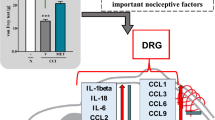
This study was designed to investigate the analgesic effect of perineural injection of BoNT/A on neuropathic pain induced by sciatic nerve chronic constriction injury (CCI) and possible mechanisms. SD rats were randomly divided into Sham group, CCI group and BoNT/A group. Paw mechanical withdrawal threshold (pMWT) and paw thermal withdrawal latency (pTWL) of each group were detected at different time points after surgery. The expression of myelin markers, autophagy markers and NLRP3 inflammasome-related molecules in injured sciatic nerves were examined at 12 days after surgery. Moreover, C-fiber evoked potential in spinal dorsal horn was recorded. The expression of SNAP-25, neuroinflammation and synaptic plasticity in spinal dorsal horn of each group were examined. Then rats treated with BoNT/A were randomly divided into DMSO group and Wnt agonist group to further explore the regulatory effect of BoNT/A on Wnt pathway. We found that pMWT and pTWL of ipsilateral paw were significantly decreased in CCI group compared with Sham group, which could be improved by perineural injection of BoNT/A at days 7, 9 and 12 after surgery. The peripheral analgesic mechanisms of perineural injection of BoNT/A might be related to the protective effect on myelin sheath by inhibiting NLRP3 inflammasome and promoting autophagy flow, while the central analgesic mechanisms might be associated with inhibition of neuroinflammation and synaptic plasticity in spinal dorsal horn due to inhibiting SNAP-25 and Wnt pathway. As a new route of administration, perineural injection of BoNT/A can relieve CCI induced neuropathic pain probably via both peripheral and central analgesic mechanisms.














Similar content being viewed by others
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BoNT/A:
-
Botulinum toxin A
- CCI:
-
Chronic constriction injury
- pMWT:
-
Paw mechanical withdrawal threshold
- pTWL:
-
Paw thermal withdrawal latency
- LC3:
-
Microtubule-associated protein-1 light chain 3
- NLRP3:
-
NOD-like receptor family pyrin domain-containing 3
- SNAP-25:
-
Synaptosomal-associated protein 25 kDa
- LTP:
-
Long-term potentiation
- NMDAR:
-
N-methyl-d-aspartate receptors
- SD:
-
Sprague Dawley
References
Jacobson L, Dengler J, Moore AM (2020) Nerve entrapments. Clin Plast Surg 47:267–278
Finnerup NB, Attal N, Haroutounian S, McNicol E, Baron R, Dworkin RH, Gilron I, Haanpaa M, Hansson P, Jensen TS, Kamerman PR, Lund K, Moore A, Raja SN, Rice AS, Rowbotham M, Sena E, Siddall P, Smith BH, Wallace M (2015) Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol 14:162–173
Apalla Z, Sotiriou E, Lallas A, Lazaridou E, Ioannides D (2013) Botulinum toxin A in postherpetic neuralgia: a parallel, randomized, double-blind, single-dose, placebo-controlled trial. Clin J Pain 29:857–864
Yuan RY, Sheu JJ, Yu JM, Chen WT, Tseng IJ, Chang HH, Hu CJ (2009) Botulinum toxin for diabetic neuropathic pain: a randomized double-blind crossover trial. Neurology 72:1473–1478
Wu CJ, Lian YJ, Zheng YK, Zhang HF, Chen Y, Xie NC, Wang LJ (2012) Botulinum toxin type A for the treatment of trigeminal neuralgia: results from a randomized, double-blind, placebo-controlled trial. Cephalalgia 32:443–450
Fabregat G, De Andres J, Villanueva-Perez VL, Asensio-Samper JM (2013) Subcutaneous and perineural botulinum toxin type a for neuropathic pain: a descriptive review. Clin J Pain 29:1006–1012
Matak I, Bolcskei K, Bach-Rojecky L, Helyes Z (2019) Mechanisms of botulinum toxin type A action on pain. Toxins (Basel) 11
Seo M, Lim D, Kim S, Kim T, Kwon BS, Nam K (2021) Effect of botulinum toxin injection and extracorporeal shock wave therapy on nerve regeneration in rats with experimentally induced sciatic nerve injury. Toxins (Basel) 13
Marinelli S, Luvisetto S, Cobianchi S, Makuch W, Obara I, Mezzaroma E, Caruso M, Straface E, Przewlocka B, Pavone F (2010) Botulinum neurotoxin type A counteracts neuropathic pain and facilitates functional recovery after peripheral nerve injury in animal models. Neuroscience 171:316–328
Zychowska M, Rojewska E, Makuch W, Luvisetto S, Pavone F, Marinelli S, Przewlocka B, Mika J (2016) Participation of pro- and anti-nociceptive interleukins in botulinum toxin A-induced analgesia in a rat model of neuropathic pain. Eur J Pharmacol 791:377–388
Buntragulpoontawee M, Chang KV, Vitoonpong T, Pornjaksawan S, Kitisak K, Saokaew S, Kanchanasurakit S (2020) The effectiveness and safety of commonly used injectates for ultrasound-guided hydrodissection treatment of peripheral nerve entrapment syndromes: a systematic review. Front Pharmacol 11:621150
Meyer-Friessem CH, Eitner LB, Kaisler M, Maier C, Vollert J, Westermann A, Zahn PK, Avila Gonzalez CA (2019) Perineural injection of botulinum toxin-A in painful peripheral nerve injury - a case series: pain relief, safety, sensory profile and sample size recommendation. Curr Med Res Opin 35:1793–1803
Xiao PY, Chen JY, Zeng Q, Huang Z, Huang BX, Yu J, Liao SJ (2022) UNC5B overexpression alleviates peripheral neuropathic pain by stimulating netrin-1-dependent autophagic flux in schwann cells. Mol Neurobiol 59:5041–5055
Xu J, Wei X, Gao F, Zhong X, Guo R, Ji Y, Zhou X, Chen J, Yao P, Liu X, Wei X (2020) Nicotinamide adenine dinucleotide phosphate oxidase 2-derived reactive oxygen species contribute to long-term potentiation of C-fiber-evoked field potentials in spinal dorsal horn and persistent mirror-image pain following high-frequency stimulus of the sciatic nerve. Pain 161:758–772
Xiao L, Cheng J, Zhuang Y, Qu W, Muir J, Liang H, Zhang D (2013) Botulinum toxin type A reduces hyperalgesia and TRPV1 expression in rats with neuropathic pain. Pain Med 14:276–286
Hughey S, Campbell D, Rapp-Santos K, Cole J, Booth G, Longwell J, Stedje-Larsen E (2022) Refining the rat sciatic nerve block: a novel ultrasound-guided technique. Lab Anim 56:191–195
He JJ, Wei XM, Dou ZL, Zhang JS, Wei ZH, Zhang WX, Jiang L (2021) Ultrasound-guided nerve hydrodissection with 5% dextrose 4 weeks after steroid injection in treatment of carpal tunnel syndrome: a retrospective study. Front Neurol 12:782319
Xu T, Li D, Zhou X, Ouyang HD, Zhou LJ, Zhou H, Zhang HM, Wei XH, Liu G, Liu XG (2017) Oral application of magnesium-L-threonate attenuates vincristine-induced allodynia and hyperalgesia by normalization of tumor necrosis factor-alpha/nuclear factor-kappaB signaling. Anesthesiology 126:1151–1168
Huang Z, Xiao PY, Chen JY, Zeng Q, Huang BX, Yu J, Liao SJ (2022) Mammalian sterile 20-like kinase 1 mediates neuropathic pain associated with its effects on regulating mitophagy in Schwann cells. Oxid Med Cell Longev 2022:3458283
Biasizzo M, Kopitar-Jerala N (2020) Interplay between NLRP3 inflammasome and autophagy. Front Immunol 11:591803
Liu XG (2022) Normalization of neuroinflammation: a new strategy for treatment of persistent pain and memory/emotional deficits in chronic pain. J Inflamm Res 15:5201–5233
Sheehan GD, Martin MK, Young VA, Powell R, Bhattacharjee A (2021) Thermal hyperalgesia and dynamic weight bearing share similar recovery dynamics in a sciatic nerve entrapment injury model. Neurobiol Pain 10:100079
Lam KHS, Hung CY, Chiang YP, Onishi K, Su DCJ, Clark TB, Reeves KD (2020) Ultrasound-guided nerve hydrodissection for pain management: rationale, methods, current literature, and theoretical mechanisms. J Pain Res 13:1957–1968
Tumpaj T, Potocnik Tumpaj V, Albano D, Snoj Z (2022) Ultrasound-guided carpal tunnel injections. Radiol Oncol 56:14–22
Moon YE, Choi JH, Park HJ, Park JH, Kim JH (2016) Ultrasound-guided nerve block with botulinum toxin Type A for intractable neuropathic pain. Toxins (Basel) 8
Kwon SY, Jun EH, Park SJ, Kim Y (2022) Botulinum toxin injection strategy of intractable and relapsed piriformis syndrome: a case report. Medicine (Baltimore) 101:e30950
Cobianchi S, Jaramillo J, Luvisetto S, Pavone F, Navarro X (2017) Botulinum neurotoxin A promotes functional recovery after peripheral nerve injury by increasing regeneration of myelinated fibers. Neuroscience 359:82–91
Gomez-Sanchez JA, Carty L, Iruarrizaga-Lejarreta M, Palomo-Irigoyen M, Varela-Rey M, Griffith M, Hantke J, Macias-Camara N, Azkargorta M, Aurrekoetxea I, De Juan VG, Jefferies HB, Aspichueta P, Elortza F, Aransay AM, Martinez-Chantar ML, Baas F, Mato JM, Mirsky R, Woodhoo A, Jessen KR (2015) Schwann cell autophagy, myelinophagy, initiates myelin clearance from injured nerves. J Cell Biol 210:153–168
Marinelli S, Nazio F, Tinari A, Ciarlo L, D’Amelio M, Pieroni L, Vacca V, Urbani A, Cecconi F, Malorni W, Pavone F (2014) Schwann cell autophagy counteracts the onset and chronification of neuropathic pain. Pain 155:93–107
Li W, Liang J, Li S, Wang L, Xu S, Jiang S, Song M, Meng H, Zhai D, Tang L, Yang Y, Zhang L, Zhang B (2022) Research progress of targeting NLRP3 inflammasome in peripheral nerve injury and pain. Int Immunopharmacol 110:109026
Cui M, Liang J, Xu D, Zhao L, Zhang X, Zhang L, Ren S, Liu D, Niu X, Zang YJ, Zhang B (2020) NLRP3 inflammasome is involved in nerve recovery after sciatic nerve injury. Int Immunopharmacol 84:106492
Luvisetto S (2021) Botulinum neurotoxins in central nervous system: an overview from animal models to human therapy. Toxins (Basel) 13
Liu S, Liu YP, Huang ZJ, Zhang YK, Song AA, Ma PC, Song XJ (2015) Wnt/Ryk signaling contributes to neuropathic pain by regulating sensory neuron excitability and spinal synaptic plasticity in rats. Pain 156:2572–2584
Mika J, Rojewska E, Makuch W, Korostynski M, Luvisetto S, Marinelli S, Pavone F, Przewlocka B (2011) The effect of botulinum neurotoxin A on sciatic nerve injury-induced neuroimmunological changes in rat dorsal root ganglia and spinal cord. Neuroscience 175:358–366
Zhang YK, Huang ZJ, Liu S, Liu YP, Song AA, Song XJ (2013) WNT signaling underlies the pathogenesis of neuropathic pain in rodents. J Clin Invest 123:2268–2286
Acknowledgements
We are very grateful to our corporate sponsor Lanzhou Biotechnique Development Co., LTD.
Funding
This study was supported by grants from the Natural Science Foundation of Guangdong Province (2021A1515010313), the Guangdong Provincial Key Laboratory of Diagnosis and Treatment of Major Neurological Diseases (2020B1212060017), Guangdong Provincial Clinical Research Center for Neurological Diseases (2020B1111170002), Southern China International Joint Research Center for Early Intervention and Functional Rehabilitation of Neurological Diseases (2015B050501003 and 2020A0505020004), Guangdong Provincial Engineering Center for Major Neurological Disease Treatment, Guangdong Provincial Translational Medicine Innovation Platform for Diagnosis and Treatment of Major Neurological Disease, Guangzhou Clinical Research and Translational Center for Major Neurological Diseases (201604020010), Guangzhou Science and Technology Program (20220610195), a crosswise project of First Affiliated Hospital, Sun Yat-sen University (2020027).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Conceived and designed the experiments: WZ and LJ. Material preparation, data collection and analysis: JH, MW and ZS. Wrote the paper: JH and XW. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors confirm that there is no conflict of interest.
Ethical Approval
This study was approved by Institutional Animal Care and Use Committee, Sun Yat-Sen University (Approval NO. SYSU-IACUC-2022-000798).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, Jj., Wei, Xm., Wu, Ml. et al. Analgesic Effect of Perineural Injection of BoNT/A on Neuropathic Pain Induced by Chronic Constriction Injury of Sciatic Nerve in Rats. Neurochem Res 48, 2161–2174 (2023). https://doi.org/10.1007/s11064-023-03893-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-023-03893-0