Abstract
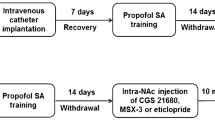
Propofol has been proven to be potentially abused by humans and laboratory animals; however, studies that have examined propofol relapse behavior are limited, and its underlying mechanism remains unclear. In this study, we examined whether basolateral amygdala-specific or systematic administration of the dopamine receptor antagonist alters cue-induced propofol-seeking behaviors in a rat model. Male Sprague–Dawley rats first received 14 days of propofol self-administration training, where active nose poke resulted in the delivery of propofol infusion paired with a tone and light cues. After 1–30 days of forced abstinence, the cue-induced propofol-seeking behaviors were tested in the operant chamber. We demonstrated, for the first time, after a few days of withdrawal from intravenous bolus administration of propofol, propofol-related cues could induce robust reinstatement of drug-seeking behavior. Systematic administration of dopamine D1 receptor antagonist (SCH-23390) or dopamine D2 receptor antagonist (spiperone) inhibited propofol relapse behavior induced by drug-related cues. Furthermore, we show that microinfusion of SCH-23390 into basolateral amygdala dose-dependently attenuated cue-induced propofol drug-seeking behavior, whereas infusion of spiperone had no effect on the propofol relapse behavior. Our results reveal the involvement of dopamine receptors within the basolateral amygdala in the cue-induced propofol relapse behavior in rats.



Similar content being viewed by others
References
Wischmeyer PE, Johnson BR, Wilson JE et al (2007) A survey of propofol abuse in academic anesthesia programs. Anesth Analg 105:1066–1071
Fry RA, Fry LE, Castanelli DJ (2015) A retrospective survey of substance abuse in anaesthetists in Australia and New Zealand from 2004 to 2013. Anaesth Intensive Care 43:111–117
Zacny JP, Lichtor JL, Coalson DW, Finn RS, Uitvlugt AM, Glosten B, Flemming DC, Apfelbaum JL (1992) Subjective and psychomotor effects of subanesthetic doses of propofol in healthy volunteers. Anesthesiology 76:696–702
Zacny JP, Lichtor L, Zaragoza JG, Coalson DW, Uitvlugt AM, Flemming DC, Binstock WB, Cutter T, Apfelbaum JL (1993) Assessing the behavioral effects and abuse potential of propofol bolus injections in healthy volunteers. Drug Alcohol Depend 32:45–57
Lian Q, Wang B, Zhou W, Jin S, Xu L, Huang Q, Lai M, Lu K, Tang L, Zhu H, Lin H, Zhang F, Cao H, Li J, Chen JF, Ge RS (2013) Self-administration of propofol is mediated by dopamine D1 receptors in nucleus accumbens in rats. Neuroscience 231:373–383
Bonnet U, Scherbaum N (2012) Craving dominates propofol addiction of an affected physician. J Psychoact Drugs 44(2):186–190
Follette JW, Farley WJ (1992) Anesthesiologist addicted to propofol. Anesthesiology 77:817–818
Soyka M, Schutz CG (1997) Propofol dependency. Addiction 92:1369–1370
Zhou W, Zhang F, Liu H, Tang S, Lai M, Zhu H, Kalivas PW (2009) Effect of training and withdrawal periods on heroin seeking induced by conditioned cue in an animal of model of relapse. Psychopharmacology 203:677–684
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O’Brien CP (1999) Limbic activation during cue-induced cocaine craving. Am J Psychiatry 156:11–18
Duncan E, Boshoven W, Harenski K, Fiallos A, Tracy H, Jovanovic T, Hu X, Drexler K, Kilts C (2007) An fMRI study of the interaction of stress and cocaine cues on cocaine craving in cocaine-dependent men. Am J Addict 16:174–182
Hamlin AS, Clemens KJ, McNally GP (2008) Renewal of extinguished cocaine-seeking. Neuroscience 151:659–670
Pelloux Y, Hoots JK, Cifani C, Adhikary S, Martin J, Minier-Toribio A, Bossert JM, Shaham Y (2018) Context-induced relapse to cocaine seeking after punishment-imposed abstinence is associated with activation of cortical and subcortical brain regions. Addict Biol 23:699–712
Fuchs RA, Evans KA, Ledford CC, Parker MP, Case JM, Mehta RH, See RE (2005) The role of the dorsomedial prefrontal cortex, basolateral amygdale, and dorsal hippocampus contextual reinstatement of cocaine seeking in rats. Neuropsychopharmacology 30:296–309
Chaudhri N, Woods CA, Sahuque LL, Gill TM, Janak PH (2013) Unilateral inactivation of the basolateral amygdala attenuates context-induced renewal of Pavlovian-conditioned alcohol-seeking. Eur J Neurosci 38:2751–2761
Stuber GD, Sparta DR, Stamatakis AM, van Leeuwen WA, Hardjoprajitno JE, Cho S, Tye KM, Kempadoo KA, Zhang F, Deisseroth K, Bonci A (2011) Excitatory transmission from the amygdala to nucleus accumbens facilitates reward seeking. Nature 475:377–380
Ambroggi F, Ishikawa A, Fields HL, Nicola SM (2008) Basolateral amygdala neurons facilitate reward seeking behavior by exciting nucleus accumbens neurons. Neuron 59:648–661
Yang B, Wang BF, Lai MJ, Zhang FQ, Yang XW, Zhou WH, Lian QQ (2011) Differential involvement of GABAA and GABAB receptor in propofol self-administration in rats. Acta Pharmacol Sin 32:1460–1465
Weiss F, Maldonado-Vlaar CS, Parsons LH, Kerr TM, Smith DL, Ben-Shahar O (2000) Control of cocaine-seeking behavior by drug-associated stimuli in rats: effects on recovery of extinguished operant-responding and extracellular dopamine levels in amygdala and nucleus accumbens. Proc Natl Acad Sci USA 97:4321–4326
Meador-Woodruff JH, Mansour A, Healy DJ, Kuehn R, Zhou QY, Bunzow JR, Akil H, Civelli O, Watson SJ Jr (1991) Comparison of the distributions of D1 and D2 dopamine receptor mRNAs in rat brain. Neuropsychopharmacology 5:231–242
Scibilia RJ, Lachowicz JE, Kilts CD (1992) Topographic nonoverlapping distribution of D1 and D2 dopamine receptors in the amygdaloid nuclear complex of the rat brain. Synapse 11:146–154
Kröner S, Rosenkranz JA, Grace AA, Barrionuevo G (2005) Dopamine modulates excitability of basolateral amygdala neurons in vitro. J Neurophysiol 93:1598–1610
Lintas A, Chi N, Lauzon NM, Bishop SF, Sun N, Tan H, Laviolette SR (2012) Inputs from the basolateral amygdala to the nucleus accumbens shell control opiate reward magnitude via differential dopamine D1 or D2 receptor transmission. Eur J Neurosci 35:279–290
See RE, Kruzich PJ, Grimm JW (2001) Dopamine, but not glutamate, receptor blockade in the basolateral amygdala attenuates conditioned reward in a rat model of relapse to cocaine-seeking behavior. Psychopharmacology 154:301–310
Khaled MA, Pushparaj A, Di Ciano P, Diaz J, Le Foll B (2014) Dopamine D3 receptors in the basolateral amygdala and the lateral habenula modulate cue-induced reinstatement of nicotine seeking. Neuropsychopharmacology 39:3049–3058
Zhou W, Liu H, Zhang F, Tang S, Zhu H, Lai M, Kalivas PW (2007) Role of acetylcholine transmission in nucleus accumbens and ventral tegmental area in heroin-seeking induced by conditioned cues. Neuroscience 144:1209–1218
Paxinos G, Watson D (1986) The rat brain in stereotaxic coordinates. Academic Press, New York
Zhou W, Zhang F, Tang S, Liu H, Gu J, Yang G (2005) The dissociation of heroin-seeking patterns induced by contextual, discriminative, or discrete conditioned cues in a model of relapse to heroin in rats. Psychopharmacology 181:197–206
Liu X, Weiss F (2002) Additive effect of stress and drug cues on reinstatement of ethanol seeking: exacerbation by history of dependence and role of concurrent activation of corticotropin-releasing factor and opioid mechanisms. J Neurosci 22:7856–7861
Shaham Y, Miczek KA (2003) Reinstatement-toward a model of relapse. Psychopharmacology 168:1–2
Koob GF, Le Moal M (1997) Drug abuse: hedonic homeostatic dysregulation. Science 278:52–58
O’Brien CP, Childress AR, McLellan AT, Ehrman R (1992) Classical conditioning in drug-dependent humans. Ann NY Acad Sci 654:400–415
Bossert JM, Marchant NJ, Calu DJ, Shaham Y (2013) The reinstatement model of drug relapse: recent neurobiological findings, emerging research topics, and translational research. Psychopharmacology 229:453–476
Epstein DH, Preston KL (2003) The reinstatement model and relapse prevention: a clinical perspective. Psychopharmacology 168:31–41
Katz JL, Higgins ST (2003) The validity of the reinstatement model of craving and relapse to drug use. Psychopharmacology 168:21–30
Loweth JA, Scheyer AF, Milovanovic M, LaCrosse AL, Flores-Barrera E, Werner CT, Li X, Ford KA, Le T, Olive MF, Szumlinski KK, Tseng KY, Wolf ME (2014) Synaptic depression via mGluR1 positive allosteric modulation suppresses cue-induced cocaine craving. Nat Neurosci 17:73–80
Li X, Rubio FJ, Zeric T, Bossert JM, Kambhampati S, Cates HM, Kennedy PJ, Liu QR, Cimbro R, Hope BT, Nestler EJ, Shaham Y (2015) Incubation of methamphetamine craving is associated with selective increases in expression of Bdnf and trkb, glutamate receptors, and epigenetic enzymes in cue-activated fos-expressing dorsal striatal neurons. J Neurosci 35:8232–8244
Airavaara M, Pickens CL, Stern AL, Wihbey KA, Harvey BK, Bossert JM, Liu QR, Hoffer BJ, Shaham Y (2011) Endogenous GDNF in ventral tegmental area and nucleus accumbens does not play a role in the incubation of heroin craving. Addict Biol 16:261–272
Grimm JW, Hope BT, Wise RA, Shaham (2001) Neuroadaptaion. Incubation of cocaine craving after withdrawal. Nature 412:141–142
Bienkowski P, Rogowski A, Korkosz A, Mierzejewski P, Radwanska K, Kaczmarek L, Bogucka-Bonikowska A, Kostowski W (2004) Time-dependent changes in alcohol-seeking behavior during abstinence. Eur Neuropsychopharmacol 14:355–360
Abdolahi A, Acosta G, Breslin FJ, Hemby SE, Lynch WJ (2010) Incubation of nicotine seeking is associated with enhanced protein kinase A-regulated signaling of dopamine-and cAMP-regulated phosphoprotein of 32 kDa in the insular cortex. Eur J Neurosci 31:733–741
Stefanik MT, Kalivas PW (2013) Optogenetic dissection of basolateral amygdala projections during cue-induced reinstatement of cocaine seeking. Front Behav Neurosci 7:213. https://doi.org/10.3389/fnbeh.2013.00213
Weiss F, Maldonado-Vlaar CS, Parsons LH et al (2000) Control of cocaine-seeking behavior by drug-associated stimuli in rats: effects on recovery of extinguished operant-responding and extracellular dopamine levels in amygdala and nucleus accumbens. Proc Natl Acad Sci USA 97:4321–4326
Li YQ, Li FQ, Wang XY, Wu P, Zhao M, Xu CM, Shaham Y, Lu L (2008) Central amygdala extracellular signal-regulated kinase signaling pathway is critical to incubation of opiate craving. J Neurosci. 28(49):13248–13257. https://doi.org/10.1523/JNEUROSCI.3027-08.2008
Lu L, Hope BT, Dempsey J, Liu SY, Bossert JM, Shaham Y (2005) Central amygdala ERK signaling pathway is critical to incubation of cocaine craving. Nat Neurosci 8(2):212–219
Berglind WJ, Case JM, Parker MP, Fuchs RA, See RE (2006) Dopamine D1 or D2 receptor antagonism within the basolateral amygdala differentially alters the acquisition of cocaine-cue associations necessary for cue-induced reinstatement of cocaine-seeking. Neuroscience 137:699–706
Ciccocioppo R, Sanna PP, Weiss F (2001) Cocaine-predictive stimulus induces drug-seeking behavior and neural activation in limbic brain regions after multiple months of abstinence: reversal by D(1) antagonists. Proc Natl Acad Sci USA 98:1976–1981
Wells AM, Arguello AA, Xie X, Blanton MA, Lasseter HC, Reittinger AM, Fuchs RA (2013) Extracellular signal-regulated kinase in the basolateral amygdala, but not the nucleus accumbens core, is critical for context-response-cocaine memory reconsolidation in rats. Neuropsychopharmacology 38:753–762
Li Z, Luan W, Chen Y, Chen M, Dong Y, Lai B, Ma L, Zheng P (2011) Chronic morphine treatment switches the effect of dopamine on excitatory synaptic transmission from inhibition to excitation in pyramidal cells of the basolateral amygdala. J Neurosci 31:17527–17536
Bissiere S, Humeau Y, Luthi A (2003) Dopamine gates LTP induction in lateral amygdala by suppressing feedforward inhibition. Nat Neurosci 6:587–592
Ferrari F, Giuliani D (1995) Behavioural assessment in rats of the antipsychotic potential of the potent dopamine D2 receptor antagonist, (-)eticlopride. Pharmacol Res 31:261–267
Cervo L, Carnovali F, Stark JA, Mennini T (2003) Cocaine-seeking behavior in response to drug-associated stimuli in rats: involvement of D3 and D2 dopamine receptors. Neuropsychopharmacology 28:1150–1159
Liu X, Jernigen C, Gharib M (2010) Effects of dopamine antagonists on drug cue-induced reinstatement of nicotine-seeking behavior in rats. Behav Pharmacol 21:153–160
Liu X, Jernigen C, Gharib M, Booth S, Caggiula AR, Sved AF (2010) Effects of dopamine antagonists on drug cue-induced reinstatement of nicotine-seeking behavior in rats. Behav Pharmacol 21:153–160
Seeman P, Van Tol HH (1994) Dopamine receptor pharmacology. Trends Pharmacol Sci 15:264–270
Wise RA (2004) Dopamine, learning and motivation. Nat Rev Neurosci 5:483–494
Fuster JM (2008) The prefrontal cortex. Academic, New York, pp 333–385
Kolb B, Cioe J (2004) Organization and plasticity of the prefrontal cortex of the rat Prefrontal Cortex. In Otani S (ed) From synaptic plasticity to cognition. Kluwer, Dordrecht, pp 1–32
Chiu CQ, Puente N, Grandes P, Castillo PE (2010) Dopaminergic modulation of endocannabinoid-mediated plasticity at GABAergic synapses in the prefrontal cortex. J Neurosci 30:7236–7248
Xu TX, Yao WD (2010) D1 and D2 dopamine receptors in separate circuits cooperate to drive associative long-term potentiation in the prefrontal cortex. Proc Natl Acad Sci USA 107:16366–16371
Acknowledgements
This work was supported in part by the National Nature Science Foundation of China (81771431) and Science & Technology Foundation of Wenzhou (2017Y0643).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, S., Wang, X., Lin, W. et al. Dopamine D1 Receptor Within Basolateral Amygdala Is Involved in Propofol Relapse Behavior Induced by Cues. Neurochem Res 43, 2393–2403 (2018). https://doi.org/10.1007/s11064-018-2667-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-018-2667-8