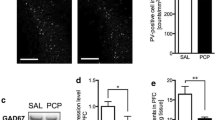
Abstract
It has been reported that specific environmental influences during the postpartum period might contribute to the development of schizophrenia (SZ). Administration of MK801 during early development led to persistent brain pathology. Glutamate decarboxylase 1 (GAD67) and parvalbumin (PV), and neuregulin 1 (NRG1)/ErbB4 signaling were closely associated with SZ pathology. We postulated therefore that NMDA receptor antagonists exposure during the postpartum period may be associated with expression dysregulation of some of the SZ candidate proteins. To test this, we used mouse primary hippocampal neurons and neonatal male mice treated with the NMDA receptor antagonist, MK801 at postnatal day 4 (P4) or P7, followed by the treatments of antipsychotic drugs (i.e., olanzapine, risperidone, and haloperidol). The expressions of GAD67, PV, NRG1, and ErbB4 in in vitro and in vivo SZ models were detected with Western blot analysis and immunohistochemistry, respectively. Behavioral tests (locomotion activity, social interaction, novel object recognition and prepulse inhibition) were measured. We found MK801 decreased the expression of GAD67, PV, NRG1 and ErbB4, and induced obvious behavioral alterations, while antipsychotics reversed these alterations. These results suggest that exposure to the NMDA receptor antagonist in early development may lead to long-lasting influence on the expression of specific proteins, such as GAD67, PV, NRG1, and ErbB4. Moreover, our results suggest that rescue of the activation of the NRG1/ErbB4 signaling pathway may be one of the mechanisms by which antipsychotic drugs have an antipsychotic effect.












Similar content being viewed by others
Abbreviations
- SZ:
-
Schizophrenia
- CNS:
-
The central nervous system
- GAD67:
-
Glutamate decarboxylase 1 (GAD67)
- PV:
-
Parvalbumin
- NRG1:
-
Neuregulin 1
- NMDA:
-
N-methyl-d-aspartate
- PPI:
-
Prepulse inhibition
- NORT:
-
Novel object recognition test
References
Murray CJ, Lopez AD (1997) Alternative projections of mortality and disability by cause 1990–2020: Global Burden of Disease Study. Lancet 349(9064):1498–1504. doi:10.1016/s0140-6736(96)07492-2 (London, England)
Owen MJ, O’Donovan MC, Thapar A, Craddock N (2011) Neurodevelopmental hypothesis of schizophrenia. Br J Psychiatry 198(3):173–175. doi:10.1192/bjp.bp.110.084384
van Os J, Kapur S (2009) Schizophrenia. Lancet 374(9690):635–645. doi:10.1016/S0140-6736(09)60995-8
Orlovska S, Pedersen MS, Benros ME, Mortensen PB, Agerbo E, Nordentoft M (2014) Head injury as risk factor for psychiatric disorders: a nationwide register-based follow-up study of 113,906 persons with head injury. Am J Psychiatry 171(4):463–469. doi:10.1176/appi.ajp.2013.13020190
Berger GE, Wood S, McGorry PD (2003) Incipient neurovulnerability and neuroprotection in early psychosis. Psychopharmacol Bull 37(2):79–101
McBain CJ, Fisahn A (2001) Interneurons unbound. Nat Rev Neurosci 2(1):11–23. doi:10.1038/35049047
Hoftman GD, Volk DW, Bazmi HH, Li S, Sampson AR, Lewis DA (2015) Altered cortical expression of GABA-related genes in schizophrenia: illness progression vs developmental disturbance. Schizophr Bull 41(1):180–191. doi:10.1093/schbul/sbt178
Arnold SE, Talbot K, Hahn CG (2005) Neurodevelopment, neuroplasticity, and new genes for schizophrenia. Prog Brain Res 147:319–345. doi:10.1016/s0079-6123(04)47023-x
Glausier JR, Fish KN, Lewis DA (2014) Altered parvalbumin basket cell inputs in the dorsolateral prefrontal cortex of schizophrenia subjects. Mol Psychiatry 19(1):30–36. doi:10.1038/mp.2013.152
Gos T, Myint AM, Schiltz K, Meyer-Lotz G, Dobrowolny H, Busse S, Muller UJ, Mawrin C, Bernstein HG, Bogerts B, Steiner J (2014) Reduced microglial immunoreactivity for endogenous NMDA receptor agonist quinolinic acid in the hippocampus of schizophrenia patients. Brain Behav Immun 41:59–64. doi:10.1016/j.bbi.2014.05.012
Ozdemir H, Ertugrul A, Basar K, Saka E (2012) Differential effects of antipsychotics on hippocampal presynaptic protein expressions and recognition memory in a schizophrenia model in mice. Prog Neuropsychopharmacol Biol Psychiatry 39(1):62–68. doi:10.1016/j.pnpbp.2012.05.009
Kim TW, Kang HS, Park JK, Lee SJ, Baek SB, Kim CJ (2014) Voluntary wheel running ameliorates symptoms of MK-801-induced schizophrenia in mice. Mol Med Rep 10(6):2924–2930. doi:10.3892/mmr.2014.2644
Coleman LG Jr, Jarskog LF, Moy SS, Crews FT (2009) Deficits in adult prefrontal cortex neurons and behavior following early post-natal NMDA antagonist treatment. Pharmacol Biochem Behav 93(3):322–330. doi:10.1016/j.pbb.2009.04.017
Jones KS, Corbin JG, Huntsman MM (2014) Neonatal NMDA receptor blockade disrupts spike timing and glutamatergic synapses in fast spiking interneurons in a NMDA receptor hypofunction model of schizophrenia. PLoS One 9(10):e109303. doi:10.1371/journal.pone.0109303
Williams NM, Preece A, Spurlock G, Norton N, Williams HJ, Zammit S, O’Donovan MC, Owen MJ (2003) Support for genetic variation in neuregulin 1 and susceptibility to schizophrenia. Mol Psychiatry 8(5):485–487. doi:10.1038/sj.mp.40013485
Correll CU (2010) From receptor pharmacology to improved outcomes: individualising the selection, dosing, and switching of antipsychotics. Eur Psychiatry 25(Suppl 2):S12–S21. doi:10.1016/s0924-9338(10)71701-6
Stefansson H, Steinthorsdottir V, Thorgeirsson TE, Gulcher JR, Stefansson K (2004) Neuregulin 1 and schizophrenia. Ann Med 36(1):62–71
Fischbach GD, Rosen KM (1997) ARIA: a neuromuscular junction neuregulin. Annu Rev Neurosci 20:429–458. doi:10.1146/annurev.neuro.20.1.429
Lemke G (2001) Glial control of neuronal development. Annu Rev Neurosci 24:87–105. doi:10.1146/annurev.neuro.24.1.87
Harrison PJ, Weinberger DR (2005) Schizophrenia genes, gene expression, and neuropathology: on the matter of their convergence. Mol Psychiatry 10(1):40–68. doi:10.1038/sj.mp.4001558 (image 45)
Wakuda T, Iwata K, Iwata Y, Anitha A, Takahashi T, Yamada K, Vasu MM, Matsuzaki H, Suzuki K, Mori N (2015) Perinatal asphyxia alters neuregulin-1 and COMT gene expression in the medial prefrontal cortex in rats. Prog Neuropsychopharmacol Biol Psychiatry 56:149–154. doi:10.1016/j.pnpbp.2014.08.002
Dejaegere T, Serneels L, Schafer MK, Van Biervliet J, Horre K, Depboylu C, Alvarez-Fischer D, Herreman A, Willem M, Haass C, Hoglinger GU, D’Hooge R, De Strooper B (2008) Deficiency of Aph1B/C-γ-secretase disturbs Nrg1 cleavage and sensorimotor gating that can be reversed with antipsychotic treatment. Proc Natl Acad Sci USA 105(28):9775–9780. doi:10.1073/pnas.0800507105
Savonenko AV, Melnikova T, Laird FM, Stewart KA, Price DL, Wong PC (2008) Alteration of BACE1-dependent NRG1/ErbB4 signaling and schizophrenia-like phenotypes in BACE1-null mice. Proc Natl Acad Sci USA 105(14):5585–5590. doi:10.1073/pnas.0710373105
Barros CS, Calabrese B, Chamero P, Roberts AJ, Korzus E, Lloyd K, Stowers L, Mayford M, Halpain S, Muller U (2009) Impaired maturation of dendritic spines without disorganization of cortical cell layers in mice lacking NRG1/ErbB signaling in the central nervous system. Proc Natl Acad Sci USA 106(11):4507–4512. doi:10.1073/pnas.0900355106
Engl J, Laimer M, Niederwanger A, Kranebitter M, Starzinger M, Pedrini MT, Fleischhacker WW, Patsch JR, Ebenbichler CF (2005) Olanzapine impairs glycogen synthesis and insulin signaling in L6 skeletal muscle cells. Mol Psychiatry 10(12):1089–1096
Türkez H, Toğar B (2010) The genotoxic and oxidative damage potential of olanzapine in vitro. Toxicol Ind Health 26(9):583–588. doi:10.1177/0748233710373090 (Epub 2010 Jun 11)
Schmidt AJ, Krieg JC, Clement HW, Hemmeter UM, Schulz E, Vedder H, Heiser P (2010) Effects of quetiapine, risperidone, 9-hydroxyrisperidone and ziprasidone on the survival of human neuronal andimmune cells in vitro. J Psychopharmacol 24(3):349–354. doi:10.1177/0269881108096506
Niu J, Mei F, Li N, Wang H, Li X, Kong J, Xiao L (2010) Haloperidol promotes proliferation but inhibits differentiation in rat oligodendrocyte progenitor cell cultures. Biochem Cell Biol 88(4):611–620. doi:10.1139/O09-178
Mutlu O, Ulak G, Celikyurt IK, Akar FY, Erden F (2011) Effects of olanzapine, sertindole and clozapine on learning and memory in the Morris water maze test in naive and MK-801-treated mice. Pharmacol Biochem Behav 98(3):398–404. doi:10.1016/j.pbb.2011.02.009
Sams-Dodd F (1995) Automation of the social interaction test by a video-tracking system: behavioural effects of repeated phencyclidine treatment. J Neurosci Methods 59(2):157–167
Schneider T, Przewlocki R (2005) Behavioral alterations in rats prenatally exposed to valproic acid: animal model of autism. Neuropsychopharmacology 30(1):80–89. doi:10.1038/sj.npp.1300518
Mizoguchi H, Takuma K, Fukakusa A, Ito Y, Nakatani A, Ibi D, Kim HC, Yamada K (2008) Improvement by minocycline of methamphetamine-induced impairment of recognition memory in mice. Psychopharmacology 196(2):233–241. doi:10.1007/s00213-007-0955-0
Gunduz-Bruce H (2009) The acute effects of NMDA antagonism: from the rodent to the human brain. Brain Res Rev 60(2):279–286. doi:10.1016/j.brainresrev.2008.07.006
du Bois TM, Huang XF (2007) Early brain development disruption from NMDA receptor hypofunction: relevance to schizophrenia. Brain Res Rev 53(2):260–270. doi:10.1016/j.brainresrev.2006.09.001
Markham JA, Koenig JI (2011) Prenatal stress: role in psychotic and depressive diseases. Psychopharmacology 214(1):89–106. doi:10.1007/s00213-010-2035-0
Light GA, Hsu JL, Hsieh MH, Meyer-Gomes K, Sprock J, Swerdlow NR, Braff DL (2006) Gamma band oscillations reveal neural network cortical coherence dysfunction in schizophrenia patients. Biol Psychiatry 60(11):1231–1240. doi:10.1016/j.biopsych.2006.03.055
Zhang ZJ, Reynolds GP (2002) A selective decrease in the relative density of parvalbumin-immunoreactive neurons in the hippocampus in schizophrenia. Schizophr Res 55(1–2):1–10
Rudolph U, Mohler H (2014) GABAA receptor subtypes: therapeutic potential in down syndrome, affective disorders, schizophrenia, and autism. Annu Rev Pharmacol Toxicol 54:483–507. doi:10.1146/annurev-pharmtox-011613-135947
Stefansson H, Sigurdsson E, Steinthorsdottir V, Bjornsdottir S, Sigmundsson T, Ghosh S, Brynjolfsson J, Gunnarsdottir S, Ivarsson O, Chou TT, Hjaltason O, Birgisdottir B, Jonsson H, Gudnadottir VG, Gudmundsdottir E, Bjornsson A, Ingvarsson B, Ingason A, Sigfusson S, Hardardottir H, Harvey RP, Lai D, Zhou M, Brunner D, Mutel V, Gonzalo A, Lemke G, Sainz J, Johannesson G, Andresson T, Gudbjartsson D, Manolescu A, Frigge ML, Gurney ME, Kong A, Gulcher JR, Petursson H, Stefansson K (2002) Neuregulin 1 and susceptibility to schizophrenia. Am J Hum Genet 71(4):877–892. doi:10.1086/342734
Fazzari P, Paternain AV, Valiente M, Pla R, Lujan R, Lloyd K, Lerma J, Marin O, Rico B (2010) Control of cortical GABA circuitry development by Nrg1 and ErbB4 signalling. Nature 464(7293):1376–1380. doi:10.1038/nature08928
Yamada A, Inoue E, Deguchi-Tawarada M, Matsui C, Togawa A, Nakatani T, Ono Y, Takai Y (2013) Necl-2/CADM1 interacts with ErbB4 and regulates its activity in GABAergic neurons. Mol Cell Neurosci 56:234–243. doi:10.1016/j.mcn.2013.06.003
Wen L, Lu YS, Zhu XH, Li XM, Woo RS, Chen YJ, Yin DM, Lai C, Terry AV Jr, Vazdarjanova A, Xiong WC, Mei L (2010) Neuregulin 1 regulates pyramidal neuron activity via ErbB4 in parvalbumin-positive interneurons. Proc Natl Acad Sci USA 107(3):1211–1216. doi:10.1073/pnas.0910302107
Yin DM, Sun XD, Bean JC, Lin TW, Sathyamurthy A, Xiong WC, Gao TM, Chen YJ, Mei L (2013) Regulation of spine formation by ErbB4 in PV-positive interneurons. J Neurosci 33(49):19295–19303. doi:10.1523/jneurosci.2090-13.2013
Lang UE, Puls I, Muller DJ, Strutz-Seebohm N, Gallinat J (2007) Molecular mechanisms of schizophrenia. Cell Physiol Biochem 20(6):687–702. doi:10.1159/000110430
Grunze HC, Rainnie DG, Hasselmo ME, Barkai E, Hearn EF, McCarley RW, Greene RW (1996) NMDA-dependent modulation of CA1 local circuit inhibition. J Neurosci 16(6):2034–2043
Law AJ, Lipska BK, Weickert CS, Hyde TM, Straub RE, Hashimoto R, Harrison PJ, Kleinman JE, Weinberger DR (2006) Neuregulin 1 transcripts are differentially expressed in schizophrenia and regulated by 5′ SNPs associated with the disease. Proc Natl Acad Sci USA 103(17):6747–6752. doi:10.1073/pnas.0602002103
Chong VZ, Thompson M, Beltaifa S, Webster MJ, Law AJ, Weickert CS (2008) Elevated neuregulin-1 and ErbB4 protein in the prefrontal cortex of schizophrenic patients. Schizophr Res 100(1–3):270–280. doi:10.1016/j.schres.2007.12.474
Banerjee A, Macdonald ML, Borgmann-Winter KE, Hahn CG (2010) Neuregulin 1-erbB4 pathway in schizophrenia: from genes to an interactome. Brain Res Bull 83(3–4):132–139
Wang XD, Su YA, Guo CM, Yang Y, Si TM (2008) Chronic antipsychotic drug administration alters the expression of neuregulin 1beta, ErbB2, ErbB3, and ErbB4 in the rat prefrontal cortex and hippocampus. Int J Neuropsychopharmacol 11(4):553–561
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81171268).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Li, C., Tang, Y., Yang, J. et al. Sub-chronic Antipsychotic Drug Administration Reverses the Expression of Neuregulin 1 and ErbB4 in a Cultured MK801-Induced Mouse Primary Hippocampal Neuron or a Neurodevelopmental Schizophrenia Model. Neurochem Res 41, 2049–2064 (2016). https://doi.org/10.1007/s11064-016-1917-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1917-x