Abstract
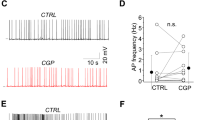
Medium spiny projection neurons (MSNs) are the main neuronal population in the neostriatum. MSNs are inhibitory and GABAergic. MSNs connect with other MSNs via local axon collaterals that produce lateral inhibition, which is thought to select cell assemblies for motor action. MSNs also receive inhibitory inputs from GABAergic local interneurons. This work shows, through the use of the paired pulse protocol, that somatostatin (SST) acts presynaptically to regulate GABA release from the terminals interconnecting MSNs. This SST action is reversible and not mediated through the release of dopamine. It is blocked by the SST receptor (SSTR) antagonist ciclosomatostatin (cicloSST). In contrast, SST does not regulate inhibition coming from interneurons. Because, SST is released by a class of local interneuron, it is concluded that this neuron helps to regulate the selection of motor acts.





Similar content being viewed by others
References
Wilson CJ (2004) Basal ganglia. In: The synaptic organization of the brain. Oxford University Press, Oxford, pp 361–414
Graybiel AM (1995) Building action repertoires: memory and learning functions of the basal ganglia. Curr Opin Neurobiol 5:733–741
Grillner S, Hellgren J, Menard A et al (2005) Mechanisms for selection of basic motor programs—roles for the striatum and pallidum. Trends Neurosci 28:364–370
Barnes TD, Kubota Y, Hu D et al (2005) Activity of striatal neurons reflects dynamic encoding and recoding of procedural memories. Nature 437:1158–1161
Czubayko U, Plenz D (2002). Fast synaptic transmission between striatal spiny projection neurons. Proc Natl Acad Sci 99:15764–15769
Tunstall MJ, Oorschot DE, Kean A et al (2002) Inhibitory interactions between spiny projection neurons in the rat striatum. J Neurophysiol 88:1263–1269
Koos T, Tepper JM, Wilson CJ (2004) Comparison of IPSCs evoked by spiny and fast-spiking neurons in the neostriatum. J Neurosci 24:7916–7922
Bar-Gad I, Bergman H (2001) Stepping out of the box: information processing in the neural networks of the basal ganglia. Curr Opin Neurobiol 11:689–695
Koós T, Tepper JM (1999) Inhibitory control of neostriatal projection neurons by GABAergic interneurons. Nat Neurosci 2:467–472
Di Figlia M, Aronin N (1982) Ultrastructural features of immunoreactivity somatostatin neurons in the rat caudate nucleus. J Neurosci 2:1267–1274
Chesselet MF, Graybiel AM (1982) Striatal neurons expressing somatostatin-like immunoreactivity: evidence for a peptidergic interneuronal system in the cat. Neuroscience 17:107–126
Ramirez JL, Mouchantaf R, Kumar U et al (2002) Brain somatostatin receptors are upregulated in somatostatin deficient mice. Mol Endocrinol 16:1951–1963
Low MJ (2004) Clinical endocrinology and metabolism. The somatostatin neuroendocrine system: physiology and clinical relevance in gastrointestinal and pancreatic disorders. Best Pract Res Clin Endocrinol Metab 18:607–622
Vilchis C, Bargas J, Pérez-Roselló T et al (2002) Somatostatin modulates Ca2+ currents in neostriatal neurons. Neuroscience 109:555–567
Galarraga E, Vilchis C, Tkatch T et al (2007) Somatostatinergic modulation of firing pattern and calcium activated potassium currents in medium spiny neostriatal neurons. Neuroscience 146:537–554
Izhikevich EM (2007) Dynamical systems in Neuroscience. MIT Press, Cambridge
Boehm S, Betz H (1997) Somatostatin inhibits excitatory transmission at rat hippocampal synapses via presynaptic receptors. J Neurosci 17:4066–4075
Tallent MK, Siggins GR (1997) Somatostatin depresses excitatory but not inhibitory neurotransmission in rat CA1 hippocampus. J Neurophysiol 78:3008–3018
Leresche N, Asprodini E, Emri Z, et al (2000) Somatostatin inhibits GABAergic transmission in the sensory thalamus via presynaptic receptors. Neuroscience 98:513–522
Lanneau C, Peineau S, Petit F et al (2000) Somatostatin modulation of excitatory synaptic transmission between periventricular and arcuate hypothalamic nuclei in vitro. J Neurophysiol 84:1464–1474
Momiyama T, Zaborszky L (2006) Somatostatin presynaptically inhibits both GABA and glutamate release onto rat basal forebrain cholinergic neurons. J Neurophysiol 96:686–694
Sun Q-Q, Huguenard JR, Prince DA (2002) Somatostatin inhibits thalamic network oscillations in vitro: actions on the GABAergic neurons of reticular nucleus. J Neurosci 22:5374–5386
Guzman JN, Hernandez A, Galarraga E et al (2003) Dopaminergic modulation of axon collaterals interconnecting spiny neurons of the rat striatum. J Neurosci 23:8931–8940
Salgado H, Tecuapetla F, Perez-Rosello T et al (2005) A reconfiguration of CaV2 Ca2+ channels current and its dopaminergic D2 modulation in developing neostriatal neurons. J Neurophysiol 94:3771–3787
Pérez-Rosello T, Figueroa A, Salgado H et al (2005) The cholinergic control of firing pattern and neurotransmission in rat neostriatal projection neurons: role of CaV2.1 and CaV2.2 Ca2+ channels. J Neurophysiol 93:2507–2519
Tecuapetla F, Carrillo-Reid L, Bargas J et al (2007) Dopaminergic modulation of short term synaptic plasticity at striatal inhibitory synapses. Proc Natl Acad Sci USA 104:10258–10263
Bargas J, Galarraga E, Aceves J (1989) An early outward conductance modulates the firing latency and frequency of neostriatal neurons of the rat brain. Exp Brain Res 75:146–156
Tecuapetla F, Carrillo-Reid L, Guzmán JN et al (2005) Different inhibitory inputs onto neostriatal projection neurons as revealed by field stimulation. J Neurophysiol 93:1119–1126
Kita H (1993) GABAergic circuits of the striatum. Prog Brain Res 99:51–72
Cheselet MF, Reisine TD (1983) Somatostatin regulates dopaminérgico release in striatal slices and caudate nuclei. J Neurosci 3:232–236
Beal MF, Martin JB (1984) The effect of somatostatin on striatal catecholamines. Neurosci Lett 24:271–276
Thermos K, Radke J, Kastellakis A et al (1996) Dopamine–somatostatin interactions in the rat striatum: an in vivo microdialysis study. Synapse 22:209–216
Hathway GJ, Humphrey PPA, Kendrick KM (1999) Evidence that somatostatin SST2 receptors mediate striatal dopamine release. Br J Pharmacol 128:1346–1352
Meyer DK, Conzelmann U, Shultheiss K (1989) Effects of somatostatin-14 on the in vitro release of [3H]GABA from slices of rat caudatoputamen. Neuroscience 28:61–68
Vasilaki A, Papasava D, Hoyer D et al (2004) The somatostatin receptor (SST1) modulates the release of somatostatin in the nucleus accumbens of the rat. Neuropharmacology 47:612–618
Acknowledgment
We thank D. Tapia and J. A. Laville for technical assistance. L. Carrillo made the acquisition program. This work was supported by CONACyT grants: 42636 to EG and 49484 to JB, DGAPA-UNAM (IN201507, IN201607) and IMPULSA03 grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue article in honor of Dr. Ricardo Tapia.
Rights and permissions
About this article
Cite this article
Lopez-Huerta, V.G., Tecuapetla, F., Guzman, J.N. et al. Presynaptic Modulation by Somatostatin in the Neostriatum. Neurochem Res 33, 1452–1458 (2008). https://doi.org/10.1007/s11064-007-9579-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9579-3