Abstract
Objective
To investigate the association between the magnetic resonance imaging (MRI) signal characteristics of skull base chordoma and radiosurgical outcomes.
Methods
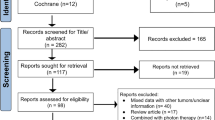
Twenty-four patients with skull base chordomas treated with Gamma Knife radiosurgery (GKRS) after previous surgical resection were retrospectively (2001–2021) examined. Pre-GKRS MRIs were analyzed for RT2 (tumor-to-brainstem signal intensity ratio on T2-weighted imaging), RCE (tumor-to-brainstem signal intensity ratio on contrast-enhanced T1-weighted imaging), and mean apparent diffusion coefficient (ADC). Correlations of the parameters with patient survival and local tumor progression were made by using Cox regression and Kaplan–Meier analyses.
Results
During a median follow-up of 46 months after GKRS, 9 patients died with significantly more local tumor progression events (median number: 2 vs 0, P = .012) than did 15 alive patients. On multivariable analysis, higher mean ADC was associated with longer patient survival (P = .016) after GKRS. The actuarial 5-year overall survival rates were 88.9% versus 54.7% for chordomas with an ADC of ≥ 1270 × 10–6 mm2/s versus < 1270 × 10–6 mm2/s. RT2 < 1.5 (P = .038) and RCE > 1.57 (P = .022) were associated with a lower probability of local tumor control.
Conclusion
Lower mean ADC values are associated with shorter patient survival in skull base chordomas after GKRS. Diffusion-weighted imaging may help in GKRS planning and outcome prediction for these patients.




Similar content being viewed by others
Data availability
The datasets generated during the current study are not publicly available due to information that could compromise the privacy of research participants, but are available from the corresponding author on reasonable request.
References
Erdem E, Angtuaco EC, Van Hemert R, Park JS, Al-Mefty O (2003) Comprehensive review of intracranial chordoma. Radiographics 23:995–1009. https://doi.org/10.1148/rg.234025176
Walcott BP, Nahed BV, Mohyeldin A, Coumans JV, Kahle KT, Ferreira MJ (2012) Chordoma: current concepts, management, and future directions. Lancet Oncol 13:e69-76. https://doi.org/10.1016/s1470-2045(11)70337-0
Wang L, Wu Z, Tian K, Wang K, Li D, Ma J, Jia G, Zhang L, Zhang J (2017) Clinical features and surgical outcomes of patients with skull base chordoma: a retrospective analysis of 238 patients. J Neurosurg 127:1257–1267. https://doi.org/10.3171/2016.9.jns16559
Catton C, O’Sullivan B, Bell R, Laperriere N, Cummings B, Fornasier V, Wunder J (1996) Chordoma: long-term follow-up after radical photon irradiation. Radiother Oncol 41:67–72. https://doi.org/10.1016/s0167-8140(96)91805-8
Takagi M, Demizu Y, Nagano F, Terashima K, Fujii O, Jin D, Mima M, Niwa Y, Katsui K, Suga M, Yamashita T, Akagi T, Sakata KI, Fuwa N, Okimoto T (2018) Treatment outcomes of proton or carbon ion therapy for skull base chordoma: a retrospective study. Radiat Oncol 13:232. https://doi.org/10.1186/s13014-018-1173-0
Pikis S, Mantziaris G, Peker S, Samanci Y, Nabeel AM, Reda WA, Tawadros SR, El-Shehaby AMN, Abdelkarim K, Eldin RME, Sheehan D, Sheehan K, Liscak R, Chytka T, Tripathi M, Madan R, Speckter H, Hernández W, Barnett GH, Hori YS, Dabhi N, Aldakhil S, Mathieu D, Kondziolka D, Bernstein K, Wei Z, Niranjan A, Kersh CR, Lunsford LD, Sheehan JP (2022) Stereotactic radiosurgery for intracranial chordomas: an international multiinstitutional study. J Neurosurg. https://doi.org/10.3171/2021.12.jns212416
Santegoeds RGC, Temel Y, Beckervordersandforth JC, Van Overbeeke JJ, Hoeberigs CM (2018) State-of-the-art imaging in human chordoma of the skull base. Curr Radiol Rep 6:16. https://doi.org/10.1007/s40134-018-0275-7
Guler E, Ozgen B, Mut M, Soylemezoglu F, Oguz KK (2017) The added value of diffusion magnetic resonance imaging in the diagnosis and posttreatment evaluation of skull base chordomas. J Neurol Surg B Skull Base 78:256–265. https://doi.org/10.1055/s-0036-1597824
Yeom KW, Lober RM, Mobley BC, Harsh G, Vogel H, Allagio R, Pearson M, Edwards MS, Fischbein NJ (2013) Diffusion-weighted MRI: distinction of skull base chordoma from chondrosarcoma. AJNR Am J Neuroradiol 34(1056–1061):s1051. https://doi.org/10.3174/ajnr.A3333
Tian K, Wang L, Ma J, Wang K, Li D, Du J, Jia G, Wu Z, Zhang J (2017) MR imaging grading system for skull base chordoma. AJNR Am J Neuroradiol 38:1206–1211. https://doi.org/10.3174/ajnr.A5152
Snell JW, Sheehan J, Stroila M, Steiner L (2006) Assessment of imaging studies used with radiosurgery: a volumetric algorithm and an estimation of its error. Technical note J Neurosurg 104:157–162. https://doi.org/10.3171/jns.2006.104.1.157
Lee CC, Chou CL, Chen CJ, Yang HC, Wu HM, Shiau CY, Pan DH, Chung WY (2018) Stereotactic radiosurgery for hypervascular intracranial tumors. J Neurooncol 140:547–558. https://doi.org/10.1007/s11060-018-2980-8
Kano H, Iqbal FO, Sheehan J, Mathieu D, Seymour ZA, Niranjan A, Flickinger JC, Kondziolka D, Pollock BE, Rosseau G, Sneed PK, McDermott MW, Lunsford LD (2011) Stereotactic radiosurgery for chordoma: a report from the North American Gamma Knife Consortium. Neurosurgery 68:379–389. https://doi.org/10.1227/NEU.0b013e3181ffa12c
US Department of Health and Human Services (2017) Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5x11.pdf. Accessed 1 May 2022
Raza SM, Bell D, Freeman JL, Grosshans DR, Fuller GN, DeMonte F (2018) multimodality management of recurrent skull base chordomas: factors impacting tumor control and disease-specific survival. Oper Neurosurg (Hagerstown) 15:131–143. https://doi.org/10.1093/ons/opx201
Zhou J, Yang B, Wang X, Jing Z (2018) Comparison of the effectiveness of radiotherapy with photons and particles for chordoma after surgery: a meta-analysis. World Neurosurg 117:46–53. https://doi.org/10.1016/j.wneu.2018.05.209
Malayeri AA, El Khouli RH, Zaheer A, Jacobs MA, Corona-Villalobos CP, Kamel IR, Macura KJ (2011) Principles and applications of diffusion-weighted imaging in cancer detection, staging, and treatment follow-up. Radiographics 31:1773–1791. https://doi.org/10.1148/rg.316115515
Ginat DT, Mangla R, Yeaney G, Johnson M, Ekholm S (2012) Diffusion-weighted imaging for differentiating benign from malignant skull lesions and correlation with cell density. AJR Am J Roentgenol 198:W597-601. https://doi.org/10.2214/ajr.11.7424
Oh HC, Hong CK, Lee KS, Cha YJ, Ahn SJ, Suh SH, Park HH (2021) Apparent diffusion coefficient as a prognostic factor in clival chordoma. Sci Rep 11:486. https://doi.org/10.1038/s41598-020-79894-8
Sasaki T, Moritani T, Belay A, Capizzano AA, Sato SP, Sato Y, Kirby P, Ishitoya S, Oya A, Toda M, Takahashi K (2018) Role of the apparent diffusion coefficient as a predictor of tumor progression in patients with chordoma. AJNR Am J Neuroradiol 39:1316–1321. https://doi.org/10.3174/ajnr.A5664
Wang K, Xie SN, Wang L, Du J, Ma JP, Huo XL, Tian KB, Zhang LW, Zhang JT, Wu Z (2020) Natural growth dynamics of untreated skull base chordomas in vivo. World Neurosurg 136:e310–e321. https://doi.org/10.1016/j.wneu.2019.12.164
Tian K, Wang L, Wang K, Ma J, Li D, Hao S, Yang Y, Du J, Jia G, Zhang L, Wu Z, Zhang J (2016) Analysis of clinical features and outcomes of skull base chordoma in different age-groups. World Neurosurg 92:407–417. https://doi.org/10.1016/j.wneu.2016.05.035
Holtzman AL, Rotondo RL, Rutenberg MS, Indelicato DJ, De Leo A, Rao D, Patel J, Morris CG, Mendenhall WM (2021) Clinical outcomes following dose-escalated proton therapy for skull-base chordoma. Int J Part Ther 8:179–188. https://doi.org/10.14338/ijpt-20-00066.1
Shinya Y, Hasegawa H, Shin M, Kawashima M, Koga T, Hanakita S, Katano A, Sugiyama T, Nozawa Y, Saito N (2022) High dose radiosurgery targeting the primary tumor sites contributes to survival in patients with skull base chordoma. Int J Radiat Oncol Biol Phys 113:582–587. https://doi.org/10.1016/j.ijrobp.2022.02.024
Acknowledgements
The authors would like to thank Hsin-Yi Huang (Biostatistics Task Force, Taipei Veterans General Hospital) for providing statistical analysis assistance.
Funding
This work was supported by Taipei Veterans General Hospital (Grant Number: V110C-056) and Taiwan’s Ministry of Science and Technology (Grant Number: MOST-109-2628-B-010-014-MY2).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Y-SH, C-CL and C-AW. The first draft of the manuscript was written by Y-SH and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Taipei Veterans General Hospital (2021–07-025CC).
Consent to participate
The Taipei Veterans General Hospital institutional review board approved the study protocol and waived the need for consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, YS., Lee, CC., Wu, CA. et al. Magnetic resonance imaging signal characteristics associated with prognosis of skull base chordoma after gamma knife radiosurgery. J Neurooncol 161, 45–56 (2023). https://doi.org/10.1007/s11060-022-04199-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04199-x