Abstract
Purpose
The health-related quality of life (HRQoL) for patients with glioblastoma is known to be largely affected. Little is known about the HRQoL for relatives and the relationship between these two. To optimize family care, such issues need to be addressed early on, preferably from the time of diagnosis. This study aimed to describe and compare the HRQoL of patients with glioblastoma and their relatives before surgery.
Methods
A prospective cohort study including 89 patients diagnosed with glioblastoma and their relatives. HRQoL (Short Form Health Survey, SF-36) and emotional well-being (hospital anxiety and depression scale, HADS) were analysed with descriptive, comparative and multivariable regression analyses.
Results
Relatives scored worse for mental HRQoL (p < 0.001) and for symptoms of anxiety (p < 0.001) and depression (p = 0.022) compared to patients. The multivariable regression showed an increased risk of affected mental HRQoL in relatives of patients with poor functional status (WHO) (p = 0.01) and higher levels in symptoms of anxiety (p = 0.03), or when relatives had low physical HRQoL themselves (p = 0.01). There was increased risk of affected mental HRQoL in patients with comorbidities (p = 0.003), and when the respective relative showed higher levels in symptoms of anxiety (p = 0.005).
Conclusion
Relatives scored worse for mental HRQoL and emotional well-being than patients, suggesting that HRQoL in patients and relatives might be connected to symptoms of anxiety in the respective individual at disease onset. The results illustrate the need to screen HRQoL and emotional well-being in both patients and relatives from an early stage—before surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glioblastoma is the most common and aggressive primary malignant brain tumor in adults with an incidence of approximately 3.2/100 000 [1]. The mainstay of treatment for glioblastoma is surgical resection, followed by chemo- and radiotherapy. Despite combined therapy, the median survival for glioblastoma is about 1–2 years [2, 3] and 5-year survival rate at around 5–9% [1, 4, 5].
Patients with glioblastoma present with a variety of symptoms and signs, such as neurological deficits and epileptic seizures, cognitive problems and neuropsychiatric symptoms of anxiety and depression, caused by the tumor itself, tumor-related treatment, or a combination of these factors [6]. These symptoms may all have a negative effect on the health-related quality of life (HRQoL) [7, 8], leading to inactivity [7] and the ability to continue a normal working life [7, 9]. The patient’s accelerating symptoms may also affect the nearest relative’s HRQoL [10]. In studies comparing gender, female generally score lower HRQoL then men [11, 12]. In addition, the individual situation of the patient will be affected by the strains placed upon the family members by the disease and the altered roles within the family—with the relatives themselves being affected by the increased burden imposed upon them [7, 9, 13, 14]. To our knowledge no studies have examined the relationship between the experiences of patients and relatives HRQoL and emotional well-being—before surgery.
To optimize our current de facto palliative oncological care, and develop and implement useful support to promote well-being, information about baseline HRQoL, emotional well-being, and family relationships is necessary. Identifying potential problems at early stage, and addressing these appropriately, is essential and could—in the long run—improve the HRQoL and emotional well-being in this group of patients and their relatives.
It is important to address and fill out this knowledge gap of the relation between patients and relatives before surgery. Relatives are likely to play an essential role in the HRQoL and emotional well-being of the patients with glioblastoma, as well as being affected themselves by the changes caused by the disease. The aim of this study was to describe and compare the HRQoL and emotional well-being in patients with glioblastoma and their relatives before surgery.
Material and methods
Participants
This was a prospective cohort study and patients with glioblastoma were identified from a population-based study at the University Hospital in Gothenburg, Sweden [15]. Patients over 18 years old with a radiological diagnosis of glioblastoma in the South-West region of Sweden with approximately 1.7 million inhabitants (one of the two lager regions in Sweden) were approached prior to surgery. Patients and their relatives were thoroughly informed about the suspected diagnosis. The following exclusion criteria were used: emergency surgery, severe cognitive impairment, poor performance of the Swedish language, operated elsewhere, reoperation. In addition, patients with other histological diagnosis than glioblastoma were excluded after surgery (Fig. 1). For recruitment of relatives, the patients were asked to select a person closest to them (this occurred 1–3 days before surgery), regardless of they were living together or not. The relative was then contacted by the study coordinator and asked to participate in the study.
Measures
Short Form Health Survey (SF-36) is a validated and reliable questionnaire when measuring HRQoL, both for general population [16, 17] and for patients with brain tumors [18], the questionnaire is generic and one of the most widely used measures of HRQoL in clinical studies. SF-36 includes 36 items, with 35 items divided into eight domains, which are subsequently divided into two major health components—physical component summary (PCS) and mental component summary (MCS) [16, 17]. The domains and dimensions range from 0 to 100 (worst possible health state to best possible health state) [17], with a population mean of 50 and a standard deviation (SD) of 10 [19].
The hospital anxiety and depression scale (HADS) is a validated [20] questionnaire. It consists of a self-assessment scale with 14 items divided into two subscales, including the person’s own experience of anxiety (HADa) and depression (HADd). Items range from 0 to 3 on a four-point Likert scale. The score for each subscale ranges from 0 to 21. Seven points or lower indicate the absence of significant anxiety or depression, scores between 8 and 10 indicate doubtful cases, and scores over 11 indicate definite cases of anxiety or depression [21].
One way to measure functional status is Eastern Cooperative Oncology Group Performance Status (ECOG PS), this questionnaire range from 0 to 5: 0; no limitations of activity, 1; restricted in physically exhausting activity, 2; ambulatory and capable of all self-care, but unable to carry out any work activities, 3; capable of only limited self-care, 4; completely disabled, 5; dead. This was later adapted by the WHO and has the title ECOG/WHO performance status [22, 23].
Data collection
Data collection occurred from October 2012 to November 2016. Prior to admission for surgery, the patients and relatives received information about the suspected tumor diagnosis. A research nurse distributed the questionnaires to the patients and relatives in connection with enrolment before surgery. The questionnaires were completed by the individual participant without input from others. Patients and relatives scored their own HRQoL and emotional well-being.
The following data were collected from the medical records: gender, age at surgery, days from diagnosis to surgery, first symptom (neurological deficit, epilepsy, cognitive effects, headache, several symptoms, incidental findings), effects of corticosteroids (e.g. relieved headache or improved neurological function), functional status (according to the ECOG/WHO performance status scale), location of the tumor (unilateral, bilateral, central), tumor side (right, left, bilateral), comorbidity (according to an adapted Charlson Comorbidity Index (CCI) [24]) hypertension, diabetes and epilepsy and surgical planning (radical resection, subtotal resection, biopsy).
Statistics
To present data descriptive statistics were used. Wilcoxon signed-rank test was used for the analysis of paired comparisons between patients and relatives. Mann–Whitney U test was used to analyse differences between groups. Univariable linear regression analysis applying forward selection was performed to test the impact of various variables on SF-36 MCS in patients as well as in relatives. These variables were: age of patient at surgery; days from diagnosis to surgery; first symptom (neurological deficit, epilepsy, cognitive effects); functional status (WHO); location of the tumor; operation side; comorbidity; surgical planning; and the patient’s and relative’s own estimates of SF-36 and HADS. Variables that predicted SF-36 MCS with p < 0.20 in the univariable analyses were entered and tested as independent variables in the multivariable stepwise linear regression. All tests were 2-tailed with a significance level of 5%. Statistical analysis was performed using IBM SPSS Statistics 23.
Results
Demographic data
A total of 89 patients were included in the study (Fig. 1). Their median age was 64 years, 57 were male and 32 were female. These 89 patients were asked to choose a relative for participating in the study. Of the 63 relatives who participated in the study, 18 were males and 45 were females; 49 were spouses or cohabitants (15 males/34 females), 12 were offspring of adult age (3 males/9 females), one was parent, and one was daughter-in-law. An overview of clinical and demographic data is provided in Table 1. A flow chart with sample numbers, reasons for exclusions and non-participation is shown in Fig. 1. An overview of the estimated self-assessed SF-36 and HADS for the patient group (n = 89), for the patients whose relatives were included (n = 63), and the group of relatives (n = 63) is shown in Table 2. Hypertension was the most common comorbidity, followed by diabetes and epilepsy. There were no differences in terms of age, sex, and days from diagnosis to surgery, comorbidity, or preoperative intention between the patients who participated compared with the drop-outs. When WHO was merged into the groups 0–1 and 2–4 there was a tendency that the WHO was worse against the drop-outs (WHO 2–4 = 33.3% in the drop-outs vs. 15.9% in the participated patients).
Comparisons between patients and relatives
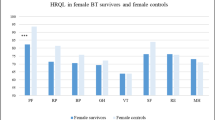
Paired comparisons between patients and relatives showed that patients scored worse levels for the physical parameters in SF-36; physical functioning (p < 0.01), role physical (p < 0.01), bodily pain (p = 0.047), general health (p = 0.007), and total PCS (p < 0.001), while relatives scored lower levels for the psychologic parameters in SF-36, i.e., mental health (p = 0.001) and total MCS (p < 0.001). In addition, relatives scored significantly worse symptoms for HADa (p < 0.001) and HADd (p = 0.022) (Table 2).
Compared to male relatives, female relatives scored worse levels in SF-36 vitality (p = 0.018), SF (p = 0.037), and in HADd (p = 0.016). In the case of patients, no differences were found between males and females.
The relationship between patients’ and relatives’ mental HRQoL and independent variables
As stated, all variables with p-values < 0.20 in the univariable analysis were entered in a step-wise multivariable regression model (Table 3). Of these, the presence of comorbidity (p = 0.003) in patients and HADa (p = 0.005) in relatives retained their significance in the multivariable model, i.e. identified as factors that affected patients’ mental health (Table 3).
Regarding the group of relatives, variables with p-values < 0.20 also were entered in a step-wise multivariable regression model (Table 4). Of these, the presence of patients’ functional status (WHO) (p = 0.01) and HADa (p = 0.03), as well as SF-36 PCS (p = 0.01) in relatives retained their significance in the multivariable model, i.e. those were factors that affected relatives mental health (Table 4).
Discussion
In this study the mental HRQoL of patients with glioblastoma and their relatives were found to be connected to symptoms of anxiety in the respective individual. In addition, there were differences between the patients and relatives in terms of self-reported HRQoL and emotional well-being. Relatives scored worse for items covering mental HRQoL and reported more frequent symptoms of anxiety and depression than patients.
It is well known that HRQoL and emotional well-being deteriorate in patients with glioblastoma who have had—or are about to undergo—tumor treatment [7, 25, 26]. Anxiety and emotional wellbeing may be more skewed towards patients with major neurological deficits such as significant aphasia or a dominant side hemiparesis. However, this has not been analysed in the study. In this study, relatives reported worse mental states than patients, including worse scores for MCS, impaired mental health and more symptoms of depression and anxiety. It has previously been shown that relatives are a vulnerable group in terms of mental health, with the relatives’ anxiety being at its highest before chemoradiotherapy and remaining high over time [27]. Worse scores among relatives can be related to a deeper insight into the disease and its prognosis, the personality changes that patients undergo [28, 29], as well as the consequence of these combined factors on family life [27].
Other possible explanations for worse scores in the group of relatives might be uncertainty about the surgery, the fear of being forced to take care of the patient, or being left alone. These feelings can at the other hand also be mixed with feelings of gratitude and privilege of taking care of the patient [29, 30].
There was a relation between patients and relatives with regard to symptoms of anxiety and MCS. This indicates that if impaired mental HRQoL and/or anxiety are present in one group, there is a likelihood that the other group will experience anxiety and/or lower mental HRQoL. The results help to identify especially vulnerable persons, such as patients with comorbidities and patients that have relatives with symptoms of anxiety.
Little is known about how relatives and patients affect each other mentally, especially in case of a glioblastoma diagnosis. Although the notion of close relatives influencing each other seems intuitively an obvious, there are few data on this subject. A previous study did identify a relationship between male relatives and patients with cancer regarding their mental HRQoL. In the same study, however, no relationship was found between female relatives and patients with cancer in terms of mental HRQoL [31].
In the present study, female relatives scored worse levels than male relatives for vitality and social functioning in SF-36, and for symptoms of depression in HAD. Little is known about gender-related differences occurring with relatives of patients with glioblastoma, but it is known that females generally score lower HRQoL than men [11, 12], and it is therefore relevant to study. A previous study reported similar findings—with female relatives of patients with cancer scoring worse overall HRQoL than male relatives [31]. Regardless of whether or not gender is a contributing cause of the reported differences in HRQoL, our data show that relatives estimate themselves worse than patients. Compared with a normative database with selected age and gender-matched sample [19] patients included in our study have lower estimates in HRQoL. The fact that both patients and relatives have affected HRQoL already before surgery illustrates the importance of a person-centered support already early in health care.
Higher age can be an indirect factor that negatively affects HRQoL and emotional well-being. Although some studies indicate that elderly patients still benefit from treatment [32, 33], increasing age will increase the probability of comorbidity, which has a negative impact on mental HRQoL for patients. Thus, the present study has identified comorbidity as a factor contributing towards patients deteriorated mental HRQoL. However, since hypertension was the most common comorbidity it is not possible to ratiocinate too much. Likewise, at older age, lower physical HRQoL can be presumed in patients with glioblastoma and this can affect patients mental HRQoL.
As expected, patients scored lower physical HRQoL than their relatives. Physical impairment can be one of the symptoms of glioblastoma and decreased physical function in patients can affect relatives negatively. Relatives to patients with low functional status (WHO) had a higher risk of poor mental well-being. WHO was still a significant factor despite the fact that in the sample group there was a slightly better WHO than in the dropout group.
The relation between patients and their relatives in the present study suggests that a family serves as a unit and needs to be treated as such. Thus, the focus of care should not only be on the ill person, but also include the relative. For this, it will be important for the relatives to feel welcomed, acknowledged and listened to and be considered as an essential part of the family [34]. Furthermore, research needs to be done regarding interventions for relatives of patients with glioblastoma, on which the body of literature regarding brain tumors is small and inconclusive [35].
Therefore, in the care of seriously ill patients, person-centered care is preferred and in case there are close relatives, he or she should be valued as an important part of this care. The typically heavy workload and frenetic pace associated with an acute neurosurgical care unit may make it difficult to provide and maintain this particular type of care, but our data clearly point out the importance of support and attention for relatives already before surgery.
Glioblastoma is incurable and has a poor prognosis and treatment focus more on QoL for the remaining time, not only for the patient but also for the relatives. This study shows that patients’ and relatives’ emotional well-being are dependent on each other and this even before surgery. In the clinical situation, the propose is that patients and relatives are screened for quality of life and mental health before surgery, to design a support model for early interception and support to patients at risk and their respective relatives.
Conclusion
When a patient is presented with glioblastoma, the disease affects the entire family’s mental health and emotional well-being. This study confirms that HRQoL and emotional well-being are affected in both patients and relatives. In fact, the relatives are more vulnerable at the time point before surgery regarding mental health and emotional well-being than the patients themselves. The results emphasize the importance of providing support, not only for the patients, but also for their relatives, early on at the time point before surgery. It would be attractive if early support, focusing on the caregiver/relative, also benefit patients since close relatives are likely to play an essential role for the wellbeing of patients.
Strengths and limitations
The strength of this study is the study design with a direct comparison between patients and relatives, allowing a family view that includes awareness of relatives’ well-being. To compare data between patients and relatives, well validated generic questionnaires as SF-36 and HADS were chosen. In future studies, it will be of interest to use disease-specific instruments, which however do not allow comparison of pair’s estimates as was the focus of the present study. From an ethical point of view, the number of instruments should also be minimized on already exposed groups. A further strength is that it focuses on an early time point—prior to surgery—since both patients and relatives are already affected at that point and probably need support other than what is currently provided—and indeed more of it.
Concerning limitations, further knowledge of the dynamics of patients’ and relatives’ HRQoL throughout the entire course of the disease would be highly valuable and follow-up studies will be conducted to this end. A relatively large drop-out, could be a limitation, demonstrated in the flow-chart. Although this is a limitation, it is important to study HRQoL issues in diseases like glioblastoma with short survival, which involve severe and disabling symptoms of cognitive decline, personality changes and neurological deficits—even if this leads to fewer participants and larger drop-out rates. A national population based study would of course be of value, but it was not possible to realize. Further, it would be of value to compare the results with the general population, but updated comparable data were not available. Another limitation is that data on education and cultural background are missing. It can be assumed that there may be different experiences depending on the relationship of the relatives to the patient, but due to small samples, these analyses could not be performed. It should be noted though that patients were asked to choose their closest relative, so even if it was not a partner living together with the patient, it was a person standing close to him/her.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ostrom QT, Gittleman H, Truitt G, Boscia A, Kruchko C, Barnholtz-Sloan JS (2018) CBTRUS Statistical Report: primary brain and other central nervous system tumors diagnosed in the United States in 2011–2015. Neuro Oncol. 20(suppl_4):iv1–iv86
Thakkar JP, Dolecek TA, Horbinski C, Ostrom QT, Lightner DD, Barnholtz-Sloan JS et al (2014) Epidemiologic and molecular prognostic review of glioblastoma. Cancer Epidemiol Biomark Prev 23(10):1985–1996
Rønning PA, Helseth E, Meling TR, Johannesen TB (2012) A population-based study on the effect of temozolomide in the treatment of glioblastoma multiforme. Neuro Oncol 14(9):1178–1184
Gittleman H, Boscia A, Ostrom QT, Truitt G, Fritz Y, Kruchko C et al (2018) Survivorship in adults with malignant brain and other central nervous system tumor from 2000–2014. Neuro Oncol 20(suppl_7):vii6–vii16
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC et al (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10(5):459–466
Sizoo EM, Dirven L, Reijneveld JC, Postma TJ, Heimans JJ, Deliens L et al (2014) Measuring health-related quality of life in high-grade glioma patients at the end of life using a proxy-reported retrospective questionnaire. J Neurooncol 116(2):283–290
Edelstein K, Coate L, Massey C, Jewitt NC, Mason WP, Devins GM (2016) Illness intrusiveness and subjective well-being in patients with glioblastoma. J Neurooncol 126(1):127–135
Sagberg LM, Solheim O, Jakola AS (2016) Quality of survival the 1st year with glioblastoma: a longitudinal study of patient-reported quality of life. J Neurosurg 124(4):989–997
McConigley R, Halkett G, Lobb E, Nowak A (2010) Caring for someone with high-grade glioma: a time of rapid change for caregivers. Palliat Med 24(5):473–479
Boele FW, Hoeben W, Hilverda K, Lenting J, Calis AL, Sizoo EM et al (2013) Enhancing quality of life and mastery of informal caregivers of high-grade glioma patients: a randomized controlled trial. J Neurooncol 111(3):303–311
Zahran HS, Kobau R, Moriarty DG, Zack MM, Holt J, Donehoo R (2005) Health-related quality of life surveillance–United States, 1993–2002. Morb Mortal Wkly Rep Surveill Summ 54(4):1–35
Cherepanov D, Palta M, Fryback DG, Robert SA (2010) Gender differences in health-related quality-of-life are partly explained by sociodemographic and socioeconomic variation between adult men and women in the US: evidence from four US nationally representative data sets. Qual Life Res Int J Qual Life Asp Treat Care Rehabil 19(8):1115–1124
Moore G, Collins A, Brand C, Gold M, Lethborg C, Murphy M et al (2013) Palliative and supportive care needs of patients with high-grade glioma and their carers: a systematic review of qualitative literature. Patient Educ Couns 91(2):141–153
Russell B, Collins A, Dally M, Dowling A, Gold M, Murphy M et al (2014) Living longer with adult high-grade glioma: setting a research agenda for patients and their caregivers. J Neurooncol 120(1):1–10
Fekete BWK, Carén H, Ozanne A, Roengren L, Zetterberg H, Tisell M, Smits A, Pontén F, Lindskog C, OlssonBontell T, Jakola AS, Rydenhag B (2019) The Gothenburg population-based glioblastoma research database: methodological aspects and potential impact. Neurooncol Neurosurg 2:4–6
Persson LO, Karlsson J, Bengtsson C, Steen B, Sullivan M (1998) The Swedish SF-36 Health Survey II. Evaluation of clinical validity: results from population studies of elderly and women in Gothenborg. J Clin Epidemiol 51(11):1095–1103
Sullivan M, Karlsson J, Ware JE Jr (1995) The Swedish SF-36 Health Survey--I. Evaluation of data quality, scaling assumptions, reliability and construct validity across general populations in Sweden. Social Sci Med 41(10):1349–1358
Bunevicius A (2017) Reliability and validity of the SF-36 Health Survey Questionnaire in patients with brain tumors: a cross-sectional study. Health Qual Life Outcomes 15(1):92
Sullivan M, Karlsson J, Taft C (2002) SF-36 Hälsoenkät. Svensk Manual och Tolknings guide: SF-36 Health Survey: Swedish Manual and Interpretation Guide. Sahlgrenska University Hospital, Göteborg
Bjelland I, Dahl AA, Haug TT, Neckelmann D (2002) The validity of the Hospital Anxiety and Depression Scale: an updated literature review. J Psychosomatic Res 52:69–77
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67(6):361–370
World Health Organization (1979) WHO handbook for reporting results of cancer treatment. WHO, Geneva
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET et al (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5(6):649–655
Charlson ME, Pompei P, Ales KL, Mackenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Bunevicius A, Tamasauskas S, Deltuva V, Tamasauskas A, Radziunas A, Bunevicius R (2014) Predictors of health-related quality of life in neurosurgical brain tumor patients: focus on patient-centered perspective. Acta Neurochir 156(2):367–374
Rooney AG, Carson A, Grant R (2011) Depression in cerebral glioma patients: a systematic review of observational studies. J Natl Cancer Inst 103(1):61–76
Halkett GK, Lobb EA, Shaw T, Sinclair MM, Miller L, Hovey E et al (2017) Distress and psychological morbidity do not reduce over time in carers of patients with high-grade glioma. Support Care Cancer Off J Multinatl Assoc Support Care Cancer 25(3):887–893
Pace A, Dirven L, Koekkoek JAF, Golla H, Fleming J, Rudà R et al (2017) European Association for Neuro-Oncology (EANO) guidelines for palliative care in adults with glioma. Lancet Oncol 18(6):e330–e340
Coolbrandt A, Sterckx W, Clement P, Borgenon S, Decruyenaere M, de Vleeschouwer S et al (2015) Family caregivers of patients with a high-grade glioma: a qualitative study of their lived experience and needs related to professional care. Cancer Nurs 38(5):406–413
Breen LJ, Aoun SM, O’Connor M, Howting D, Halkett GKB (2018) Family caregivers’ preparations for death: a qualitative analysis. J Pain Symptom Manag 55(6):1473–1479
Bergelt C, Koch U, Petersen C (2008) Quality of life in partners of patients with cancer. Qual Life Res Int J Qual Life Aspects Treat Care Rehabil 17(5):653–663
Keime-Guibert F, Chinot O, Taillandier L, Cartalat-Carel S, Frenay M, Kantor G et al (2007) Radiotherapy for glioblastoma in the elderly. N Engl J Med 356(15):1527–1535
Youssef M, Ludmir EB, Mandel JJ, Patel AJ, Jalali A, Treiber J et al (2019) Treatment strategies for glioblastoma in older patients: age is just a number. J Neurooncol 145(2):357–364
Gervais C, Verdon C, deMontigny F, Leblanc L, Lalande D (2020) Creating a space to talk about one’s experience of suffering: families’ experience of a family nursing intervention. Scand J Caring Sci 34(2):446–455
Boele FW, Rooney AG, Bulbeck H, Sherwood P (2019) Interventions to help support caregivers of people with a brain or spinal cord tumour. Cochrane Database Syst Rev 7(7):Cd012582
Acknowledgements
The authors would like to acknowledge all patients and their relatives who generously participated in this study. The authors also acknowledge research assistants and nurses Anneli Johansson, Lena Nilsson, Stina Svensson and Ünzüle Yildiz for excellent data management.
Funding
Open access funding provided by University of Gothenburg. The agreement between the Swedish government and the county councils, the ALF-agreement ALFGBG-716671, ALFGBG-717021, Health Medical Care Committee at Västra Götaland Regional FoU-support, VGFOUREG-750851, AFA Research Foundation, Margaretahemmet foundation, Gothenburg Foundation for Neurological Research and Sahlgrenska Academy and Institute of Health and Care Science, University of Gothenburg.
Author information
Authors and Affiliations
Contributions
Conceptualization (PS, BR, AO), Data curation (PS, BF, BR, AO), Formal analysis (PS, IH, AO), Investigation (PS, BF, BR, AO) Methodology (PS, IH, AJ, AS, BR, AO), Validation (PS, IH, AS, AJ, BR, AO), Visualization (PS, AO), Writing—original draft (PS, AO), Writing—review & editing (PS, BF, IH, AJ, AS, BR, AO). All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest.
The authors declare that they have no conflict of interest.
Consent to participate
Informed consent was obtained from all participants in this study.
Consent to publication
Consent to publish was obtained from all the participants in this study.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Regional Ethical Review Board in Gothenburg, dnr. 559-12. License number for SF-36 is QM037698.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ståhl, P., Fekete, B., Henoch, I. et al. Health-related quality of life and emotional well-being in patients with glioblastoma and their relatives. J Neurooncol 149, 347–356 (2020). https://doi.org/10.1007/s11060-020-03614-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-020-03614-5