Abstract
Background
Molecular markers of WHO grade II/III glioma are known to have important prognostic and predictive implications and may be associated with unique imaging phenotypes. The purpose of this study is to determine whether three clinically relevant molecular markers identified in gliomas—IDH, 1p/19q, and MGMT status—show distinct quantitative MRI characteristics on FLAIR imaging.
Methods
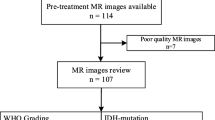
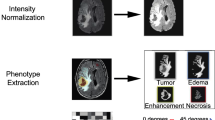
Sixty-one patients with grade II/III gliomas who had molecular data and MRI available prior to radiation were included. Quantitative MRI features were extracted that measured tissue heterogeneity (homogeneity and pixel correlation) and FLAIR border distinctiveness (edge contrast; EC). T-tests were conducted to determine whether patients with different genotypes differ across the features. Logistic regression with LASSO regularization was used to determine the optimal combination of MRI and clinical features for predicting molecular subtypes.
Results
Patients with IDH wildtype tumors showed greater signal heterogeneity (p = 0.001) and lower EC (p = 0.008) within the FLAIR region compared to IDH mutant tumors. Among patients with IDH mutant tumors, 1p/19q co-deleted tumors had greater signal heterogeneity (p = 0.002) and lower EC (p = 0.005) compared to 1p/19q intact tumors. MGMT methylated tumors showed lower EC (p = 0.03) compared to the unmethylated group. The combination of FLAIR border distinctness, heterogeneity, and pixel correlation optimally classified tumors by IDH status.
Conclusion
Quantitative imaging characteristics of FLAIR heterogeneity and border pattern in grade II/III gliomas may provide unique information for determining molecular status at time of initial diagnostic imaging, which may then guide subsequent surgical and medical management.



Similar content being viewed by others
References
Cancer Genome Atlas Research Network, Brat DJ, Verhaak RGW, Aldape KD, Yung WKA, Salama SR et al. (2015) Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N Engl J Med 372, 2481–2498. https://doi.org/10.1056/NEJMoa1402121
Buckner J, Giannini C, Eckel-Passow J, Lachance D, Parney I, Laack N et al (2017) Management of diffuse low-grade gliomas in adults - use of molecular diagnostics. Nat Rev Neurol 13:340–351. https://doi.org/10.1038/nrneurol.2017.54
Eckel-Passow JE, Lachance DH, Molinaro AM, Walsh KM, Decker PA, Sicotte H et al (2015) Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N Engl J Med 372:2499–2508. https://doi.org/10.1056/NEJMoa1407279
Wang Y, Zhang T, Li S, Fan X, Ma J, Wang L et al (2015) Anatomical localization of isocitrate dehydrogenase 1 mutation: a voxel-based radiographic study of 146 low-grade gliomas. Eur J Neurol 22:348–354. https://doi.org/10.1111/ene.12578
Sanson M, Marie Y, Paris S, Idbaih A, Laffaire J, Ducray F et al (2009) Isocitrate dehydrogenase 1 codon 132 mutation is an important prognostic biomarker in gliomas. J Clin Oncol 27:4150–4154. https://doi.org/10.1200/JCO.2009.21.9832
Jenkins RB, Blair H, Ballman KV, Giannini C, Arusell RM, Law M et al (2006) A t(1;19)(q10;p10) mediates the combined deletions of 1p and 19q and predicts a better prognosis of patients with oligodendroglioma. Can Res 66:9852–9861. https://doi.org/10.1158/0008-5472.CAN-06-1796
van den Bent MJ, Erdem-Eraslan L, Idbaih A, de Rooi J, Eilers PHC, Spliet WGM et al (2013) MGMT-STP27 methylation status as predictive marker for response to PCV in anaplastic oligodendrogliomas and oligoastrocytomas. A report from EORTC study 26951. Clin Cancer Res 19:5513–5522. https://doi.org/10.1158/1078-0432.CCR-13-1157
Reifenberger J, Reifenberger G, Liu L, James CD, Wechsler W, Collins VP (1994) Molecular genetic analysis of oligodendroglial tumors shows preferential allelic deletions on 19q and 1p. Am J Pathol 145:1175–1190
Cairncross G, Wang M, Shaw E, Jenkins R, Brachman D, Buckner J et al (2013) Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402. J Clin Oncol 31:337–343. https://doi.org/10.1200/JCO.2012.43.2674
van den Bent MJ, Brandes AA, Taphoorn MJB, Kros JM, Kouwenhoven MCM, Delattre J-Y et al (2013) Adjuvant procarbazine, lomustine, and vincristine chemotherapy in newly diagnosed anaplastic oligodendroglioma: long-term follow-up of EORTC brain tumor group study 26951. J Clin Oncol 31:344–350. https://doi.org/10.1200/JCO.2012.43.2229
Watanabe T, Nobusawa S, Kleihues P, Ohgaki H (2009) IDH1 mutations are early events in the development of astrocytomas and oligodendrogliomas. Am J Pathol 174:1149–1153. https://doi.org/10.2353/ajpath.2009.080958
Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W et al (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773. https://doi.org/10.1056/NEJMoa0808710
Hartmann C, Meyer J, Balss J, Capper D, Mueller W, Christians A et al (2009) Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: a study of 1,010 diffuse gliomas. Acta Neuropathol 118:469–474. https://doi.org/10.1007/s00401-009-0561-9
Duncan CG, Barwick BG, Jin G, Rago C, Kapoor-Vazirani P, Powell DR et al (2012) A heterozygous IDH1R132H/WT mutation induces genome-wide alterations in DNA methylation. Genome Res 22:2339–2355. https://doi.org/10.1101/gr.132738.111
Noushmehr H, Weisenberger DJ, Diefes K, Phillips HS, Pujara K, Berman BP et al (2010) Identification of a CpG island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell 17:510–522. https://doi.org/10.1016/j.ccr.2010.03.017
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK et al (2016) The 2016 world health organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Wick W, Meisner C, Hentschel B, Platten M, Schilling A, Wiestler B et al (2013) Prognostic or predictive value of MGMT promoter methylation in gliomas depends on IDH1 mutation. Neurology 81:1515–1522. https://doi.org/10.1212/WNL.0b013e3182a95680
Dean BL, Drayer BP, Bird CR, Flom RA, Hodak JA, Coons SW et al (1990) Gliomas: classification with MR imaging. Radiology 174:411–415. https://doi.org/10.1148/radiology.174.2.2153310
Hajnal JV, Bryant DJ, Kasuboski L, Pattany PM, De Coene B, Lewis PD et al (1992) Use of fluid attenuated inversion recovery (FLAIR) pulse sequences in MRI of the brain. J Comput Assist Tomogr 16:841–844
Law M, Oh S, Babb JS, Wang E, Inglese M, Zagzag D et al (2006) Low-grade gliomas: dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging—prediction of patient clinical response. Radiology 238:658–667. https://doi.org/10.1148/radiol.2382042180
Sugahara, T., Korogi, Y., Kochi, M., Ikushima, I., Shigematu, Y., Hirai, T. et al. (1999) Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging, 9, 53–60.
Forst DA, Nahed BV, Loeffler JS, Batchelor TT (2014) Low-grade gliomas. Oncologist 19:403–413. https://doi.org/10.1634/theoncologist.2013-0345
Narang AK, Chaichana KL, Weingart JD, Redmond KJ, Lim M, Olivi A et al (2017) Progressive low-grade glioma: assessment of prognostic importance of histologic reassessment and MRI findings. World Neurosurg 99:751–757. https://doi.org/10.1016/j.wneu.2016.04.030
Cuccarini V, Erbetta A, Farinotti M, Cuppini L, Ghielmetti F, Pollo B et al (2016) Advanced MRI may complement histological diagnosis of lower grade gliomas and help in predicting survival. J Neuro-Oncol 126:279–288. https://doi.org/10.1007/s11060-015-1960-5
QI S, L. YU, LI, H., OU, Y., QIU, X., DING, Y. et al (2014) Isocitrate dehydrogenase mutation is associated with tumor location and magnetic resonance imaging characteristics in astrocytic neoplasms. Oncol Lett 7:1895–1902. https://doi.org/10.3892/ol.2014.2013
Tietze A, Choi C, Mickey B, Maher EA, Parm Ulhøi B, Sangill R et al (2018) Noninvasive assessment of isocitrate dehydrogenase mutation status in cerebral gliomas by magnetic resonance spectroscopy in a clinical setting. J Neurosurg 128:391–398. https://doi.org/10.3171/2016.10.JNS161793
Metellus P, Coulibaly B, Colin C, de Paula AM, Vasiljevic A, Taieb D et al (2010) Absence of IDH mutation identifies a novel radiologic and molecular subtype of WHO grade II gliomas with dismal prognosis. Acta Neuropathol 120:719–729. https://doi.org/10.1007/s00401-010-0777-8
Chawla S, Krejza J, Vossough A, Zhang Y, Kapoor GS, Wang S et al (2013) Differentiation between oligodendroglioma genotypes using dynamic susceptibility contrast perfusion-weighted imaging and proton MR spectroscopy. Am J Neuroradiol 34:1542–1549. https://doi.org/10.3174/ajnr.A3384
Weller M, Stupp R, Hegi ME, van den Bent M, Tonn JC, Sanson M et al (2012) Personalized care in neuro-oncology coming of age: why we need MGMT and 1p/19q testing for malignant glioma patients in clinical practice. Neuro Oncol 14(Suppl 4):iv100-108. https://doi.org/10.1093/neuonc/nos206
Kim JW, Park C-K, Park S-H, Kim YH, Han JH, Kim C-Y et al (2011) Relationship between radiological characteristics and combined 1p and 19q deletion in World Health Organization grade III oligodendroglial tumours. J Neurol Neurosurg Psychiatry 82:224–227. https://doi.org/10.1136/jnnp.2009.178806
Megyesi JF, Kachur E, Lee DH, Zlatescu MC, Betensky RA, Forsyth PA et al (2004) Imaging correlates of molecular signatures in oligodendrogliomas. Clin Cancer Res 10:4303–4306. https://doi.org/10.1158/1078-0432.CCR-04-0209
Jenkinson MD, du Plessis DG, Smith TS, Joyce KA, Warnke PC, Walker C (2006) Histological growth patterns and genotype in oligodendroglial tumours: correlation with MRI features. Brain 129:1884–1891. https://doi.org/10.1093/brain/awl108
Eoli M, Menghi F, Bruzzone MG, De Simone T, Valletta L, Pollo B et al (2007) Methylation of O6-methylguanine DNA methyltransferase and loss of heterozygosity on 19q and/or 17p are overlapping features of secondary glioblastomas with prolonged survival. Clin Cancer Res 13:2606–2613. https://doi.org/10.1158/1078-0432.CCR-06-2184
Darlix A, Deverdun J, Menjot de Champfleur N, Castan F, Zouaoui S, Rigau V et al (2017) IDH mutation and 1p19q codeletion distinguish two radiological patterns of diffuse low-grade gliomas. J Neuro Oncol 133:37–45. https://doi.org/10.1007/s11060-017-2421-0
Delfanti R, Piccioni D, Bahrami N, Dale A, McDonald CR, Farid N (2017) Imaging correlates for the 2016 update on WHO classification of grade II/III gliomas: implications for IDH, 1p/19q and ATRX status. J Neuro Oncol
Smits M, van den Bent MJ (2017) Imaging correlates of adult glioma genotypes. Radiology 284:316–331. https://doi.org/10.1148/radiol.2017151930
Jovicich J, Czanner S, Greve D, Haley E, van der Kouwe A, Gollub R et al (2006) Reliability in multi-site structural MRI studies: effects of gradient non-linearity correction on phantom and human data. NeuroImage 30:436–443. https://doi.org/10.1016/j.neuroimage.2005.09.046
Matlab—graycoprops [Internet]
Mahmoud-Ghoneim D, Toussaint G, Constans JM, de Certaines JD (2003) Three dimensional texture analysis in MRI: a preliminary evaluation in gliomas. Magn Reson Imaging 21:983–987
Nachimuthu DS, Baladhandapani A (2014) Multidimensional texture characterization: on analysis for brain tumor tissues using MRS and MRI. J Digit Imaging 27:496–506. https://doi.org/10.1007/s10278-013-9669-5
Li X, Xia H, Zhou Z, Tong L (2010) 3D texture analysis of hippocampus based on MR images in patients with alzheimer disease and mild cognitive impairment. In: 2010 3rd international conference on biomedical engineering and informatics, p. 1–4. https://doi.org/10.1109/BMEI.2010.5639520
Suoranta S, Holli-Helenius K, Koskenkorva P, Niskanen E, Könönen M, Äikiä M et al (2013) 3D texture analysis reveals imperceptible MRI textural alterations in the thalamus and putamen in progressive myoclonic epilepsy type 1, EPM1. PLoS ONE 8:e69905. https://doi.org/10.1371/journal.pone.0069905
Zhang J, Yu C, Jiang G, Liu W, Tong L (2012) 3D texture analysis on MRI images of Alzheimer’s disease. Brain Imaging Behav 6:61–69. https://doi.org/10.1007/s11682-011-9142-3
Bahrami N, Dale A, White NS, Hattangadi-Gluth JA, Farid N, Piccioni DE et al. (2017) Edge contrast of the FLAIR hyperintense region predicts survival in patients with high grade gliomas following treatment with bevacizumab. Am J Neuroradiol
Claes A, Idema AJ, Wesseling P (2007) Diffuse glioma growth: a guerilla war. Acta Neuropathol 114:443–458. https://doi.org/10.1007/s00401-007-0293-7
Wang Y, Fan X, Zhang C, Zhang T, Peng X, Li S et al (2014) Anatomical specificity of O6-methylguanine DNA methyltransferase protein expression in glioblastomas. J Neuro Oncol 120:331–337. https://doi.org/10.1007/s11060-014-1555-6
Carrillo JA, Lai A, Nghiemphu PL, Kim HJ, Phillips HS, Kharbanda S et al (2012) Relationship between tumor enhancement, edema, IDH1 mutational status, MGMT promoter methylation, and survival in glioblastoma. AJNR Am J Neuroradiol 33:1349–1355. https://doi.org/10.3174/ajnr.A2950
Bahrami N, Seibert TM, Karunamuni R, Bartsch H, Krishnan A, Farid N et al (2017) Altered network topology in patients with primary brain tumors after fractionated radiotherapy. Brain Connect 7:299–308. https://doi.org/10.1089/brain.2017.0494
Drabycz S, Roldán G, de Robles P, Adler D, McIntyre JB, Magliocco AM et al (2010) An analysis of image texture, tumor location, and MGMT promoter methylation in glioblastoma using magnetic resonance imaging. NeuroImage 49:1398–1405. https://doi.org/10.1016/j.neuroimage.2009.09.049
Cohen A, Holmen S, Colman H (2013) IDH1 and IDH2 mutations in gliomas. Curr Neurol Neurosci Rep 13:345. https://doi.org/10.1007/s11910-013-0345-4
Claus EB, Walsh KM, Wiencke J, Molinaro AM, Wiemels JL, Schildkraut JM et al (2015) Survival and low grade glioma: the emergence of genetic information. Neurosurg Focus 38:E6. https://doi.org/10.3171/2014.10.FOCUS12367
Acknowledgements
We would like to thank patients at the UCSD Moores Cancer Center Neuro-Oncology Program for their generous participation.
Funding
We also acknowledge the funding from National Institutes of Health grants R01NS065838 (C.R.M.); National Institutes of Health UL1TR000100 (J.A.H.) and KL2TR00144 (J.A.H.); American Cancer Society Award ACS-IRG 70-002 (J.A.H.) and American Cancer Society RSG-15-229-01-CCE (C.R.M).
Author information
Authors and Affiliations
Contributions
NB—primary conception and design of the study, gathering, analysis and interpretation of data, primary drafting and editing of the manuscript. SH—assistance in conception of the study and gathering the data. DEP—assistance in design of the study, gathering, analysis and interpretation of data. RK—analysis and interpretation of data. YHC—analysis and interpretation of data. NSW—analysis and interpretation of data. RLD—gathering, analysis and interpretation of data. TM—analysis and interpretation of data. JAHG—analysis and interpretation of data. AMD—analysis and interpretation of data. NF—conception and design of the study, acquisition, analysis and interpretation of data. CR—conception and design of the study, gathering, analysis and interpretation of data.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any personal or financial interest in drugs, materials, or devices described in this submission.
Rights and permissions
About this article
Cite this article
Bahrami, N., Hartman, S.J., Chang, YH. et al. Molecular classification of patients with grade II/III glioma using quantitative MRI characteristics. J Neurooncol 139, 633–642 (2018). https://doi.org/10.1007/s11060-018-2908-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2908-3