Abstract
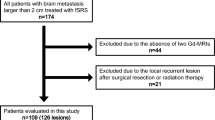
Stereotactic radiotherapy (SRT) of brain metastasis can lead to lesion growth caused by radiation toxicity. The pathophysiology of this so-called pseudo-progression is poorly understood. The purpose of this study was to evaluate the use of MRI cine-loops for describing the consecutive events in this radiation induced lesion growth. Ten patients were selected from our department’s database that had received SRT of brain metastases and had lesion growth caused by pseudo-progression as well as at least five follow-up MRI scans. Pre- and post SRT MRI scans were co-registered and cine-loops were made using post-gadolinium 3D T1 axial slices. The ten cine loops were discussed in a joint meeting of the authors. The use of cine-loops was superior to evaluation of separate MRI scans for interpretation of events after SRT. There was a typical lesion evolution pattern in all patients with varying time course. Initially regression of the metastases was observed, followed by an enlarging area of new contrast enhancement in the surrounding brain tissue. Analysis of consecutive MRI’s using cine-loops may improve understanding of pseudo-progression. It probably represents a radiation effect in brain tissue surrounding the irradiated metastasis and not enlargement of the metastasis itself.



Similar content being viewed by others
References
Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47:207–214
Brandsma D, Stalpers L, Taal W, Sminia P, van den Bent MJ (2008) Clinical features, mechanisms, and management of pseudoprogression in malignant gliomas. Lancet Oncol 9:453–461
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Ruzevick J, Kleinberg L, Rigamonti D (2014) Imaging changes following stereotactic radiosurgery for metastatic intracranial tumors: differentiating pseudoprogression from tumor progression and its effect on clinical practice. Neurosurg Rev 37:193–201
Wiggenraad R, Verbeek-de KA, Mast M et al (2012) Local progression and pseudo progression after single fraction or fractionated stereotactic radiotherapy for large brain metastases. A single centre study. Strahlenther Onkol 188:696–701
Lin NU, Lee EQ, Aoyama H et al (2013) Challenges relating to solid tumour brain metastases in clinical trials, part 1: patient population, response, and progression. A report from the RANO group. Lancet Oncol 14:e396–e406
Lawrence YR, Li XA, El Naqa I et al (2010) Radiation dose-volume effects in the brain. Int J Radiat Oncol Biol Phys 76(3):S20–S27
Minniti G, Clarke E, Lanzetta G et al (2011) Stereotactic radiosurgery for brain metastases: analysis of outcome and risk of brain radionecrosis. Radiat Oncol 6:48
Jagannathan J, Petit JH, Balsara K, Hudes R, Chin LS (2004) Long-term survival after gamma knife radiosurgery for primary and metastatic brain tumors. Am J Clin Oncol 27:441–444
Kocher M, Soffietti R, Abacioglu U et al (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol 29:134–141
Wiggenraad R, Verbeek-de KA, Kal HB et al (2011) Dose-effect relation in stereotactic radiotherapy for brain metastases. A systematic review. Radiother Oncol 98:292–297
Patel TR, McHugh BJ, Bi WL et al (2011) A comprehensive review of MR imaging changes following radiosurgery to 500 brain metastases. AJNR Am J Neuroradiol 32:1885–1892
Molenaar R, Wiggenraad R, Verbeek-de KA, Walchenbach R, Vecht C (2009) Relationship between volume, dose and local control in stereotactic radiosurgery of brain metastasis. Br J Neurosurg 23:170–178
Hoefnagels FW, Lagerwaard FJ, Sanchez E et al (2009) Radiological progression of cerebral metastases after radiosurgery: assessment of perfusion MRI for differentiating between necrosis and recurrence. J Neurol 256:878–887
Dinkel J, Hintze C, Tetzlaff R et al (2009) 4D-MRI analysis of lung tumor motion in patients with hemidiaphragmatic paralysis. Radiother Oncol 91:449–454
Sonke JJ, Belderbos J (2010) Adaptive radiotherapy for lung cancer. Semin Radiat Oncol 20:94–106
Barretto RP, Ko TH, Jung JC et al (2011) Time-lapse imaging of disease progression in deep brain areas using fluorescence microendoscopy. Nat Med 17:223–228
Fink J, Born D, Chamberlain MC (2012) Radiation necrosis: relevance with respect to treatment of primary and secondary brain tumors. Curr Neurol Neurosci Rep 12:276–285
Giglio P, Gilbert MR (2003) Cerebral radiation necrosis. Neurologist 9:180–188
Nonoguchi N, Miyatake S, Fukumoto M et al (2011) The distribution of vascular endothelial growth factor-producing cells in clinical radiation necrosis of the brain: pathological consideration of their potential roles. J Neurooncol 105:423–431
Conflict of interest
R. Wiggenraad has received speakers fee from Brainlab.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MPG 1686 kb)
Supplementary material 2 (MPG 5452 kb)
Rights and permissions
About this article
Cite this article
Wiggenraad, R., Bos, P., Verbeek-de Kanter, A. et al. Pseudo-progression after stereotactic radiotherapy of brain metastases: lesion analysis using MRI cine-loops. J Neurooncol 119, 437–443 (2014). https://doi.org/10.1007/s11060-014-1519-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-014-1519-x