Abstract
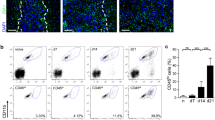
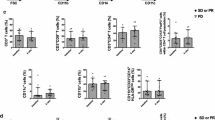
Glioblastoma multiforme (GBM) is the most aggressive primary human brain tumor. The relatively high amount of T regulatory lymphocytes present in the tumor, contributes to the establishment of an immunosuppressive microenvironment. Samples of peripheral blood were collected from GBM patients and healthy controls and a purified population of Treg (CD4+/CD25bright) was isolated using flow cytometric cell sorting. Treg migrating capacities toward human glioma cell line conditioned medium were evaluated through an in vitro migration test. Our data show that supernatants collected from GBM cell lines were more attractant to Treg when compared to complete standard medium. The addition of an anti-CCL2 antibody to conditioned medium decreased conditioned medium-depending Treg migration, suggesting that CCL2 (also known as Monocyte Chemoattractant Protein, MCP-1) is implicated in the process. The number of circulating CD4+/μL or Treg/μL was similar in GBM patients and controls. Specific Treg markers (FOXP3; CD127; Helios; GITR; CTLA4; CD95; CCR2, CCR4; CCR7) were screened in peripheral blood and no differences could be detected between the two populations. These data confirm that the tumor microenvironment is attractive to Treg, which tend to migrate toward the tumor region changing the immunological response. Though we provide evidence that CCL2 is implicated in Treg migration, other factors are needed as well to provide such effect.





Similar content being viewed by others
References
Wainwright DA, Nigam P, Thaci B et al (2012) Recent developments on immunotherapy for brain cancer. Expert Opin Emerg Drugs 17(2):181–202
Behin A, Hoang-Xuan K, Carpentier AF et al (2003) Primary brain tumors in adults. Lancet 361:323–331
Curtin JF, Candolfi M, Fakhouri TM et al (2008) Treg depletion inhibits efficacy of cancer immunotherapy: implications for clinical trials. PLoS ONE 3(4):e1983
Crane CA, Ahn BJ, Han SJ et al (2012) Soluble factors secreted by glioblastoma cell lines facilitate recruitment, survival, and expansion of regulatory T cells: implications for immunotherapy. Neuro Oncol 14(5):584–595
Fujimoto S, Greene MI, Sehon AH (1976) Regulation of the immuneresponse to tumor antigens. II. The nature of immunosuppressor cells in tumor-bearing hosts. J Immunol 116:800–806
Ha TY, Park HJ, On KK et al (1995) Potentiation of anti-tumor immunity by modulation of suppressor T cells. II. Regression of tumor by modulation of suppressor T cells. Korean J Immunol 17:101–118
Ha TY (2009) The role of regulatory T cells in cancer. Immune Netw 9(6):209–235
Chen X, Oppenheim JJ (2011) Resolving the identity myth: key markers of functional CD4+ FoxP3+ regulatory T cells. Int Immunopharmacol 11(10):1489–1496
Wang RF (2006) Regulatory T cells and innate immune regulation in tumor immunity. Springer Semin Imunopathol 28:17–23
Fietta AM, Morosini M, Passadore I et al (2009) Systemic inflammatory response and downmodulation of peripheral CD25+ Foxp3+ T-regulatory cells in patients undergoing radiofrequency thermal ablation for lung cancer. Human Immunol 70:477–486
Liu Z, Kim JH, Falo LD et al (2009) Tumor regulatory T cells potently abrogate antitimor immunity. J Immunol 182:6160–6167
El Andaloussi A, Lesniak MS (2006) An increase in CD4+ CD25+ FOXP3+ regulatory T cells in tumor infiltrating lymphocytes of human glioblastoma multiforme. Neuro Oncol 8:234–243
Heimberger AB, Abou-Ghazal M, Reina-Ortiz C et al (2008) Incidence and prognostic impact of FoxP3+ regulatory T cells in human gliomas. Clin Cancer Res 14:5166–5172
Jacobs JF, Idema AJ, Bol KF et al (2010) Prognostic significance and mechanism of Treg infiltration in human brain tumors. J Neuroimmunol 225:195–199
Fecci PE, Mitchell DA, Whitesides JF et al (2006) Increased regulatory T-cell fraction amidst a diminished CD4 compartment explains cellular immune defects in patients with malignant glioma. Cancer Res 66(6):3294–3302
Zhang J, Patel L, Pienta KJ (2010) Targeting chemokine (C–C motif) ligand 2 (CCL2) as an example of translation of cancer molecular biology to the clinic. Prog Mol Biol Transl Sci 95:31–53
Deshmane SL, Kremlev S, Amini S et al (2009) Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interferon Cytokine Res 29:313–326
Del Pozo MA, Cabanas C, Montoya MC et al (1997) ICAMs redistributed by chemokines to cellular uropods as a mechanism for recruitment of T lymphocytes. J Cell Biol 137:493–508
Carrillo-de Sauvage MA, Gómez A, Ros CM (2012) CCL2-expressing astrocytes mediate the extravasation of T lymphocytes in the brain. Evidence from patients with glioma and experimental models in vivo. PLoS ONE 7:30762
Calatozzolo C, Canazza A, Pollo B et al (2011) Expression of the new CXCL12 receptor, CXCR7, in gliomas. Cancer Biol Ther 11(242–53):17
Conductier G, Blondeau N, Guyon A et al (2010) The role of monocyte chemoattractant protein CCL2/CCL2 in neuroinflammatory diseases. J Neuroimmunol 224:93–100
Kim YH, Jung TY, Jung S et al (2012) Tumor-infiltrating T-cell subpopulations in glioblastomas. Br J Neurosurg 26:21–27
Donson AM, Birks DK, Schittone SA et al (2012) Increased immune gene expression and immune cell infiltration in high grade astrocytoma distinguish long-term from short-term survivors. J Immunol 189:1920–1927
Lowther DE, Hafler DA (2012) Regulatory T cells in the central nervous system. Immunol Rev 248(1):156–169
Jordan JT, Sun W, Hussain SF et al (2008) Preferential migration of regulatory T cells mediated by glioma-secreted chemokines can be blocked with chemotherapy. Cancer Immunol Immunother 57(1):123–131
Parney IF, Waldron J, Parsa AT (2009) Flow citometry and in vitro analysis of human glioma associated macrophages. J Neurosurg 110(3):572–582
Lohr J, Ratliff T, Huppertz A et al (2011) Effector T-cell infiltration positively impacts survival of glioblastoma patients and is impaired by tumor-derived TGF-β. Clin Cancer Res 17(13):4296–4308
de Chaisemartin L, Goc J, Damotte D et al (2011) Characterization of chemokines and adhesion molecules associated with T cell presence in tertiary lymphoid structures in human lung cancer. Cancer Res 71(20):6391–6399
Wei S, Kryczek I, Zou W (2006) Regulatory T-cell compartmentalization and trafficking. Blood 108(2):426–431
Ménétrier-Caux C, Faget J, Biota C, Gobert M, Blay JY, Caux C (2012) Innate immune recognition of breast tumor cells mediates CCL2 secretion favoring Treg recruitment within tumorenvironment. Oncoimmunology 1(5):759–761
Weller M (2012) More on the immune privilege of glioblastoma. Neuro Oncol 14(5):527–528
Nalla AK, Gogineni VR, Gupta R et al (2011) Suppression of uPA and uPAR blocks radiation-induced CCL2 mediated recruitment of endothelial cells in meningioma. Cell Signal 23(8):1299–1310
Yan M, Jene N, Byrne D et al (2011) Recruitment of regulatory T cells is correlated with hypoxia-induced CXCR4 expression, and is associated with poor prognosis in basal-like breast cancers. Breast Cancer Res 13(2):R47
Wainwright DA, Balyasnikova IV, Chang AL et al (2012) IDO Expression in brain tumors increases the recruitment of regulatory T cells and negatively impacts survival. Clin Cancer Res 18(22):6110–6121
Coe D, Begom S, Addey C et al (2010) Depletion of regulatory T cells by anti-GITR mAb as a novel mechanism for cancer immunotherapy. Cancer Immunol Immunother 59:1367–1377
Cohen AD, Schaer DA, Liu C et al (2010) Agonist anti-GITR monoclonal antibody induces melanoma tumor immunity in mice by altering regulatory T cell stability and intra-tumor accumulation. PLoS ONE 5:e10436
Faget J, Bendriss-Vermare N, Gobert M et al (2012) ICOS-ligand expression on plasmacytoid dendritic cells supports breast cancer progression by promoting the accumulation of immunosuppressive CD4+ T cells. Cancer Res 72:6130–6141
Acknowledgments
This work was supported by research Grant of the Ministero della Salute (LR9 IRCCS Foundation Neurological Institute “C. Besta”, Milan Italy). The authors are grateful to Luisa Fumagalli and Francesco Ghielmetti from Radiotherapy Unit (Foundation IRCCS Neurological Institute C. Besta) for the skillful technical help with irradiation experiments. We are also thankful to Marta Dossena from Cellular Neurobiology Laboratory, Cerebrovascular Diseases Unit (Foundation IRCCS Neurological Institute C. Besta) for her contribution on data analysis.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Chiara Vasco and Alessandra Canazza have equally contributed to this work.
Rights and permissions
About this article
Cite this article
Vasco, C., Canazza, A., Rizzo, A. et al. Circulating T regulatory cells migration and phenotype in glioblastoma patients: an in vitro study. J Neurooncol 115, 353–363 (2013). https://doi.org/10.1007/s11060-013-1236-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-013-1236-x