Summary
Purpose
To determine the response rate, time to disease progression, survival, and toxicity of intravenous carboplatin and chronic oral high-dose tamoxifen in patients with recurrent malignant gliomas.
Patients and methods
Patients with histological confirmation of recurrent malignant gliomas were eligible for this multicenter phase II trial. Treatment consisted of 400 mg/m2 carboplatin intravenously every 4 weeks and oral high dose chronic tamoxifen (80 mg bid in women and 100 mg bid in men).
Results
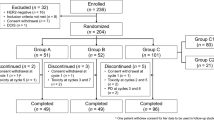
Twenty seven patients met the eligibility criteria and were evaluable for response. The histological subtypes were: 16 (59%) glioblastoma multiforme (GBM), malignant astrocytoma (5 patients), malignant mixed glioma (5 patients), and glioblastoma/gliosarcoma (1 patient). Twenty-two patients (82%) had an ECOG performance status of 0 or 1. No complete responses were observed, 4 patients (15%) achieved a partial response, and 14 patients (52%) had stable disease. Median time to progression was 3.65 months (95%CI 2.56, 4.83). Median overall survival was 14.09 months (95%CI 7.06, 19.91). One patient with a recurrent GBM had a sustained partial response and is progression free 81 months since starting treatment. Another patient with mixed malignant oligoastrocytoma also had a prolonged partial response (lasting 63 months) and is alive 84 months after treatment for recurrence. The most frequently reported grade 3 or 4 toxicities were fatigue (19%), nausea (11%) and anorexia (11%).
Conclusions
Carboplatin and high dose tamoxifen has similar response rates to other regimens for recurrent malignant gliomas and are probably equivalent to those found using tamoxifen as monotherapy. Long-lasting periods of disease free survival in some patients (particularly those with malignant mixed oligo astrocytomas) were found.
Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, Radiotherapy plus concomitant and adjuvant temozolomide for glioblastomaN Eng J Med352: 987–996, 2005
Scott JN, Rewcastle NB, Hagen NA, Brasher PMA, Fulton D, MacKinnon JA, Hamiltong M, Cairncross JG, Forsyth P, Which glioblastoma multiforme patient will become a long-term survivor? A population based studyAnn Neurol46:183–188, 1999
Dropcho EJ, 2001. Novel chemotherapeutic approaches to brain tumorsHematol Oncol Clin North Am 15:1027–1052
Wong ET, Hess KR, Gleason MJ, Jaeckle KA, Kyritsis AP, Prados MD, Levin VA, Yung WK, Outcomes and prognostic factors in recurrent glioma patients enrolled onto phase II clinical trialsJ Clin Oncol17:2572–2578, 1999
Go RS, Adjei AA, Review of the comparative pharmacology and clinical activity of cisplatin and carboplatinJ Clin Oncol17:409–422, 1999
Riccardi R, Riccardi A, Di Rocco C, Carelli G, Tartaglia RL, Lasorella A, Servidei T, Mastrangelo R, Cerebrospinal fluid pharmacokinetics of carboplatin in children with brain tumorsCancer Chemother Pharmacol30:21–24, 1992
Jacobs SS, Fox E, Dennie C, Morgan LB, McCully CL, Balis FM, Plasma and cerebrospinal fluid pharmacokinetics of intravenous oxaliplatin, cisplatin, and carboplatin in nonhuman primatesClin Cancer Res11:1669–1674, 2005
Wolff JEA, Trilling T, Molenkamp G, Egleler RM, Jurgens H, Chemosensitivity of glioma cells in vitro: a meta analysisJ Cancer Res Clin Oncol125:481–486, 1999
Poisson M, Pereon Y, Chiras J, Delattre JY, Treatment of recurrent malignant supratentorial gliomas with carboplatin J Neurooncol10:139–144, 1992
Yung WKA, Mechtler L, Gleason MJ, Intravenous carboplatin for recurrent malignant glioma: a phase II study J Clin Oncol9:860–864, 1991
Warnick RE, Prados MD, Mack EE, Chandler KL, Doz F, Rabbitt JE, Malec MK, A phase II study of intravenous carboplatin for the treatment of recurrent gliomasJ Neurooncol19:69–74, 1994
Robertson JF, 2004. Selective oestrogen receptor modulators/new antioestrogens: a clinical perspectiveCancer Treat Rev30(8):695–706
Couldwell WT, Uhm J, Antel JP, Yong VW, Enhanced protein kinase C activity correlates with the growth rate of malignant human gliomasNeurosurgery29:880–887, 1991
Couldwell WT, Antel JP, Apuzzo MLJ, Yong VW, Inhibition of growth of established human glioma lines by modulators of the protein kinase C second messenger systemJ Neurosurg73:594–600, 1990
Couldwell WT, Antel JP, Yong VW, Protein kinase C activity and glioma growth (letter)J Neurosurg74:686–687, 1991
Couldwell WT, Antel JP, Yong VW, Enhanced protein kinase C activity correlates with the growth rate of malignant gliomas: Part II. Effects of glioma mitogens and modulators of PKCNeurosurgery31:717–724, 1992
Pollack IF, Randall MS, Kristofik MP, Kelly RH, Selker RG, Vertosik FT, Effect of tamoxifen on DNA synthesis and proliferation of human malignant glioma lines in vitroCancer Res50:7134–7138, 1990
O’Brian CA, Liskamp RM, Solomon DH, Weinstein IB, Inhibition of protein kinase C by tamoxifenCancer Res45:2462–2465, 1985
O’Brian CA, Liskamp RM, Solomon DH, Weinstein IB, Triphenylethylenes: a new class of protein kinase C inhibitorsJ Natl Cancer Inst76:1243–1246, 1986
Zhang W, Yamada H, Sakai N, Niikawa S, Nozawa Y, Enhancement of radiosensitivity by tamoxifen in C6 glioma cellsNeurosurgery31:725–730, 1992
Vertosick FT, Selker RG, Pollack IF, Arena V, The treatment of intracranial malignant gliomas using orally administered tamoxifen therapy: preliminary results in a series of “failed” patientsNeurosurgery30:897–903, 1992
Chamberlain MC, Kormanik PA, Salvage chemotherapy with tamoxifen for recurrent anaplastic astrocytomasArch Neurol56:703–708, 1999
Couldwell WT, Hinton DR, Surnock AA, DeGiorgio C, Weiner LP, Apuzzo ML, Masri L, Law RE, Weiss MH, Treatment of recurrent malignant gliomas with chronic oral high-dose tamoxifenClin Cancer Res2:619–622, 1996
McClay EF, Albright KB, Jones JA, Eastman A, Christen R, Howell SB, Modulation of cisplatin resistance in human malignant melanoma cellsCancer Res52:6790–6796, 1992
McClay EF, Albright KB, Jones JA, Christen RD, Howell SB, Tamoxifen modulation of cisplatin sensitivity in human malignant melanoma cellsCancer Res53:1571–1576, 1993
Pollack IF, Kawecki BS, Lazo JS, Blocking of glioma proliferation in vitro and in vivo and potentiating the effects of BCNU and cisplatin: UCN-01, a selective protein kinase C inhibitorJ Neurosurg84(6):1024–1032, 1996
Mastronardi L, Farah JO, Puzzilli F, Ruggeri A, Tamoxifen modulation of carboplatin cytotoxicity in human U-138 glioma cell lineClin Neurol Neurosurg100:89–93, 1998
Basu A, Lazo JS, Sensitization of human cervical carcinoma cells to cis-diamminedichloroplatinum (II) by bryostatin 1Cancer Res52:3119–3124, 1992
Endicott JA, Ling V, The biochemistry of P-gIycoprotein-mediated multidrug resistanceAnnu Rev Biochem58:137–174, 1989
Mastronardi L, Puzzilli F, Couldwell WT, Farah JO, Lunardi P, Tamoxifen and carboplatin combination treatment of high-grade gliomas. Results of a clinical trial on newly diagnosed patientsJ Neurooncl38(1):59–68, 1998
Puchner MJA, Herrmann H, Berger J, Cristante L, Surgery, tamoxifen, carboplatin, and radiotherapy in the treatment of newly diagnosed glioblastoma patientsJ Neurooncol49:147–55, 2000
Macdonald DR, Cascino TL, Schold SC, Cairncross JG, Response criteria for phase II studies of malignant gliomaJ Clin Oncol8:1277–1280, 1990
Fleming T, One-sample multiple testing procedure for phase II clinical trialsBiometrics38:143–151 1982
Yung WKA, Prados MD, Yaya-Tur R, Rosenfeld SS, Brada M, Friedman HS, Albright R, Olson J, Chang SM, O’Neill AM, Friedman AH, Bruner J, Yue N, Dugan M, Zaknoen S, Levin VA, Multicenter Phase II Trial of Temozolomide in Patients With Anaplastic Astrocytoma or Anaplastic Oligoastrocytoma at First RelapseJ Clin Oncol17: 2762–2771, 1999
Yung WKA, Albright RE, Olson J, Fredericks R, Fink K, Prados MD, Brada M, Spence A, Hohl RJ, Shapiro W, Glantz M, Greenberg H, Selker RG, Vick NA, Rampling R, Friedman H, Phillips P, Bruner J, Yue N, Osoba D, Zaknoen S, Levin VA, A phase II study of temozolomide vs procarbazine in patients with glioblastoma multiforme at first relapseBr J Cancer83:588–593, 2000
Fulton D, Urtasun R, Forsyth P, Phase II study of prolonged oral therapy with etoposide (VP16) for patients with recurrent malignant gliomaJ Neurooncol27(2):149–155, 1996
Brandes AA, Tosoni A, Basso U, Reni M, Valduga F, Monfardini S, Amista P, Nicolardi I, Sotti G. Ermani M, Second-line chemotherapy with irinotecan plus carmustine in glioblastoma recurrent or progressive after first-line temozolomide chemotherapy: a Phase II study of the Gruppo Italiano Cooperativo di Neuro-Oncologia (GICNO)J Clin Oncol22(23): 4779–4786, 2004
Kappelle AC, Postma TJ, Taphorn MJ, Groeneveld GJ, van den Bent MJ, van Groeningen CJ, Zonnenberg BA, Sneeuw KC, Heimans JJ, PCV chemotherapy for recurrent glioblastoma multiformeNeurology56:118–120, 2001
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tang, P., Roldan, G., Brasher, P. et al. A phase II study of carboplatin and chronic high-dose tamoxifen in patients with recurrent malignant glioma. J Neurooncol 78, 311–316 (2006). https://doi.org/10.1007/s11060-005-9104-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-005-9104-y