Abstract
Seed orchards play a critical role in reforestation, but often struggle to consistently produce enough seed. A mixture of the less polar gibberellins, GA4 and GA7, has been shown to promote flowering in Pinaceae species, but the responses vary significantly based on species, genotype and provenance, application method, timing and dosage. Previous studies with lodgepole pine (Pinus contorta Dougl. var. latifolia Engelm.) suggest that the response to exogenous GA4/7 varied among genotypes. Gibberellins also play a critical role in seed development and germination but it remains unclear how exogenous GA4/7 will affect seed quality. We used 25 lodgepole pine genotypes in a commercial seed orchard in Alberta, Canada to explore the impact of genotype and geographic origin on the efficacy and optimal timing of GA4/7 stem injections to promote female cone bud initiation, cones and seed production. Overall, we found that GA4/7 stem injections promoted female conelets in 40% of genotypes included in this study. For these 10 genotypes, we found that the latitude of their provenance was positively correlated with later effective application times. Of the remaining 15 genotypes, five were hypersensitive to exogenous GA4/7 showing needle discolouration and loss, and 10 genotypes showed no response. In general, stem-injected GA4/7 did not affect the number of seeds per cone while two genotypes showed a reduced seed yield and one genotype showed increased seed yield per cone. Seed weights were not affected by the stem-injected GA4/7, while the July application date resulted in higher germination rates compared to control trees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seed orchards play a critical role in supplying high-quality seed for reforestation, but often struggle to consistently produce enough seed (Giertych 1987; Eriksson et al. 1998). Natural fluctuations, pests and diseases, as well as insufficient pollination, unfavourable environmental conditions and high ovule and conelet abortion rates can result in low numbers of mature cones, few seeds per cone and ultimately low seed yields (White et al. 1977; Yates and Ebel 1978; Katovich et al. 1989; Webber 1995; Bates et al. 2002; Owens et al. 2005; Owens and Fernando 2007). Research conducted in the late 1970s and 80s showed that in many conifers, cone bud initiation can be promoted through the application of exogenous gibberellins (GA) and other plant growth regulators (Ross and Pharis 1976; Pharis et al. 1980, 1987; Owens and Blake 1985; Bonnet-Masimbert 1987). A mixture of the less polar gibberellins, GA4 and GA7, has been shown to promote flowering and thereby enhance conelet production in Pinaceae species, but the responses can vary significantly based on species, genotype and provenance, as well as application method, timing and dosage (Eriksson et al. 1998; Zhao et al. 2011).
Wheeler et al. (1980) found that exogenous GA4/7, applied as a spray or droplets, can be used to enhance female conelet production in lodgepole pine (Pinus contorta Dougl. var. latifolia Engelm.); however, the response to exogenous GA4/7 appeared to differ among genotypes. Likewise, in a recent study with only two genotypes in a lodgepole pine seed orchard, GA4/7 stem injections promoted female cone production in one of the genotypes, while the other genotype showed no response (Kong et al. 2018). Lodgepole pine is known to display high genetic diversity among populations along with a strong adaptation to local climate (Rehfeldt et al. 1999; Liepe et al. 2016). It is reasonable to assume that trees from different geographic origins will display differences in phenology, such as the timing of bud-break in the spring, when grown together in a seed orchard. It is therefore reasonable to consider that the optimal timing of any GA4/7 treatment will vary among seed orchard trees based on the geographic origin of the parent tree.
Gibberellins are also known to play a critical regulatory role in seed development and germination and GA’s are often found in high concentrations in developing seeds (Singh et al. 2002; Hedden and Thomas 2012). It is currently unclear how the application of exogenous GA to promote cone production will affect seed development and germination rates in lodgepole pine.
In the present study we set out to explore the relationship between genotype, geographic origin of the parent and the efficacy and optimal timing of GA4/7 stem injections to promote female conelet development, cone and seed production. To address these questions, we selected a total of 25 genotypes in an under-producing commercial lodgepole pine seed orchard in Alberta, Canada. We then subjected individual ramets belonging to these genotypes to one of four GA4/7 application treatments (June, July, August, none). Specifically we wanted to test the following hypotheses: (1) GA4/7 stem injections will increase the number of female conelets/cones in all genotypes in a lodgepole pine seed orchard; (2) the optimal time for GA4/7 stem injections will vary among genotypes based on the geographic origin of the parent trees from which scions for grafting in the orchard originated from; and (3) GA4/7 stem injections will cause no difference in (a) the number of seeds per cone, (b) the 1000-seed weights, or (c) the germination rates.
Materials and methods
Study design
This study was conducted in a high elevation clonal lodgepole pine seed orchard, located approximately 40 km west of Grande Prairie, Alberta, Canada (55.066545°N, − 119.294858°W, 690 m.a.s.l.). Establishment of the orchard began in 1994 by planting trees developed from scion material collected from 34 parent genotypes which were grafted onto 3-year-old seedling rootstock for a total of 174 trees. Additional genotypes were added to the orchard until 2013 for a total of 110 genotypes and 1550 trees. Since going into production in 2006, the orchard has been unable to reach the industry target of 5.5 million seeds per year.
In summer 2015, we selected 25 of the 110 genotypes present in the orchard based on a minimum age (12 years) and number of ramets (12). For each of these genotypes, we randomly assigned three ramets each to one of three GA4/7 application times (June, July and August) with the remaining three ramets serving as a control (i.e. no GA4/7).
For each application time, a new batch of the GA4/7 mixture was prepared in the lab by mixing 17.1 g of GA4/7 with 213.75 ml of 95% ethanol, resulting in a GA4/7 concentration of 80 mg ml−1. The GA component of the mixture was composed of GA9 (0.87%), GA7 (36.88%) and GA4 (62.25%) (Zhejiang Shenghua Biok Biology Co. LTD, Deqing Zhejiang, China).
The mixture was then injected into holes drilled into the stem. The 6.35 mm wide holes were drilled just above the graft union at a 45° angle to a depth of approximately 2 cm and below the first whorl of branches. All holes were immediately sealed with grafting wax (Green Earth Pruning Sealer) to prevent infection. The number of holes, and with it the administered GA4/7 dosage, varied based on stem diameter above the graft union (see Table 1). The treatment concentration of the GA4/7 however, did not vary. Following the stem-injections, all treated trees were monitored visually for any sign of negative effects caused by our treatment. Findings from this monitoring effort were summarized in an internal report (Bhardwaj and Thomas 2016).
Tracking cone development
In order to track the impact of the GA4/7 treatments on the number of female conelets, we marked three randomly selected branches in the middle and upper part of the crown on each of the selected 300 trial trees. We then tracked the developing conelets on these marked branches, counting them at the beginning (June) and end (late August) of the 2016 and 2017 growing seasons, covering the entire 26 month reproductive cycle of lodgepole pine (Owens 2006). At the end of each growing season (2015–2017), all cones present on our trial trees were harvested to obtain a total cone count per tree. After each cone collection, five cones were selected randomly from each ramet and seeds were extracted in 2015, 2016 and 2017.
Seed extraction and testing
Immediately after each operational cone harvest (2015–2017), we collected a random sample of five cones from each trial tree. In some cases, the sample size was smaller because the total number of cones per tree was less than five. The cone samples were stored in labelled brown paper bags and transported at room temperature back to the University of Alberta where they were spread out on the lab bench to await further processing. Prior to extracting the seeds, all samples were weighed to determine the average weight per cone. In order to extract the seeds from these serotinous cones, samples were initially submerged into an 80 °C water bath (Grant, SUB Aqua Pro, Grant Instruments Ltd.) for 1 min to dissolve the resin and then immediately transferred into labelled sealed aluminum trays and placed into a drying oven at 60 °C for 8 h to facilitate the opening of cone scales. Seeds were then extracted by hand with the help of a dissecting probe and de-winged using tweezers. The extracted seeds were subsequently weighed to determine the 1000-seed weight and scanned on a flatbed scanner (EPSON Perfection v800) using the image analysis program WinSeedle (Regent Instruments Inc., QC, Canada) to determine the number, size, shape and colour of extracted seeds. Seeds were then pooled for each genotype and treatment and stored in a fridge at 4 °C. In July 2018, we randomly selected 100 seeds from each of these composite samples, from the 2017 harvest, for germination testing. In accordance with the current seed testing standard for Alberta (Government of Alberta Agriculture and Forestry Division 2016), seeds were placed on moist tissue paper in labelled petri dishes and placed in an incubator set at 25 °C with a 12 h day/night cycle for 21 days. Petri dishes were monitored every other day and germination counts were conducted on day 7, 14 and 21. Fully germinated seeds (i.e. those with a radicle at least four times the length of the seed itself) were removed after each count.
Data analysis
We assessed the effect of GA4/7 stem injections on female conelet and cone numbers, seed counts per cone and 1000-seed weights using a 2-way ANOVA with GA treatment and genotype as fixed factors. The effect on germination rates was assessed using one-way ANOVA with GA treatment as a fixed factor. All analyses were executed using R (R Core Team 2013).We used Fisher’s LSD test to conduct pairwise comparisons of least-square means using the lsmeans and cld commands in the lsmeans package (Lenth 2016).
In order to assess the relationship between parent geographic origin and the effect of GA4/7 application timing, we assigned a ranking factor (effective timing) to all genotypes that showed a positive response to the GA4/7 injection based on the respective month(s) during which the treatments were applied [i.e. effective timing (month): June = 6, June/July = 6.5, July = 7, July/August = 7.5, August = 8]. We used this ‘effective timing’ factor in combination with the geographic origin (latitude, longitude, elevation) of the parents to build a predictive model using multiple regression. The final model was determined by stepwise selection based on the Akaike Information Criterion (AIC) using the stepAIC command in the MASS package (Venables and Ripley 2002).
Results
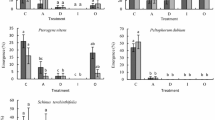
The initial (i.e. spring 2016) conelet production response to the summer 2015 GA4/7 stem injections varied significantly among genotypes (Table 2). Overall, 15 of 25 genotypes showed no significant increase in female conelet numbers for any of the three application times when compared to the control trees (Fig. 1). Among these 15 genotypes, five (648, 661, 677, 767, 770) appeared to be hypersensitive to the GA4/7 stem injections (genotype × treatment, p < 0.001) resulting in varying degrees of needle discoloration and loss (Fig. 2). In all five cases, hypersensitivity occurred irrespective of the time of GA4/7 application. For the 10 remaining genotypes that showed an increase in female conelet production in response to the exogenous GA4/7, the effective application times represented the full range of treatments (i.e. June–August) (Table 3).
Stepwise multiple regression using the 10 responsive genotypes, ranked by their effective application times, and associated geographic origins (latitude, longitude and elevation) (Table 4), revealed a significant positive correlation between effective application time and northerly latitude of the parent trees (R2= 0.48; p = 0.027) (Fig. 3).
Conelet abortion rates among the cohort of female conelets initiated in 2015 were high, with an overall mean of 72 ± 24% (± SD) over the course of the experiment. The GA4/7 treatments had no effect on abortion rates. There were, however, significant differences among genotypes (p < 0.001), with conelet abortion rates ranging from 36.6 ± 18% (genotype 664) to 99.9 ± 0.2% (genotype 668) (Fig. 4).
The September 2017 harvest of all cones initiated in 2015, showed that GA4/7 stem injections in June and July, but not August, significantly increased the total number of cones per tree compared to the control trees (p < 0.001) (Fig. 5a). Total cone production also differed significantly among genotypes (p < 0.001) (Fig. 5b). As was expected, the GA4/7 treatments had no effect on total cone production per tree in the two previous years of collection (2015, initiated in 2013 and 2016, initiated in 2014).
The number of seeds per cone showed a significant treatment × genotype interaction (p < 0.001) in 2017. For 21 of the 25 genotypes, the number of seeds per cone did not differ significantly among GA4/7 treatments, averaging 20.6 ± 8.7 seeds per cone. For two genotypes (662, 775) however, the August GA4/7 stem injections resulted in a lower number of seeds per cone (10.3 ± 9.3, 12.1 ± 12.4) (± SD) compared to the control trees (27.1 ± 3.5, 31.7 ± 8.8), while another genotype (1035), responded with lower numbers of seeds per cone when GA4/7 was injected in June (21.6 ± 14.3) when compared to control trees for this genotype (38.0 ± 17.9). Only one genotype (765), showed an increase in the number of seeds per cone in response to one of the GA4/7 treatments, with stem injections in July resulting in 32.9 ± 14.3 seeds per cone, compared to 19.1 ± 7.7 seeds per cone for control trees of the same genotype. For comparison, each cone contained on average 29.0 ± 10.5 seeds in 2015 and 29.9 ± 10.5 in 2016, irrespective of which GA4/7 treatment trees had received.
In 2017, we collected a sample of five cones from each trial tree and extracted all viable seeds. Our analysis of seed weights showed no differences in the 1000-seed weights among the GA4/7 treatments (p = 0.12), while there were strong differences among genotypes (p < 0.001) (Fig. 6).
Seeds from control trees and seeds from trees that received their GA4/7 in August showed significantly higher germination rates, than seeds from trees that received either the June or July application (p < 0.001) on day 7 of the germination trial (Table 5). After 14 days, seeds from trees that received their GA4/7 in July showed higher germination rates (46%) than all other treatments (31%). At the end of the trial (day 21) the July application still resulted in a higher germination rate compared to the control trees, as well as those that received GA4/7 in June.
Discussion
We hypothesized that at least one of our GA4/7 stem injection treatments would increase the number of female conelets, and therefore cones for all genotypes included in our study. Instead, we found that only 40% of the sample population responded with increased female conelet production (spring 2016) to at least one of our GA4/7 treatments at the given dosage. Moreover, we found that all three application times (June, July and August) were effective in promoting female conelet production, although which timing proved effective varied strongly among genotypes.
Among the 60% of genotypes that did not respond positively to any of the GA4/7 stem injection treatments, one-third appeared to be hypersensitive to the dosage of exogenous GA applied. These genotypes showed varying degrees of needle loss and discoloration within a few weeks after the stem injections. It may still be possible to promote flowering in these genotypes, however, finding an appropriate dosage for such genotypes may not be feasible as it risks significant damage to valuable orchard trees. Instead, it may be advisable to test all genotypes for hypersensitivity to exogenous GA4/7 prior to operational stem injections and to simply exclude those showing a negative reaction. A simple and inexpensive test consists of spraying a GA solution onto an individual branch early in the spring and observing the needles for several weeks (Kong and von Aderkas, personal communication) to determine which genotypes may be hypersensitive. Conversely, the remaining two-thirds of ‘non-responsive’ genotypes may need a higher dose of exogenous GA to enhance female conelet production. Further testing would be required to identify if there is an effective dosage for these genotypes.
Our findings thus showed that the initial conelet induction response to GA4/7 stem injections in lodgepole pine was genotype specific (clonal). Kong et al. (2018) likewise found clonal differences in the efficacy of GA4/7 stem injections among two lodgepole pine genotypes. At present, it remains unclear what causes the clonal differences in conelet induction response to the injection of exogenous GA4/7. However, it has been suggested that clonal differences in endogenous hormone levels, as well as the ability to metabolize exogenous GA4/7 may play a critical role (Kong et al. 2018).
Contrary to our expectations, the results from the 2017 cone harvest did not match the findings from the initial conelet numbers observed in spring 2016. Rather than showing a strong genotypic response, the total cone counts suggested a main treatment effect, with GA4/7 stem injections in June and July leading to an overall higher number of cones per tree compared to the control trees. The apparent contradiction between these two findings may be rooted in differences in the underlying methodology (i.e. 3 branches vs. whole tree) and the high level of conelet abortion among certain genotypes. In our opinion, the initial response in female conelet induction is a more accurate representation of the actual effect of the GA4/7 treatments, as the initial number of female conelets was not influenced by the high rates of conelet abortion.
We also hypothesized that the optimal timings for GA4/7 stem injections will vary among genotypes based on the geographic origin of the parent tree from which scion material was collected. Our findings appear to support this hypothesis, as we found a relationship between optimal timing of GA4/7 stem injections, leading to an increase in conelet production and the geographic origin, specifically, latitude of the parent trees, where more northerly origins were positively correlated with the later application of GA4/7 stem injections.
Lodgepole pine is one of the most wide-ranging and genetically diverse conifer species in western North America, displaying a high degree of differentiation and local adaptation among and even within populations (Rehfeldt et al. 1999; Wu and Ying 2004; Liepe et al. 2016).
Several studies have highlighted the role of climatic differences in driving genetic diversity among lodgepole pine populations (Chuine et al. 2001; Wang et al. 2010; Liepe et al. 2016).
The formation of reproductive organs represents a significant carbon cost and as a consequence plants adapt to their local climate in order to avoid losing reproductive organs during unfavourable weather conditions such as late frosts or during drought stress (Wheeler et al. 1980).
Genotypes from more northerly geographic origins are likely adapted to a cooler climate with slower spring warming and an increased risk of late frosts and may therefore show a delay in their reproductive cycle when compared to more southerly provenances (Chuine et al. 2001, 2006; Howe et al. 2003).
In order to promote the development of female conelets, the application of exogenous GA4/7 has to occur at the proper stage of cone bud initiation in late spring to early summer. Ho and Hak (1994) found that early (May–July) application of GA4/7 led to higher seed cone numbers in small potted lodgepole pine seedlings, whereas late (July–August) application resulted in increased pollen cone numbers.
The exact timing of these developmental stages, however, may vary based on both genotype and geographic origin, in particular the northern latitude, of the parent trees (O’Reilly and Owens 1987, 1988). Kong et al. (2016) found that exogenous GA4/7 applied to developing buds of lodgepole pine genotypes in a seed orchard in late spring was metabolized quickly with concentrations dropping drastically 2–4 weeks after application.
Given that the intent of treating commercial orchard trees with exogenous GA4/7 is typically to increase seed production, it would be counterproductive if the treatments negatively affected the number of seeds per cone, seed weights or germination rates. In our study, we were able to show that in a relatively small fraction of genotypes (12.5%) GA4/7 stem injections did in fact cause changes in the number of seeds per cone. In most cases at least one of the application times caused a reduction in seeds per cone when compared to the control trees. In one genotype however, one of the treatments resulted in a higher number of seeds per cone.
In our study, none of the GA4/7 treatments caused any change in the 1000-seed weight. In contrast, Eriksson et al. (1998) found a reduction in 1000-seed weights when studying the effects of GA4/7 stem injections on seed and pollen production in a Scots pine (Pinus sylvestris L.) seed orchard in Sweden. Any reduction in seed size is undesirable as larger seeds are typically associated with better viability and increased seedling vigour (Wahid and Bounoua 2013).
Although we did not find any differences in seed weights, we did find that the GA4/7 treatments had an effect on germination rates, with the July application resulting in a higher germination rate than seeds from control trees after 21 days. Gibberellins are known to play a role in seed germination and GA treatments have been used to promote germination when seed availability may be severely limited or when seeds have been stored under less than ideal conditions (Singh et al. 2002; Hedden and Thomas 2012; Vishal and Kumar 2018). Further research would be needed to determine if this positive effect carries over into increased seedling vigour after outplanting.
In summary, our study showed that GA4/7 stem injections can be used to stimulate female conelet production in lodgepole pine, however, the response to such a treatment is complex and strongly influenced by genotype and parent origin.
References
Bates SL, Strong WB, Borden JH (2002) Abortion and seed set in lodgepole and western white pine conelets following feeding by Leptoglossus occidentalis (Heteroptera: Coreidae). Environ Entomol 31:1023–1029. https://doi.org/10.1603/0046-225X-31.6.1023
Bhardwaj Y, Thomas BR (2016) B2 orchard 2015 damage assessment report. Internal report to the Huallen Seed Orchard Company
Bonnet-Masimbert M (1987) Floral induction in conifers: a review of available techniques. For Ecol Manage 19:135–146. https://doi.org/10.1016/0378-1127(87)90019-3
Chuine I, Aitken SN, Ying CC (2001) Temperature thresholds of shoot elongation in provenances of Pinus contorta. Can J For Res 31:1444–1455. https://doi.org/10.1139/cjfr-31-8-1444
Chuine I, Rehfeldt GE, Aitken SN (2006) Height growth determinants and adaptation to temperature in pines: a case study of Pinus contorta and Pinus monticola. Can J For Res 36:1059–1066. https://doi.org/10.1139/x06-005
Eriksson U, Jansson G, Almqvist C (1998) Seed and pollen production after stem injections of gibberellin A 4/7 in field-grown seed orchards of Pinus sylvestris. Can J For Res 28:340–346. https://doi.org/10.1139/x97-219
Giertych M (1987) Seed orchards in crisis. For Ecol Manage 19:1–7. https://doi.org/10.1016/0378-1127(87)90005-3
Government of Alberta, Agriculture and Forestry Division (2016) Alberta seed testing standards
Hedden P, Thomas SG (2012) Gibberellin biosynthesis and its regulation. Biochem J 444:11–25. https://doi.org/10.1042/BJ20120245
Ho RH, Hak O (1994) Optimum timing of gibberellin A4/7 sprays to promote cone production in jack pine seedlings. New For 8:61–69. https://doi.org/10.1007/BF00034131
Howe GT, Aitken SN, Neale DB et al (2003) From genotype to phenotype: unraveling the complexities of cold adaptation in forest trees. Can J Bot 81:1247–1266. https://doi.org/10.1139/b03-141
Katovich SA, Overton RP, Rush PA, Kulman HM (1989) Red-pine conelet, cone and seed losses to insects and other factors in an open-grown plantation and a seed orchard. For Ecol Manage 29:115–131. https://doi.org/10.1016/0378-1127(89)90060-1
Kong L, von Aderkas P, Irina Zaharia L (2016) Effects of exogenously applied gibberellins and thidiazuron on phytohormone profiles of long-shoot buds and cone gender determination in lodgepole pine. J Plant Growth Regul 35:172–182. https://doi.org/10.1007/s00344-015-9517-6
Kong L, von Aderkas P, Zaharia LI (2018) Effects of stem-injected gibberellins and 6-benzylaminopurine on phytohormone profiles and cone yield in two lodgepole pine genotypes. Trees 32:765. https://doi.org/10.1007/s00468-018-1670-7
Lenth RV (2016) Least-squares means: The R Package lsmeans. J Stat Softw. https://doi.org/10.18637/jss.v069.i01
Liepe KJ, Hamann A, Smets P et al (2016) Adaptation of lodgepole pine and interior spruce to climate: Implications for reforestation in a warming world. Evol Appl 9:409–419. https://doi.org/10.1111/eva.12345
O’Reilly C, Owens JN (1987) Long-shoot bud development, shoot growth, and foliage production in provenances of lodgepole pine. Can J For Res 17:1421–1433. https://doi.org/10.1139/x87-220
O’Reilly C, Owens JN (1988) Reproductive growth and development in seven provenances of lodgepole pine. Can J For Res 18:43–53. https://doi.org/10.1139/x88-008
Owens JN (2006) The reproductive biology of lodgepole pine. Extension Note 07, Forest Genetics Council of British Columbia, Victoria, British Columbia, Canada
Owens JN, Blake MD (1985) Forest tree seed production. A review of the literature and recommendations for future research. Petawawa National Forestry Institute, Canadian Forestry Service, Information Report, PI-X-53, 161 p
Owens JN, Fernando DD (2007) Pollination and seed production in western white pine. Can J For Res 37:260–275. https://doi.org/10.1139/x06-220
Owens JN, Bennett J, L’Hirondelle S (2005) Pollination and cone morphology affect cone and seed production in lodgepole pine seed orchards. Can J For Res 35:383–400. https://doi.org/10.1139/x04-176
R Core Team (2013) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna
Pharis RP, Ross SD, McMullan E (1980) Promotion of flowering in the Pinaceae by gibberellins. III. Seedlings of Douglas fir. Physiol Plant 50:119–126. https://doi.org/10.1111/j.1399-3054.1980.tb04437.x
Pharis RP, Webber JE, Ross SD (1987) The promotion of flowering in forest trees by gibberellin and cultural treatments: a review of the possible mechanisms. For Ecol Manage 19:65–84. https://doi.org/10.1016/0378-1127(87)90012-0
Rehfeldt GE, Ying CC, Spittlehouse DL, Hamilton DA (1999) Genetic responses to climate in Pinus contorta: niche breadth, climate change, and reforestation. Ecol Monogr 69:375. https://doi.org/10.2307/2657162
Ross SD, Pharis RP (1976) Promotion of flowering in the pinaceae by Gibberellins. I. Sexually mature, non-flowering grafts of Douglas-Fir. Physiol Plant 36:182–186. https://doi.org/10.1111/j.1399-3054.1976.tb03932.x
Singh D, Jermakow A, Swain S (2002) Gibberellins are required for seed development and pollen tube growth in Arabidopsis. Plant Cell 14:3133–3147. https://doi.org/10.1105/tpc.003046.Arabidopsis
Venables WN, Ripley BD (2002) Modern applied statistics with S. Springer, New York
Vishal B, Kumar PP (2018) Regulation of seed germination and abiotic stresses by gibberellins and abscisic acid. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00838
Wahid N, Bounoua L (2013) The relationship between seed weight, germination and biochemical reserves of maritime pine (Pinus pinaster Ait.) in Morocco. New For 44:385–397. https://doi.org/10.1007/s11056-012-9348-2
Wang T, O’Neill GA, Aitken SN (2010) Integrating environmental and genetic effects to predict responses of tree populations to climate. Ecol Appl 20:153–163. https://doi.org/10.1890/08-2257.1
Webber JE (1995) Pollen management for intensive seed orchard production. Tree Physiol 15:507–514
Wheeler NC, Wample RL, Pharis RP (1980) Promotion of flowering in the Pinaceae by gibberellins. IV. Seedlings and sexually mature grafts of lodgepole pine. Physiol Plant 50:340–346. https://doi.org/10.1111/j.1399-3054.1980.tb04111.x
White TL, Harris HG Jr, Kellison RC (1977) Conelet abortion in longleaf pine. Can J For Res 7:378–382. https://doi.org/10.1139/x77-047
Wu HX, Ying CC (2004) Geographic pattern of local optimality in natural populations of lodgepole pine. For Ecol Manage 194:177–198. https://doi.org/10.1016/j.foreco.2004.02.017
Yates HO, Ebel BH (1978) Impact of insect damage on loblolly pine seed production. J Econ Entomol 71:345–349. https://doi.org/10.1093/jee/71.2.345
Zhao P, Fan J, Zhang S et al (2011) Effects of gibberellin A4/7, 6-benzylaminopurine and chlormequat chloride on the number of male and female strobili and immature cones in Chinese pine (Pinus tabuliformis) with foliar sprays. J For Res 22:353–359. https://doi.org/10.1007/s11676-011-0179-3
Acknowledgements
We thank Dr. Pharis for his help in preparing the GA solution, Dave Kamelchuk for conducting the initial tree selections with B. Thomas, stem injections and initial damage assessments, the staff at the Huallen Seed Orchard Company for their support with field work. We also thank Arial Eatherton, Aaron Chen, Michael Thomson, Esteban Galeano and Aileen Sturges for their support in the field. Funding for this manuscript has been provided through the Industrial Research Chair in Tree Improvement held by Dr. B.R. Thomas supported by the Natural Sciences Engineering Research Council (NSERC), Alberta-Pacific Forest Industries Inc., Alberta Newsprint Company Timber Ltd., Canadian Forest Products Ltd., Millar Western Forest Products Ltd., Huallen Seed Orchard Company Ltd., West Fraser Mills Ltd. (including: Alberta Plywood, Blue Ridge Lumber Inc., Hinton Wood Products, Sundre Forest Products Inc.), and Weyerhaeuser Company Ltd., (Pembina and Grande Prairie Timberlands). Additional funding was also provided by the Government of Alberta.
Author information
Authors and Affiliations
Contributions
BT, SB: study conception and design. SB, BT: acquisition of data. SB, BT: analysis and interpretation of data. SB: drafting of manuscript. BT, SB: critical revision. BT, SB: final approval of the version to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Bockstette, S.W., Thomas, B.R. Impact of genotype and parent origin on the efficacy and optimal timing of GA4/7 stem injections in a lodgepole pine seed orchard. New Forests 51, 421–434 (2020). https://doi.org/10.1007/s11056-019-09733-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-019-09733-w