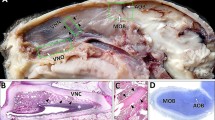
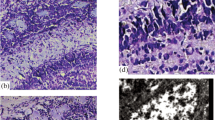
An immunohistochemical study of the olfactory bulb (OB) in human fetuses was performed. Immunohistochemical markers for nervous system-specific protein complexes S-100 and SNAP-25 were used. At 20–22 weeks of development, the development of cells expressing protein S-100 was greater in the OB than in the neocortex (the areas of the gyrus rectus and the gyrus orbitalis longitudinalis internus). These quantitative differences indicate heterochronicity in glial differentiation in the OB and cerebral cortex. Immunopositive reactions for SNAP-25 were for the first time detected at the periphery of the OB and in glomeruli in human fetuses at 15–16 and 20–22 weeks of development. Immunohistochemical staining of the OB with antibodies to SNAP-25 indicated that the human olfactory system cannot function prior to 20–22 weeks of development. In the OB of fetuses at 28–29 weeks of development, the intensity of the immunohistochemical reaction showed topological differences. The reaction with antibodies to SNAP-25 in the OB of full-term fetuses was similar to the reaction in the OB of adult humans. The results of these immunohistochemical studies using the reaction for the protein complex SNAP-25 in fetuses of different ages suggest the that the primary olfactory center in humans starts to function no earlier than the 30th week of development.
Similar content being viewed by others
References
S. V. Savel’ev, Atlas of the Human Brain [in Russian], VEDI (2005).
A. Becher, A. Drenckhahn, I. Pahner, et al., “The synaptophysin–synaptobrevin complex: a hallmark of synaptic vesicle maturation,” J. Neurosci., 19, No. 6, 1922–1931 (1999).
J. Bossy, “Development of olfactory and related structures in staged human embryos,” Anat. Embryol., 161, 225–236 (1980).
M. I. Chuah and D. R. Zheng, “Olfactory marker protein is present in olfactory receptor cells of human fetuses,” Neurosci., 23, 263–270 (1987).
M. I. Chuah and D. R. Zheng, “The human primary olfactory pathway: fine structural and cytochemical aspects during development and in adults,” Microsc. Res. Tech., 23, No. 1, 76–85 (1992).
R. Donato, “Intracellular and extracellular roles of S100 proteins,” Microsc. Res. Tech., 60, No. 6, 540–541 (2003).
T. Humphrey, “The development of the olfactory and accessory olfactory formations in human embryos and fetuses,” J. Comp. Neurol., 73, 431–468 (1940).
F. Muller and R. O’Rahilly, “Olfactory structures in staged human embryos,” Cells Tissues Organs, 178, 93–116 (2004).
A. A. Pearson, “The development of the olfactory nerve in man,” J. Comp. Neurol., 75, 199–217 (1941).
A. A. Pearson, “The development of the olfactory nerve, the nervus terminalis, and the vomeronasal nerve in man,” Ann. Otol. Rhinol. Laryngol., 51, No. 2, 317–332 (1942).
I. Salasar, P. S. Quintiero, M. Lombardero, et al., “The prenatal maturity of the accessory olfactory bulb in pigs,” Senses, 29, No. 1, 3–11 (2004).
T. Sollner, M. K. Bennett, S. W. Whiteheart, et al., “A protein assembly-disassembly pathway in vitro that may correspond to sequential steps of synaptic vesicle docking, activation, and fusion,” Cell, 75, No. 3, 409–418 (1993).
S. Takahashi, T. Iwanaga,Y. Takahashi, et al., “Neuron-specific enolase, neurofilament protein and S-100 protein in the olfactory mucosa of human fetuses. An immunohistochemical study,” Cell Tissue Res., 238, No. 2, 231–234 (1984).
N. Tateishi, T. Shimoda, N. Yada, et al., “S-100: astrocyte specific protein,” Nihon Shinkei Seishin Yakurigaku Zasshi, 26, No. 1, 11–16 (2006).
Author information
Authors and Affiliations
Additional information
Translated from Morfologiya, Vol. 135, No. 1, pp. 20–24, January–February, 2009.
Rights and permissions
About this article
Cite this article
Kharlamova, A.S., Barabanov, V.M. & Savel’ev, S.V. Development of the Olfactory Bulbs in Human Fetuses (an immunohistochemical study). Neurosci Behav Physi 40, 131–135 (2010). https://doi.org/10.1007/s11055-009-9248-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-009-9248-x