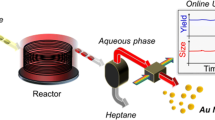
Abstract
Iron oxide nanoparticles have been of great interest due to their physical and chemical properties, and the coprecipitation method is among the main ways to produce them. Despite being the cheapest and most straightforward method, its main disadvantage during the scale-up process is the increase in the nanoparticle’s polydispersity and the reactor’s dimensions. This way, looking for an alternative to increasing the production scale in the synthesis of Fe3O4 by coprecipitation, we developed and optimized a low-cost flow reactor based on peristaltic pumps made of inexpensive DC motors and an Arduino microcontroller. A 24–1 experimental design was performed to optimize the Fe3O4 coprecipitation parameters such as reagent concentration, flow rate, and reactor size. The flow reactor parameters were verified to affect the size, homogeneity, and stability of Fe3O4 nanoparticles with sizes ranging from 5 to 10 nm and polydispersity index varying between 0.15 and 0.45 Furthermore, the product was synthesized in a matter of minutes with rates up to 150 mL min−1 in a low-cost reactor.






Similar content being viewed by others
References
de Melo FM, Grasseschi D, Brandão BBNS et al (2018) Superparamagnetic maghemite-based cdte quantum dots as efficient hybrid nanoprobes for water-bath magnetic particle inspection. ACS Appl Nano Mater 1:2858–2868. https://doi.org/10.1021/acsanm.8b00502
Asimakidou T, Makridis A, Veintemillas-Verdaguer S et al (2020) Continuous production of magnetic iron oxide nanocrystals by oxidative precipitation. Chem Eng J 393:124593. https://doi.org/10.1016/j.cej.2020.124593
Carvalho A, Domingues I, Gonçalves MC (2015) Core-shell superparamagnetic nanoparticles with interesting properties as contrast agents for MRI. Mater Chem Phys 168:42–49. https://doi.org/10.1016/j.matchemphys.2015.10.030
Cornell RM, Schwertmann U (2003) The iron oxides; structure, reaction, occurences and uses, second edition, John Wiley & Sons, pp 139-183. https://doi.org/10.1002/3527602097
de Melo F, Almeida S, Toma H (2020) Magnetic nanohydrometallurgy applied to lanthanide separation. Minerals 10:530. https://doi.org/10.3390/min10060530
Gonzalez-Moragas L, Yu S-M, Murillo-Cremaes N et al (2015) Scale-up synthesis of iron oxide nanoparticles by microwave-assisted thermal decomposition. Chem Eng J 281:87–95. https://doi.org/10.1016/j.cej.2015.06.066
Hafermann L, Michael Köhler J (2015) Small gold nanoparticles formed by rapid photochemical flow-through synthesis using microfluid segment technique. J Nanoparticle Res 17:99. https://doi.org/10.1007/s11051-015-2914-8
Haseidl F, Müller B, Hinrichsen O (2016) Continuous-flow synthesis and functionalization of magnetite: intensified process for tailored nanoparticles. Chem Eng Technol 39:2051–2058. https://doi.org/10.1002/ceat.201600163
Hong Y, Xue ZL, Terepka AD (2004) Synthesis of silver nanoparticles in a continuous flow tubular microreactor. Nano Lett 4:2227–2232. https://doi.org/10.1021/nl0485859
Hui C, Shen C, Yang T et al (2008) Large-scale Fe 3 O 4 nanoparticles soluble in water synthesized by a facile method. J Phys Chem C 112:11336–11339. https://doi.org/10.1021/jp801632p
James M, Revia RA, Stephen Z, Zhang M (2020) Microfluidic Synthesis of Iron Oxide Nanoparticles. Nanomaterials 10:2113. https://doi.org/10.3390/nano10112113
Jolivet J-P, Chanéac C, Tronc E (2004) Iron oxide chemistry. From molecular clusters to extended solid networks. Chem Commun 477–483. https://doi.org/10.1039/B304532N
Jongen N, Donnet M, Bowen P et al (2003) Development of a continuous segmented flow tubular reactor and the “scale-out” concept – in search of perfect powders. Chem Eng Technol 26:303–305. https://doi.org/10.1002/ceat.200390046
Jozwiak WK, Kaczmarek E, Maniecki TP et al (2007) Reduction behavior of iron oxides in hydrogen and carbon monoxide atmospheres. Appl Catal A Gen 326:17–27. https://doi.org/10.1016/j.apcata.2007.03.021
Kim W, Suh CY, Cho SW et al (2012) A new method for the identification and quantification of magnetite-maghemite mixture using conventional X-ray diffraction technique. Talanta 94:348–352. https://doi.org/10.1016/j.talanta.2012.03.001
Kumar A, Dixit CK (2017) Methods for characterization of nanoparticles. Adv Nanomedicine Deliv Ther Nucleic Acids 44–58. https://doi.org/10.1016/B978-0-08-100557-6.00003-1
Langford JI, Wilson AJC (1978) Scherrer after sixty years: a survey and some new results in the determination of crystallite size. J Appl Crystallogr 11:102–113. https://doi.org/10.1107/S0021889878012844
Majidi S, Zeinali Sehrig F, Samiei M et al (2016) Magnetic nanoparticles: applications in gene delivery and gene therapy. Artif Cells, Nanomedicine Biotechnol 44:1186–1193. https://doi.org/10.3109/21691401.2015.1014093
Marquez-Medina MD, Prinsen P, Li H et al (2018) Continuous-flow synthesis of supported magnetic iron oxide nanoparticles for efficient isoeugenol conversion into vanillin. Chemsuschem 11:389–396. https://doi.org/10.1002/cssc.201701884
Menegatti de Melo F, Mattioni JV, Dias F et al (2021) Solvophobic-controlled synthesis of smart magneto-fluorescent nanostructures for real-time inspection of metallic fractures. Nanoscale Adv 3:3593–3604. https://doi.org/10.1039/D1NA00149C
Montgomery DC (2006) Design and analysis of experiments. Technometrics 48:158–158. https://doi.org/10.1198/tech.2006.s372
Neto BR, Scarminio IS, Bruns ER (2001) Como fazer experimentos: Pesquisa e desenvolvimento na ciência e na indústria, 3th Ed., Bookman, Campinas, Sp, ISBN: 8577806529.
Panariello L, Mazzei L, Gavriilidis A (2018) Modelling the synthesis of nanoparticles in continuous microreactors: the role of diffusion and residence time distribution on nanoparticle characteristics. Chem Eng J 350:1144–1154. https://doi.org/10.1016/j.cej.2018.03.167
Panariello L, Damilos S, du Toit H et al (2020) Highly reproducible, high-yield flow synthesis of gold nanoparticles based on a rational reactor design exploiting the reduction of passivated Au( <scp>iii</scp> ). React Chem Eng 5:663–676. https://doi.org/10.1039/C9RE00469F
Park J, Joo J, Kwon SG et al (2007) Synthesis of monodisperse spherical nanocrystals. Angew Chem Int Ed Engl 46:4630–4660. https://doi.org/10.1002/anie.200603148
Pereira ML de O, Maia KCB, Silva WC et al (2020) Fe 3 O 4 nanoparticles as surfactant carriers for enhanced oil recovery and scale prevention. ACS Appl Nano Mater 3:5762–5772. https://doi.org/10.1021/acsanm.0c00939
Răcuciu M, Creangă DE, Airinei A (2006) Citric-acid-coated magnetite nanoparticles for biological applications. Eur Phys J E 21:117–121. https://doi.org/10.1140/epje/i2006-10051-y
Revia RA, Zhang M (2016) Magnetite nanoparticles for cancer diagnosis, treatment, and treatment monitoring: recent advances. Mater Today 19:157–168. https://doi.org/10.1016/j.mattod.2015.08.022
Sahoo Y, Goodarzi A, Swihart MT et al (2005) Aqueous ferrofluid of magnetite nanoparticles: fluorescence labeling and magnetophoretic control. J Phys Chem B 109:3879–3885. https://doi.org/10.1021/jp045402y
Salazar-Alvarez G, Muhammed M, Zagorodni AA (2006) Novel flow injection synthesis of iron oxide nanoparticles with narrow size distribution. Chem Eng Sci 61:4625–4633. https://doi.org/10.1016/j.ces.2006.02.032
Shen Z, Wu A, Chen X (2017) Iron oxide nanoparticle based contrast agents for magnetic resonance imaging. Mol Pharm 14:1352–1364. https://doi.org/10.1021/acs.molpharmaceut.6b00839
Thanh NTK, Maclean N, Mahiddine S (2014) Mechanisms of nucleation and growth of nanoparticles in solution. Chem Rev 114:7610–7630. https://doi.org/10.1021/cr400544s
Valério A, Morelhão SL (2019) Usage of Scherrer’s formula in X-ray diffraction analysis of size distribution in systems of monocrystalline nanoparticles. https://doi.org/10.48550/arXiv.1911.00701
Veiseh O, Gunn JW, Zhang M (2010) Design and fabrication of magnetic nanoparticles for targeted drug delivery and imaging. Adv Drug Deliv Rev 62:284–304. https://doi.org/10.1016/j.addr.2009.11.002
Wu W, He Q, Jiang C (2008) Magnetic iron oxide nanoparticles: synthesis and surface functionalization strategies. Nanoscale Res Lett 3:397–415. https://doi.org/10.1007/s11671-008-9174-9
Funding
This work received funding from CAPES, CNPq, FAPERJ (E-26/211.281/2019, E-26/010.002267/2019), and Instituto Serrapilheira (R-2012–37959).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare are no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 9741 kb)
Rights and permissions
About this article
Cite this article
Perissé Moreira, M., Grasseschi, D. Automation of a low-cost device for flow synthesis of iron oxide nanoparticles. J Nanopart Res 24, 93 (2022). https://doi.org/10.1007/s11051-022-05476-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-022-05476-6