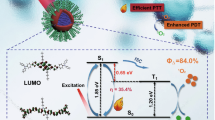
Abstract
In this experiment, we successfully developed nanocarriers in the form of gold nanoflowers coated with two layers of silica for the purposes of drug loading and NIR (near-infrared) photothermal therapy for the treatment of oral cancer. The gold nanoflowers converted NIR laser energy into heat energy. The cores were coated with a thin silica layer (AuNFs@SiO2) to protect the gold nanoflowers from intraparticle ripening. The second layer was mesoporous silica (AuNFs@SiO2@mSiO2), which acted as a nanocarrier for anticancer drug (DOX) loads. The mean effective diameter of the nanoparticles was approximately 150–200 nm, whereas the peak absorption of the AuNFs was 684 nm. After the AuNFs were encapsulated by the silica shells, the plasmonic absorption peak of AuNFs@SiO2 and AuNFs@SiO2@mSiO2 exhibited a red shift to 718 nm. When exposed to an 808 nm NIR laser, these crystals showed an obvious photothermal conversion in the NIR region and a highly efficient release of DOX. Biocompatibility was assessed in vitro using Cell Counting Kit-8 assays, and the results showed that the nanocarriers induced no obvious cytotoxicity. This nanomaterial could be considered a new type of material that shows promising potential for photothermal-chemotherapy against malignant tumours, including those of oral cancers.







Similar content being viewed by others
References
Al-Ogaidi I, Gou H, Al-Kazaz AKA et al (2014) A gold@ silica core–shell nanoparticle-based surface-enhanced Raman scattering biosensor for label-free glucose detection. Anal Chim Acta 811:76–80
An N, Rausch-fan X, Wieland M et al (2012) Initial attachment, subsequent cell proliferation/viability and gene expression of epithelial cells related to attachment and wound healing in response to different titanium surfaces. Dent Mater 28(12):1207–1214
Barbosa S, Agrawal A, Rodríguez-Lorenzo L et al (2010) Tuning size and sensing properties in colloidal gold nanostars. J Langmuir 26(18):14943–14950
Chen LB, Zhang F, Wang CC (2009) Rational synthesis of magnetic thermosensitive microcontainers as targeting drug carriers. Small 5(5):621–628
Dolmans DE, Fukumura D, Jain RK (2003) Photodynamic therapy for cancer. Nat Rev Cancer 3(5):380–387
Dong B, Xu S, Sun J et al (2011) Multifunctional NaYF4: Yb 3 + , Er 3 + @ Ag core/shell nanocomposites: integration of upconversion imaging and photothermal therapy. J Mater Chem 21(17):6193–6200
Fales AM, Yuan H, Vo-Dinh T (2011) Silica-coated gold nanostars for combined surface-enhanced Raman scattering (SERS) detection and singlet-oxygen generation: a potential nanoplatform for theranostics. Langmuir 27(19):12186–12190
Feng J, Wang Z, Shen B et al (2014) Effects of template removal on both morphology of mesoporous silica-coated gold nanorod and its biomedical application. RSC Adv 4(54):28683–28690
Fomina N, Sankaranarayanan J, Almutairi A (2012) Photochemical mechanisms of light-triggered release from nanocarriers. Adv Drug Deliv Rev 64(11):1005–1020
Gabaldon JP, Bore M, Datye AK (2007) Mesoporous silica supports for improved thermal stability in supported Au catalysts. Top Catal 44(1–2):253–262
Guo Y, Zhang Z, Kim DH et al (2013) Photothermal ablation of pancreatic cancer cells with hybrid ironoxide core gold-shell nanoparticles. Int J Nano 8: 3437
Haiss W, Thanh NTK, Aveyard J et al (2007) Determination of size and concentration of gold nanoparticles from UV–vis spectra. Anal Chem 79(11):4215–4221
Huang X, El-Sayed IH, Qian W et al (2006) Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J Am Chem Soc 128(6):2115–2120
Jiang Y, Ahn EY, Ryu SH et al (2004) Cytotoxicity of psammaplin A from a two-sponge association may correlate with the inhibition of DNA replication. BMC Cancer 4(1):70
Jiang Z, Dong B, Chen B et al (2013) Multifunctional Au@ mSiO2/rhodamine B isothiocyanate nanocomposites: cell imaging, photocontrolled drug release, and photothermal therapy for cancer cells. Small 9(4):604–612
Kwon S, Singh RK, Kim TH et al (2014) Luminescent mesoporous nanoreservoirs for the effective loading and intracellular delivery of therapeutic drugs. Acta Biomater 10(3):1431–1442
Lai CY, Trewyn BG, Jeftinija DM et al (2003) A mesoporous silica nanosphere-based carrier system with chemically removable CdS nanoparticle caps for stimuli-responsive controlled release of neurotransmitters and drug molecules. J Am Chem Soc 125(15):4451–4459
Levi-Polyachenko NH, Merkel EJ, Jones BT et al (2009) Rapid photothermal intracellular drug delivery using multiwalled carbon nanotubes. Mol Pharm 6(4):1092–1099
Pattani VP, Tunnell JW (2012) Nanoparticle-mediated photothermal therapy: a comparative study of heating for different particle types. Lasers Surg Med 44(8):675–684
Popova MD, Szegedi Á, Kolev IN et al (2012) Carboxylic modified spherical mesoporous silicas as drug delivery carriers. Int J Pharm 436(1):778–785
Stassi S, Cauda V, Canavese G et al (2012) Synthesis and characterization of gold nanostars as filler of tunneling conductive polymer composites. Eur J Inorg Chem 16:2669–2673
Stöber W, Fink A, Bohn E (1968) Controlled growth of monodisperse silica spheres in the micron size range. J Colloid Interface Sci 26(1):62–69
Storm FK, Harrison WH, Elliott RS et al (1979) Normal tissue and solid tumor effects of hyperthermia in animal models and clinical trials. Cancer Research 39(6 Part 2): 2245–2251
Van de Broek B, Devoogdt N, D’Hollander A et al (2011) Specific cell targeting with nanobody conjugated branched gold nanoparticles for photothermal therapy. ACS Nano 5(6):4319–4328
van der Zee J (2002) Heating the patient: a promising approach? Annal oncol 13(8):1173–1184
Wang J, Dong B, Chen B et al (2012) Selective photothermal therapy for breast cancer with targeting peptide modified gold nanorods. Dalton Trans 41(36):11134–11144
Wang J, Dong B, Chen B et al (2013) Glutathione modified gold nanorods with excellent biocompatibility and weak protein adsorption, targeting imaging and therapy toward tumor cells. Dalton Trans 42(32):11548–11558
Yagüe C, Arruebo M, Santamaria J (2010) NIR-enhanced drug release from porous Au/SiO 2 nanoparticles. Chem Commun 46(40):7513–7515
Yang J, Zhang F, Chen Y et al (2011) Core-shell Ag@ SiO2@ mSiO2 mesoporous nanocarriers for metal-enhanced fluorescence. Chem Commun 47(42):11618–11620
Zhao L, Ji X, Sun X et al (2009) Formation and stability of gold nanoflowers by the seeding approach: the effect of intraparticle ripening. J Phys Chem C 113(38):16645–16651
Acknowledgments
This work was supported by Grants from the Natural Science Foundation of China (Grant No: 81372900) and the Science and Technology Development Plan of Jilin Province of China (Grant No: 20110708 and 201401010 55JC). The authors are grateful to the Key Laboratory of Surface and Interface Chemistry of Jilin Province, College of Chemistry, Ms. Xiaohui Ji, Ms. Lili Zhao and Ms. Zhenzhen Huang for their assistance in preparing the nanocarrier materials.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Song, W., Gong, J., Wang, Y. et al. Gold nanoflowers with mesoporous silica as “nanocarriers” for drug release and photothermal therapy in the treatment of oral cancer using near-infrared (NIR) laser light. J Nanopart Res 18, 101 (2016). https://doi.org/10.1007/s11051-016-3377-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-016-3377-2