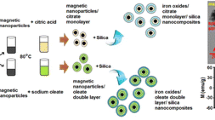
Abstract
The value of the magnetization has a strong influence on the performance of nanoparticles that act as the contrast agent material for MRI. In this article, we describe processing routes for the synthesis of FePt nanoparticles of different sizes, which, as a result, exhibit different magnetization values. “Single-core” FePt nanoparticles of different sizes (3–15 nm) were prepared via one-step or two-step synthesis, with the latter exhibiting twice the magnetization (m (1.5T) = 14.5 emu/g) of the nanoparticles formed via the one-step synthesis (m (1.5T) < 8 emu/g). Furthermore, we propose the synthesis of “multi-core” FePt nanoparticles by changing the ratio between the two surfactants (oleylamine and oleic acid). The step from smaller “single-core” FePt nanoparticles towards the larger, “multi-core” FePt nanoparticles (>20 nm) leads to an increase in the magnetization m (1.5T) from 8 to 19.5 emu/g, without exceeding the superparamagnetic limit. Stable water suspensions were prepared using two different approaches: (a) functionalization with a biocompatible, zwitterionic, catechol ligand, which was used on the FePt nanoparticles for the first time, and (b) coating with SiO2 shells of various thicknesses. These FePt-based nanostructures, the catechol- and SiO2-coated “single-core” and “multi-core” FePt nanoparticles, were investigated in terms of the relaxation rates. The higher r 2 values obtained for the “multi-core” FePt nanoparticles compared to that for the “single-core” ones indicate the superiority of the “multi-core” FePt nanoparticles as T 2 contrast agents. Furthermore, it was shown that the SiO2 coating reduces the r 1 and r 2 relaxation values for both the “single-core” and “multi-core” FePt nanoparticles. The high r 2/r 1 ratios obtained in our study put FePt nanoparticles near the top of the list of candidate materials for use in MRI.
Graphical Abstract









Similar content being viewed by others
References
Ai H, Flask C, Weinberg B et al (2005) Magnetite-loaded polymeric micelles as ultrasensitive magnetic-resonance probes. Adv Mater 17:1949–1952. doi:10.1002/adma.200401904
Aslam M, Fu L, Li S, Dravid VP (2005) Silica encapsulation and magnetic properties of FePt nanoparticles. J Colloid Interface Sci 290:444–449. doi:10.1016/j.jcis.2005.04.050
Bagaria HG, Ada ET, Shamsuzzoha M et al (2006) Understanding mercapto ligand exchange on the surface of FePt nanoparticles. Langmuir 22:7732–7737. doi:10.1021/la0601399
Balasubramaniam S, Kayandan S, Lin Y-N et al (2014) Toward design of magnetic nanoparticle clusters stabilized by biocompatible diblock copolymers for T2-weighted MRI contrast. Langmuir 30:1580–1587. doi:10.1021/la403591z
Barisik M, Atalay S, Beskok A, Qian S (2014) Size dependent surface charge properties of silica nanoparticles. J Phys Chem C 118:1836–1842. doi:10.1021/jp410536n
Berret J-F, Schonbeck N, Gazeau F et al (2006) Controlled clustering of superparamagnetic nanoparticles using block copolymers: design of new contrast agents for magnetic resonance imaging. J Am Chem Soc 128:1755–1761. doi:10.1021/ja0562999
Bin Na H, Song IC, Hyeon T (2009) Inorganic nanoparticles for MRI contrast agents. Adv Mater 21:2133–2148. doi:10.1002/adma.200802366
Bulte JWM, Kraitchman DL (2004) Iron oxide MR contrast agents for molecular and cellular imaging. NMR Biomed 17:484–499
Caravan P, Ellison JJ, McMurry TJ, Lauffer RB (1999) Gadolinium(III) chelates as MRI contrast agents: structure, dynamics, and applications. Chem Rev 99:2293–2352
Chen S, Andre P (2012) Colloidal syntheses of FePt nanoparticles. Int J Nanotechnol 9:39. doi:10.1504/IJNT.2012.044829
Chen H, Yeh J, Wang L et al (2010a) Preparation and control of the formation of single core and clustered nanoparticles for biomedical applications using a versatile amphiphilic diblock copolymer. Nano Res 3:852–862. doi:10.1007/s12274-010-0056-y
Chen S, Wang L, Duce SL et al (2010b) Engineered biocompatible nanoparticles for in vivo imaging applications. J Am Chem Soc 132:15022–15029. doi:10.1021/ja106543j
Chou SW, Zhu CL, Neeleshwar S et al (2009) Controlled growth and magnetic property of FePt nanostructure: cuboctahedron, octapod, truncated cube, and cube. Chem Mater 21:4955–4961. doi:10.1021/cm902199p
Chou SW, Shau YH, Wu PC et al (2010) In vitro and in vivo studies of FePt nanoparticles for dual modal CT/MRI molecular imaging. J Am Chem Soc 132:13270–13278. doi:10.1021/ja1035013
Dai ZR, Sun S, Wang ZL (2001) Phase transformation, coalescence, and twinning of monodisperse FePt nanocrystals. Nano Lett 1:443–447. doi:10.1021/nl0100421
Elsabahy M, Wooley KL (2012) Design of polymeric nanoparticles for biomedical delivery applications. Chem Soc Rev 41:2545. doi:10.1039/c2cs15327k
Green LA, Thuy TT, Mott DM et al (2014) Multicore magnetic FePt nanoparticles: controlled formation and properties. RSC Adv 4:1039. doi:10.1039/c3ra44319a
Hirani B, Li J, Djurovich PI et al (2007) Cyclometallated iridium and platinum complexes with noninnocent ligands. Inorg Chem 46:3865–3875. doi:10.1021/ic061556b
Huang G, Li H, Chen J et al (2014) Tunable T1 and T2 contrast abilities of manganese-engineered iron oxide nanoparticles through size control. Nanoscale 6:10404–10412. doi:10.1039/c4nr02680b
Kim DK, Kan D, Veres T et al (2005) Monodispersed Fe-Pt nanoparticles for biomedical applications. J Appl Phys 97:50–52. doi:10.1063/1.1860851
Kostevšek N, Žužek Rožman K, Arshad MS et al (2015) Multimodal hybrid FePt/SiO 2/Au nanoparticles for nanomedical applications: combining photothermal stimulation and manipulation with an external magnetic field. J Phys Chem C 119:16374–16382. doi:10.1021/acs.jpcc.5b03725
Lee DC, Mikulec FV, Pelaez JM et al (2006a) Synthesis and magnetic properties of silica-coated FePt nanocrystals. J Phys Chem B 110:11160–11166. doi:10.1021/jp060974z
Lee J-H, Jun Y-W, Yeon S-I et al (2006b) Dual-mode nanoparticle probes for high-performance magnetic resonance and fluorescence imaging of neuroblastoma. Angew Chem Int Ed Engl 45:8160–8162. doi:10.1002/anie.200603052
Liang S, Zhou Q, Wang M et al (2015) Water-soluble l-cysteine-coated FePt nanoparticles as dual MRI/CT imaging contrast agent for glioma. Int J Nanomedicine 10:2325–2333. doi:10.2147/IJN.S75174
Lu J, Liong M, Li Z et al (2010) Biocompatibility, biodistribution, and drug-delivery efficiency of mesoporous silica nanoparticles for cancer therapy in animals. Small 6:1794–1805. doi:10.1002/smll.201000538
Maenosono S, Saita S (2006) Theoretical assessment of FePt nanoparticles as heating elements for magnetic hyperthermia. IEEE Trans Magn 42:1638–1642. doi:10.1109/TMAG.2006.872198
Maenosono S, Suzuki T, Saita S (2008) Superparamagnetic FePt nanoparticles as excellent MRI contrast agents. J Magn Magn Mater 320:79–83. doi:10.1016/j.jmmm.2008.01.026
Nandwana V, Nandwana V, Elkins KE et al (2007) Size and shape control of monodisperse FePt nanoparticles. J Phys Chem C 111:4185–4189. doi:10.1021/jp068330e
Saita S, Maenosono S (2005) Formation mechanism of FePt nanoparticles synthesized via pyrolysis of Iron(III) ethoxide and platinum(II) acetylacetonate. Chem Mater 17:6624–6634. doi:10.1021/cm051760h
Shevchenko EV, Talapin DV, Schnablegger H et al (2003) Study of nucleation and growth in the organometallic synthesis of magnetic alloy nanocrystals: the role of nucleation rate in size control of CoPt3 nanocrystals. J Am Chem Soc 125:9090–9101. doi:10.1021/ja029937l
Shin T, Choi Y, Kim S, Cheon J (2015) Recent advances in magnetic nanoparticle-based multi-modal imaging. Chem Soc Rev 44:4501–4516. doi:10.1039/C4CS00345D
Slichter CP (1990) Principles of magnetic resonance. Springer, Berlin
Stanford CJ, Dagenais M, Park J-H, DeShong P (2008) Real-time monitoring of siloxane monolayer film formation on silica using a fiber Bragg grating. Curr Anal Chem 4:356–361
Sun S (2000) Monodisperse FePt nanoparticles and ferromagnetic FePt nanocrystal superlattices. Science 287:1989–1992. doi:10.1126/science.287.5460.1989
Tanaka Y, Saita S, Maenosono S (2008) Influence of surface ligands on saturation magnetization of FePt nanoparticles. Appl Phys Lett 92:47–50. doi:10.1063/1.2891083
Tanaka K, Narita A, Kitamura N et al (2010) Preparation for highly sensitive MRI contrast agents using core/shell type nanoparticles consisting of multiple SPIO cores with thin silica coating. Langmuir 26:11759–11762. doi:10.1021/la1015077
Taylor RM, Huber DL, Monson TC et al (2012) Structural and magnetic characterization of superparamagnetic iron platinum nanoparticle contrast agents for magnetic resonance imaging. J Vac Sci Technol B Nanotechnol Microelectron 30:2C101–2C1016
Thanh NTK (2012) Magnetic nanoparticles: from fabrication to clinical applications. CRC Press, London
Tong S, Hou S, Zheng Z et al (2010) Coating optimization of superparamagnetic iron oxide nanoparticles for high T2 relaxivity. Nano Lett 10:4607–4613. doi:10.1021/nl102623x
Verma A, Stellacci F (2010) Effect of surface properties on nanoparticle-cell interactions. Small 6:12–21
Wang C, Hou Y, Kim J, Sun S (2007) A general strategy for synthesizing FePt nanowires and nanorods. Angew Chem Int Ed 46:6333–6335
Wei H, Insin N, Lee J et al (2012) Compact zwitterion-coated iron oxide nanoparticles for biological applications. Nano Lett 12:22–25. doi:10.1021/nl202721q
Wu XW, Liu C, Li L et al (2004) Nonmagnetic shell in surfactant-coated FePt nanoparticles. J Appl Phys. 95:6810–6812
Xu C, Xu K, Gu H et al (2004) Dopamine as a robust anchor to immobilize functional molecules on the iron oxide shell of magnetic nanoparticles. J Am Chem Soc 126:9938–9939. doi:10.1021/ja0464802
Yang H, Zhang J, Tian Q et al (2010) One-pot synthesis of amphiphilic superparamagnetic FePt nanoparticles and magnetic resonance imaging in vitro. J Magn Magn Mater 322:973–977. doi:10.1016/j.jmmm.2009.11.039
Zeynali H, Sebt SA, Arabi H, Akbari H (2012) Size control of FePt nanoparticles produced by seed mediated growth process. J Clust Sci 23:1107–1117. doi:10.1007/s10876-012-0506-7
Acknowledgments
This work was supported by the Slovenian Research Agency (ARRS), Project J2-6760. This work was financially supported by Grant G.0618.11 N of the Fund for Scientific Research Flanders (FWO-V, www.fwo.be); the Agency for Innovation by Science and Technology in Flanders (IWT, www.iwt.be); and the KU Leuven (GOA, www.kuleuven.be). M. B. is grateful for the support from the IWT.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kostevšek, N., Šturm, S., Serša, I. et al. “Single-” and “multi-core” FePt nanoparticles: from controlled synthesis via zwitterionic and silica bio-functionalization to MRI applications. J Nanopart Res 17, 464 (2015). https://doi.org/10.1007/s11051-015-3278-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-015-3278-9