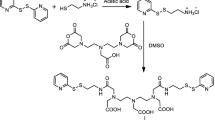
Abstract
Stable 30–50 nm polymeric polyethylene glycol–phosphatidylethanolamine (PEG–PE)-based micelles entrapping superparamagnetic iron oxide nanoparticles (SPION) have been prepared. At similar concentrations of SPION, the SPION-micelles had significantly better magnetic resonance imaging (MRI) T2 relaxation signal compared to ‘plain’ SPION. Freeze-fracture electron microscopy confirmed SPION entrapment in the lipid core of the PEG–PE micelles. To enhance the targeting capability of these micelles, their surface was modified with the cancer cell-specific anti-nucleosome monoclonal antibody 2C5 (mAb 2C5). Such mAb 2C5-SPION immunomicelles demonstrated specific binding with cancer cells in vitro and were able to bring more SPION to the cancer cells thus demonstrating the potential to be used as targeted MRI contrast agents for tumor imaging.






Similar content being viewed by others
References
Ai H, Flask C, Weinberg B, Shuai X, Pagel MD, Farell D, Duerk J, Gao J (2005) Magnetite-loaded polymeric micelles as novel magnetic resonance probes. Adv Mater 26:3995–4021
Alexiou C, Jurgons R, Schmid RJ, Bergemann C, Henke J, Erhardt W, Huenges E, Parak F (2003) Magnetic drug targeting-biodistribution of the magnetic carrier and the chemotherapeutic agent mitoxantrone after locoregional cancer treatment. J Drug Target 11:139–149. doi:10.1080/1061186031000150791
Corot C, Robert P, Idee JM, Port M (2006) Recent advances in iron oxide nanocrystal technology for medical imaging. Adv Drug Deliv Rev 58:1471–1504. doi:10.1016/j.addr.2006.09.013
Dandamudi S, Campbell RB (2007) Development and characterization of magnetic cationic liposomes for targeting tumor microvasculature. Biochim Biophys Acta 1768:427–438. doi:10.1016/j.bbamem.2006.10.006
Durand E, Raynaud JS, Bruneval P, Brigger I, Al Haj Zen A, Mandet C, Lancelot E, Lafont A (2007) Magnetic resonance imaging of ruptured plaques in the rabbit with ultrasmall superparamagnetic particles of iron oxide. J Vasc Res 44:119–128. doi:10.1159/000098484
Elbayoumi TA, Torchilin VP (2007) Enhanced cytotoxicity of monoclonal anticancer antibody 2C5-modified doxorubicin-loaded PEGylated liposomes against various tumor cell lines. Eur J Pharm Sci 32:159–168. doi:10.1016/j.ejps.2007.05.113
Elbayoumi TA, Torchilin VP (2008) Tumor-specific antibody-mediated targeted delivery of Doxil® reduces the manifestation of auricular erythema side effect in mice. Int J Pharm 357:272–279. doi:10.1016/j.ijpharm.2008.01.041
Elbayoumi TA, Pabba S, Roby A, Torchilin VP (2007) Antinucleosome antibody-modified liposomes and lipid-core micelles for tumor-targeted delivery of therapeutic and diagnostic agents. J Liposome Res 17:1–14. doi:10.1080/08982100601186474
Erdogan S, Medarova ZO, Roby A, Moore A, Torchilin VP (2008) Enhanced tumor MR imaging with gadolinium-loaded polychelating polymer-containing tumor-targeted liposomes. J Magn Reson Imaging 27:574–580. doi:10.1002/jmri.21202
Fortin JP, Wilhelm C, Servais J, Menager C, Bacri JC, Gazeau F (2007) Size-sorted anionic iron oxide nanomagnets as colloidal mediators for magnetic hyperthermia. J Am Chem Soc 129:2628–2635. doi:10.1021/ja067457e
Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26:3995–4021. doi:10.1016/j.biomaterials.2004.10.012
Gupta B, Levchenko TS, Mongayt DA, Torchilin VP (2005) Monoclonal antibody 2C5-mediated binding of liposomes to brain tumor cells in vitro and in subcutaneous tumor model in vivo. J Drug Target 13:337–343. doi:10.1080/10611860500286239
Hyeon T, Lee SS, Park J, Chung Y, Na HB (2001) Synthesis of highly crystalline and monodisperse maghemite nanocrystallites without a size-selection process. J Am Chem Soc 123:12798–12801. doi:10.1021/ja016812s
Iakoubov LZ, Torchilin VP (1997) A novel class of antitumor antibodies: nucleosome-restricted antinuclear autoantibodies (ANA) from healthy aged nonautoimmune mice. Oncol Res 9:439–446
Iakoubov LZ, Torchilin VP (1998) Nucleosome-releasing treatment makes surviving tumor cells better targets for nucleosome-specific anticancer antibodies. Cancer Detect Prev 22:470–475. doi:10.1046/j.1525-1500.1998.00055.x
Iakoubov L, Rokhlin O, Torchilin V (1995) Anti-nuclear autoantibodies of the aged reactive against the surface of tumor but not normal cells. Immunol Lett 47:147–149. doi:10.1016/0165-2478(95)00066-E
Ito A, Shinkai M, Honda H, Kobayashi T (2005) Medical application of functionalized magnetic nanoparticles. J Biosci Bioeng 100:1–11. doi:10.1263/jbb.100.1
Ito A, Honda H, Kobayashi T (2006) Cancer immunotherapy based on intracellular hyperthermia using magnetite nanoparticles: a novel concept of “heat-controlled necrosis” with heat shock protein expression. Cancer Immunol Immunother 55:320–328. doi:10.1007/s00262-005-0049-y
Jin H, Hong B, Kakar SS, Kang KA (2008) Tumor-specific nano-entities for optical detection and hyperthermic treatment of breast cancer. Adv Exp Med Biol 614:275–284. doi:10.1007/978-0-387-74911-2_31
Johannsen M, Thiesen B, Gneveckow U, Taymoorian K, Waldofner N, Scholz R, Deger S, Jung K, Loening SA, Jordan A (2006) Thermotherapy using magnetic nanoparticles combined with external radiation in an orthotopic rat model of prostate cancer. Prostate 66:97–104. doi:10.1002/pros.20324
Kang HW, Josephson L, Petrovsky A, Weissleder R, Bogdanov A Jr (2002) Magnetic resonance imaging of inducible E-selectin expression in human endothelial cell culture. Bioconjug Chem 13:122–127. doi:10.1021/bc0155521
Kubo T, Sugita T, Shimose S, Nitta Y, Ikuta Y, Murakami T (2001) Targeted systemic chemotherapy using magnetic liposomes with incorporated adriamycin for osteosarcoma in hamsters. Int J Oncol 18:121–125
Lukyanov AN, Elbayoumi TA, Chakilam AR, Torchilin VP (2004) Tumor-targeted liposomes: doxorubicin-loaded long-circulating liposomes modified with anti-cancer antibody. J Control Release 100:135–144. doi:10.1016/j.jconrel.2004.08.007
Martina MS, Fortin JP, Fournier L, Menager C, Gazeau F, Clement O, Lesieur S (2007) Magnetic targeting of rhodamine-labeled superparamagnetic liposomes to solid tumors: in vivo tracking by fibered confocal fluorescence microscopy. Mol Imaging 6:140–146
Mathieu JB, Martel S (2007) Magnetic microparticle steering within the constraints of an MRI system: proof of concept of a novel targeting approach. Biomed Microdevices 9:801–808. doi:10.1007/s10544-007-9092-0
Mulder WJ, Griffioen AW, Strijkers GJ, Cormode DP, Nicolay K, Fayad ZA (2007) Magnetic and fluorescent nanoparticles for multimodality imaging. Nanomedicine 2:307–324. doi:10.2217/17435889.2.3.307
Munshi N, De TK, Maitra A (1997) Size modulation of polymeric nanoparticles under controlled dynamics of microemulsion droplets. J Colloid Interface Sci 190:387–391. doi:10.1006/jcis.1997.4889
Nasongkla N, Bey E, Ren J, Ai H, Khemtong C, Guthi JS, Chin SF, Sherry AD, Boothman DA, Gao J (2006) Multifunctional polymeric micelles as cancer-targeted, MRI-ultrasensitive drug delivery systems. Nano Lett 6:2427–2430. doi:10.1021/nl061412u
Sawant RR, Sawant RM, Torchilin VP (2008) Mixed PEG–PE/vitamin E tumor-targeted immunomicelles as carriers for poorly soluble anti-cancer drugs: improved drug solubilization and enhanced in vitro cytotoxicity. Eur J Pharm Biopharm 70:51–57. doi:10.1016/j.ejpb.2008.04.016
Silva AK, Silva EL, Carrico AS, Egito ES (2007) Magnetic carriers: a promising device for targeting drugs into the human body. Curr Pharm Des 13:1179–1185. doi:10.2174/138161207780618993
Sun C, Veiseh O, Gunn J, Fang C, Hansen S, Lee D, Sze R, Ellenbogen RG, Olson J, Zhang M (2008) In vivo MRI detection of gliomas by chlorotoxin-conjugated superparamagnetic nanoprobes. Small 4:372–379. doi:10.1002/smll.200700784
Sykova E, Jendelova P (2007) In vivo tracking of stem cells in brain and spinal cord injury. Prog Brain Res 161:367–383. doi:10.1016/S0079-6123(06)61026-1
Tang T, Zheng JW, Chen B, Li H, Li X, Xue KY, Ai X, Zou SQ (2007) Effects of targeting magnetic drug nanoparticles on human cholangiocarcinoma xenografts in nude mice. Hepatobiliary Pancreat Dis Int 6:303–307
Torchilin VP (2004) Targeted polymeric micelles for delivery of poorly soluble drugs. Cell Mol Life Sci 61:2549–2559. doi:10.1007/s00018-004-4153-5
Torchilin VP, Levchenko TS, Lukyanov AN, Khaw BA, Klibanov AL, Rammohan R, Samokhin GP, Whiteman KR (2001) p-Nitrophenylcarbonyl-PEG–PE-liposomes: fast and simple attachment of specific ligands, including monoclonal antibodies, to distal ends of PEG chains via p-nitrophenylcarbonyl groups. Biochim Biophys Acta 1511:397–411. doi:10.1016/S0005-2728(01)00165-7
Torchilin VP, Lukyanov AN, Gao Z, Papahadjopoulos-Sternberg B (2003) Immunomicelles: targeted pharmaceutical carriers for poorly soluble drugs. Proc Natl Acad Sci USA 100:6039–6044. doi:10.1073/pnas.0931428100
Wada S, Tazawa K, Furuta I, Nagae H (2003) Antitumor effect of new local hyperthermia using dextran magnetite complex in hamster tongue carcinoma. Oral Dis 9:218–223. doi:10.1034/j.1601-0825.2003.02839.x
Yang F, de Fu L, Long J, Ni QX (2008) Magnetic lymphatic targeting drug delivery system using carbon nanotubes. Med Hypotheses 70:765–767. doi:10.1016/j.mehy.2007.07.045
Zhang C, Jugold M, Woenne EC, Lammers T, Morgenstern B, Mueller MM, Zentgraf H, Bock M, Eisenhut M, Semmler W, Kiessling F (2007) Specific targeting of tumor angiogenesis by RGD-conjugated ultrasmall superparamagnetic iron oxide particles using a clinical 1.5-T magnetic resonance scanner. Cancer Res 67:1555–1562. doi:10.1158/0008-5472.CAN-06-1668
Acknowledgment
This work was supported by the NIH grants RO1 EB01961 and RO1 EN002995 to Vladimir P. Torchilin and by Northeastern University and NSF grant DGE-0504331 to Srinivas Sridhar.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sawant, R.M., Sawant, R.R., Gultepe, E. et al. Nanosized cancer cell-targeted polymeric immunomicelles loaded with superparamagnetic iron oxide nanoparticles. J Nanopart Res 11, 1777–1785 (2009). https://doi.org/10.1007/s11051-009-9611-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9611-4