Abstract
Endemic systemic mycoses such as blastomycosis, coccidioidomycosis, histoplasmosis, talaromycosis, paracoccidioidomycosis are emerging as an important cause of morbidity and mortality worldwide. We conducted a systematic review on endemic systemic mycoses reported in Italy from 1914 to nowadays. We found out: 105 cases of histoplasmosis, 15 of paracoccidioidomycosis, 10 of coccidioidomycosis, 10 of blastomycosis and 3 of talaromycosis. Most cases have been reported in returning travelers and expatriates or immigrants. Thirtytwo patients did not have a story of traveling to an endemic area. Fortysix subjects had HIV/AIDS. Immunosuppression was the major risk factor for getting these infections and for severe outcomes. We provided an overview on microbiological characteristics and clinical management principles of systemic endemic mycoses with a focus on the cases reported in Italy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
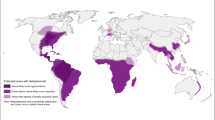
The most common endemic systemic mycoses are caused by thermally dimorphic fungi [1]. This group of fungi grows as a mold at 22–25 °C and as yeasts at 37 °C [2]. Among these diseases, blastomycosis, coccidioidomycosis, histoplasmosis, talaromycosis, paracoccidioidomycosis are emerging as an important cause of morbidity and mortality worldwide [3]. They have a peculiar geographical distribution in most cases. Migratory flows and travels allow their spread also in non-endemic countries [4]. They can affect both immunocompetent and immunocompromised people, particularly those with HIV/AIDS, where they manifest with more severe outcomes [5]. Recently the International Society for Human and Animal Mycology and the European Confederation of Medical Mycology published guidelines for the diagnosis and management of the endemic mycoses [3].
In Italy many case reports and case series have been published about endemic mycoses, mostly imported from endemic areas. Systematic reviews regarding Italian cases have been conducted only about histoplasmosis [4, 6,7,8]. A review about cases of paracoccidioidomycosis in Europe was conducted by Wagner et al. [9].
We conducted a systematic review on endemic systemic mycoses reported in Italy. A practical update of microbiological and clinical aspects is provided.
Materials and Methods
This systematic review was performed according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) [10].
Information sources were represented by three major databases, MEDLINE, CENTRAL and Embase, screened from inception until 1st April 2022 using a combination of keywords. The detailed search strategy is described in Appendix 1 (See supplementary materials).
Records were de-duplicated before entering the subsequent phase of the review. One investigator (VZ) carried out the first selection of the retrieved records by title and abstract in order to establish eligibility for full-text review. The second step consisted of further screening of full-text articles to define final inclusion in the systematic review according to the inclusion criteria. We included case reports, case series, systematic reviews about the following mycoses: histoplasmosis, coccidioidomycosis, talaromycosis, blastomycosis, and paracoccidioidomycosis. We included full texts written in English and Italian. Additional cases were sought from the reference list of included papers and reviews.
The following information was extracted from each article and entered into pilot-tested evidence tables: mycosis, author, year, number of cases, patients’ nationality, age, gender, comorbidities, immunocompromised status, country of exposure, clinical presentation and affected organs, isolated species, co-infections, diagnosis, antifungal therapy, and outcomes.
The authors confirm that the ethical policies of the journal, as noted on the journal’s author guidelines page, have been adhered to. No ethical approval was required as this is a review article with no original research data.
Results
Summary of the Literature
The literature search identified 88 articles about cases of histoplasmosis, coccidioidomycosis, talaromycosis, blastomycosis, and paracoccidioidomycosis reported in Italy (Fig. 1).
Talaromycosis
Talaromycosis, or penicilliosis, is a systemic fungal disease caused by Talaromyces marneffei (formerly Penicillium marneffei), a dimorphic fungus classified in the group of the Eurotiales order and Trichocomaceae family [11].
It is endemic in tropical and subtropical areas of South and Southeast Asia: in Northeastern India, Southern China, Hong Kong, Taiwan and Northern Thailand [11]. Talaromycosis especially affects immunocompromised patients, in particular those with HIV infection and a CD4 count < 100 cells/µL [12]. It has become the third most common HIV-related opportunistic infection in South and SouthEast Asia [12], where it is an AIDS-defining illness [11]. Travel-related talaromycosis is being increasingly reported also in non-endemic countries [11].
Human Infections occur through inhalation of T. marneffei conidia. The transmission is seasonal, coinciding with rainy seasons [11]. Bamboo rats are the main animal reservoir of T. marneffei [11]. There is no evidence of both animal-to-person or person-to-person transmission [12].
The incubation period of the disease is variable (from 1 to 3 weeks to years) [11]. Clinical manifestations appear only after hematogenous dissemination. The signs and symptoms are similar in adults and in children [13], but differ in patients with and without HIV. Patients without HIV are more likely to have bone and joint infections but are less likely to have fever, splenomegaly, skin lesions and positive fungal blood cultures [14]. Most patients present symptoms related to reticuloendothelial system involvement, including generalized lymphadenopathy, hepatomegaly, and splenomegaly. Talaromycosis can involve respiratory system (fever, dyspnea, and chest pain); gastrointestinal system (diarrhea); central nervous system (CNS) and cause mucosal or skin involvement (lesions appear as umbilicated papule, resembling molluscum contagiosum, nodules, or necrotic lesions, and they are usually located over the face and upper trunk) [15, 16].
The diagnosis is traditionally made through the identification of the fungus in clinical specimens by microscopy and culture [11]. Microscopically, T. marneffei appears as oval or round intracellular yeasts with cross-wall formations. Cultures of bone marrow, blood and biopsies of skin lesions have the highest sensitivity [12]. Talaromyces marneffei takes from 3 to 14 days to grow up in culture [11]. Talaromyces marneffei can be observed in histopathological sections with Grocott methenamine silver or periodic acid-Schiff stain. Infected tissues appear as granulomatous, suppurative reaction, and non-reactive necrosis [11]. No commercial serological tests are available. Antigens are available instead [3]. Polymerase chain reaction (PCR) based tests and next-generation sequencing (NGS) are useful tools for rapid diagnosis, even if hardly available. Galactomannan and β-D-glucan also can be helpful [11].
Disseminated disease is fatal if untreated. First choice treatment consists in liposomal amphotericin B (3–5 mg/kg per day) for 2 weeks, followed by oral itraconazole (200 mg/twice daily) for 10 weeks. If it is not possible to use amphotericin B, voriconazole or fluconazole (much less active) can be used. For immunocompromised patients itraconazole should be continued as secondary prophylaxis, until restoration of cellular immunity. For HIV patients with CD4 < 100 cells/µL living in endemic areas, primary prophylaxis with itraconazole 200 mg/day is indicated [3, 16].
Three cases of talaromycosis have been described in Italy (Table 1). All patients were immunocompromised (AIDS). Two patients were Italians who traveled to Thailand, the other one was a Chinese student living in Italy since 2016. All patients had disseminated infection, successfully treated with clinical recovery [17,18,19].
Blastomycosis
Blastomycosis is a systemic fungal disease caused by Blastomyces dermatitidis, a dimorphic fungus classified in the group of the Onygenales order and Ajellomycetaceae family [20]. Blastomyces gilchristii is another species identified in 2013, which causes similar disease in humans [21]. Recently another distinct species was identified in Canada: Blastomyces helicus [22].
Blastomycosis is endemic in the United States of America (USA) and Canada, particularly in the Mississippi and Ohio River valleys, in the Mid-western states and Canadian provinces that border the Great Lakes, and areas adjacent to the Saint Lawrence Seaway. B. dermatitidis is also present in Africa and India [23, 24]. No epidemiological data are available for Europe, where the disease has been reported only in travelers [25].
Blastomyces species are found in a specific ecological niche characterized by wet earth with animal droppings (especially dogs) and decaying vegetation [26, 27]. Infection in people occurs through inhalation of the conidia. Sometimes, the infection could be transmitted through direct skin inoculation (for trauma or insect bites). B. dermatitidis is usually not transmitted from person to person and not even from animal to person [26].
Approximately 50% of people exposed to blastomycosis develop symptoms, whereas the remaining 50% have asymptomatic or subclinical disease. Incubation period of blastomycosis ranges from 3 weeks to 3 months [28]. Pulmonary blastomycosis can present with acute pneumonia which mimic bacterial pneumonia or chronic pneumonia like tuberculosis and lung cancer [26]. Extrapulmonary dissemination (via hematogenous spread or direct inoculation) affects skin, bones, and male genitourinary system, in decreasing order of frequency. Skin lesions are frequently misdiagnosed as pyoderma gangrenosum or basal/squamous cell carcinoma. CNS involvement is uncommon in immunocompetent hosts; it may occur in HIV patients, either as meningitis or cranial abscesses [29].
The gold standard for diagnosis is the culture of clinical specimens. Growth on Sabouraud dextrose agar is very slow and could take up to four weeks. For this reason histopathological identification is very important. Yeast appears as 15 µm cells with thick, double-refractile walls and a single broad-based bud. Gomori methenamine silver or periodic–acid Schiff staining are usually used for tissue samples, calcofluor white or Papanicolaou stains are used for respiratory samples. Antigen testing (not commercially available as a kit) of the galactomannan component can be performed on urine, serum, bronchoalveolar lavage, and cerebrospinal fluid (CSF). Antigenuria has a sensitivity of 75–93% and specificity of 80% in patients with proved blastomycosis. Cross-reactivity is described for histoplasmosis, paracoccidioidomycosis, and talaromycosis [30]. Serum β-d-glucan is unreliable for diagnosis of blastomycosis because the yeast cell wall contains very little of this carbohydrate [31]. Serological tests (complement fixation or immunodiffusion) are available, but they have poor sensitivity and specificity. PCR based tests are useful (even if they are still not validated) [30].
Itraconazole is the first choice for treatment in all forms (600 mg/day for 3 days, then 200–400 mg/day for 6/12 months), except in severe or life-threatening diseases. In these cases, liposomal amphotericin B (3–5 mg/kg/day) is indicated until clinical improvement, followed by itraconazole 200 mg twice daily for 6–12 months [32]. CNS blastomycosis should be treated for at least 12 months and until CNS abnormalities resolution (liposomal amphotericin B 5 mg/kg/day for 4–6 weeks, followed by an oral azole) [33].
Ten cases of blastomycosis have been described in Italy (Table 1). All patients were immunocompetent. In all cases, the species identified was B. dermatitidis. Nevertheless as all but one of the cases were diagnosed before 2013, one cannot disregard misidentifications with the recently described species B. gilchristii and B. helicus. Only four subjects have a travel history in these countries, respectively: Ghana [25], Senegal [34], Central America [35] and Canada [36]. Only two patients developed pneumonia [36, 37], the other eight presented with extrapulmonary manifestations: bone, joints and soft tissues (n = 5) [25, 34, 36, 38], oral cavity (n = 3) [35, 39] and adrenal glands (n = 1) [40]. No deaths are reported. Seven patients recovered (for three patients this information is lacking).
Paracoccidioidomycosis
Paracoccidioidomycosis is a systemic fungal disease caused by Paracoccidioides spp, a dimorphic fungus classified in the group of the Onygenales order and Ajellomycetaceae family. Two species, P. brasiliensis sensu stricto and P. lutzii, cause paracoccidioidomycosis. Recently other species have been described: P. americana, P. restrepiensis and P. venezuelensis [41,42,43].
Paracoccidioidomycosis is endemic in Latin America, especially in Brazil, Colombia and Venezuela. Argentina (North), Ecuador (Cuenca River valley), and Paraguay (Oriental side) are areas of moderate to high endemicity. Southern Mexico, from the Gulf of Mexico to the Pacific Coast, and Central American countries are territories of low endemicity. Few data are available for Bolivia, Peru, and Uruguay, but autochthonous cases have been reported [44]. Cases of paracoccidioidomycosis have been reported in Europe, United States, Canada, Japan, Africa, and the Middle East. All of them affected patients who visited or lived in South America [9, 44].
Paracoccidioidomycosis is a noncontagious disease, transmitted through conidia inhalation from soil and humid vegetation. Men and armadillos are the main accidental hosts [44].
Paracoccidioides species usually causes a subclinical primary pneumonia [45]. Only 1–2% of infected individuals develop clinical manifestations during their lives [46]. We can distinguish two main clinical forms of paracoccidioidomycosis: the acute or subacute form (juvenile type) and the chronic form (adult type) [45]. In endemic area, the acute/subacute form occurs in children, youths, and in adults under 30 years of age [45], but travelers can show similar clinical presentation. In this case the disease develops a few weeks or months after exposure to Paracoccidioides spp [44]. Dissemination is possible and typically causes fever, lymphadenopathy, and hepatosplenomegaly. Intestinal, cutaneous and neurological involvement is also possible, while lung involvement is rare [45, 46]. The chronic form affects mostly adult males and it usually consists of a reactivation of pulmonary latent foci formed during the primary infection [45], presenting with cough, dyspnea, and sputum expectoration [46]. The chronic form occurs many years after exposure to Paracoccidioides spp [44]. Dissemination is possible and typically affects lymph nodes, skin, adrenal glands, CNS, and oral mucosa [45].
The gold standard for diagnosis is the identification of the fungus in clinical specimens [44]. It appears as spherical yeast, with multiple budding yeasts surrounding a mother cell and birefringent and greenish walls [47]. P. brasiliensis cultures usually take weeks to grow [48]. Histological examination through Grocott–Gomori staining shows granuloma with giant multinucleated cells and polymorphonuclear cell infiltrates [47]. Several immunoassays are available[47]. The most used is double agar gel immunodiffusion because of its cost-effectiveness, sensitivity (> 80%) and specificity (> 90%) [44]. Serum β-d-glucan has good sensitivity but it seems to be not useful for predicting clinical response to antifungal therapy [49]. Paracoccidioides spp can be also detected by PCR based test [47].
The first choice for treatment is itraconazole (200 mg/day administered for 6–9 months in mild disease and for 12–18 months in moderate disease) [50]. Trimethoprim-sulfamethoxazole requires a longer duration of therapy (at least 24 months), with lower cure rates, and higher relapse rates when compared to itraconazole [51]. Liposomal amphotericin B is indicated for CNS disease, severe/disseminated forms and in immunocompromised patients (3–5 mg/kg/day, followed by oral azole or trimethoprim-sulfamethoxazole) [50]. For immunocompromised patients itraconazole should be continued until restoration of cellular immunity (secondary prophylaxis). Primary prophylaxis with trimethoprim-sulfamethoxazole is effective also for paracoccidioidomycosis [50].
Fifteen cases of paracoccidioidomycosis have been described in Italy since 1914 (Table 1) [52]. All patients have traveled or lived in South America: 10 patients in Venezuela and the other 5 in Brazil [52,53,54,55,56]. Lung involvement was described in 10 patients. The other presented extrapulmonary manifestations related to bone (3) [57,58,59], skin and mucosa (6) [53, 54, 60,61,62,63] and lymph nodes (3) [61, 63, 64]. All patients were immunocompetent. Death occurs only in one case [63], an italian farmer who worked for four years in Venezuela and developed after five years chronic pulmonary paracoccidioidomycosis, disseminated to lymph nodes, skin and oral mucosa.
Coccidioidomycosis
Coccidioidomycosis, also known as ‘‘Valley fever’’[65], is a systemic fungal infection caused by some dimorphic fungi belonging to the genus Coccidioides which is classified in the Onygenales order and the Onygenaceae family [20]. Only two species, Coccidioides immitis and Coccidioides posadasii, have been differentiated [65].
Coccidioides species are endemic in the deserts of the North and in some areas of Central and South America [66]. In the United States, California and Arizona are the states with the most of the cases [67].
The transmission of coccidioidomycosis is seasonal. The highest incidence occurs in the fall. Coccidiomycosis is not transmitted from person to person but only through conidia inhalation from soil [66].
Primary pulmonary coccidioidomycosis is the typical clinical presentation of the disease. It could be asymptomatic, mild to moderate (often resolved without treatment) or severe. Some individuals develop pulmonary complications as pleural effusions, cavitations, fibrocavitations, and empyema. Extrapulmonary dissemination occurs in a small percentage of patients (HIV/AIDS patients have higher risk). Dissemination is described typically in these sites: bone, skin and soft tissues, CNS, and lymph nodes. Coccidioidal meningitis is fatal, when untreated [66].
Coccidioidomycosis can be diagnosed by direct microscopic examination and culture of clinical specimens (Fig. 2a) [66]. Colonies of Coccidioides spp develop readily, usually within 3–5 days, on Sabouraud dextrose agar (Fig. 2b) [3, 68]. Through direct microscopy coccidial spherules are observed. Mature spherules are thick-walled (80 μm diameter), with endospores (2–4 μm diameter) inside [68]. Histological examination shows granulomatous inflammation. PCR based tests are also available [66]. Pulmonary coccidioidomycosis could be diagnosed by a serological test, with the immunodiffusion for the detection of precipitating specific antibodies (IgM are produced 1–3 weeks after symptoms onset, followed by IgG 4–8 weeks later) [3]. Skin tests are also feasible. Their positivity indicates past or present infection. Skin test conversion indicates infection in the intervening time [66]. Coccidioidal antigen with an enzyme immunoassay test is available (not commercially available as a kit) [3]. β-d-glucan can be useful, even if it has low sensitivity [69].
Appropriate management of coccidioidomycosis requires treatment with fluconazole (400 mg/day orally) or itraconazole (200 mg 2 times daily) for patients with symptomatic chronic cavitary pneumonia, with soft tissue and bone involvement, immunocompromise and meningitis. Surgery may be considered in patients not responsive to treatment. For CNS disease the treatment is long-life (higher dosages are required). For immunocompromised patients, itraconazole should be continued until restoration of cellular immunity (secondary prophylaxis). For HIV patients in endemic areas primary prophylaxis is not indicated, but serological screening is recommended. Severe disease should be treated with liposomal amphotericin B (3–5 mg/kg/day) [70].
Ten cases of coccidioidomycosis have been described in Italy (Table 1). In particular, three cases were imported from the USA [71,72,73], while others were from South America (Argentina and Venezuela) [74, 75]. In four cases no travel history was reported [76]. All patients were Italian citizens. Only one case was attributed to C. posadasii [74], while in the other cases C. immitis was detected as an etiologic agent. Only one patient was immunocompromised (AIDS) [71]. No deaths were reported. In 1955 Sotgiu and Corbelli described a case of pneumonia in an Italian man who worked in the port of Genoa, who stayed in close contact with grain from North America. In this paper the authors referred to this case as the 4th described in Italy, after Castellani, Jacono and Boeri in 1933 and 1932 [73]. In 1991 Scanarini et al. reported a rare case of primary intrasellar localization of coccidioidomycosis [76]. Gobbi et al. in 2012 reported the case of a 28-years-old Italian man living in Tucson, Arizona for study purposes. He traveled in California and Nevada for visiting Sonora Desert and developed pneumonia, once coming back to Italy [72]. Tortorano et al. (2015) reported a case of a 56-years-old man with persistent erythematous papular plaque without other symptoms [74]. D'Avino et al. in 2012 reported a rare case of AIDS patient with cervical-node coccidiomycosis [71]. Four patients fully recovered [71, 72, 75, 76], two showed clinical improvement [73, 77]. For the other patients no follow-up data are available.
Histoplasmosis
Histoplasmosis is a systemic fungal disease caused by Histoplasma capsulatum, a dimorphic fungus classified in the group of the Onygenales order and Ajellomycetaceae family.
Three different varieties have been historically recognised, each with a typical geographic distribution: H. capsulatum var. capsulatum (New World, mainly in the Ohio and Mississippi River Valleys in USA, and Latin America), responsible for classic human histoplasmosis, H. capsulatum var. duboisii (Central and West Africa), responsible for African human histoplasmosis, H. capsulatum var. farciminosum (Old World, mainly in Asia) responsible for histoplasmosis in equines [78, 79]. Recent studies demonstrated that Histoplasma genus shows high diversity and four geographical clusters have been proposed for american isolates (H. capsulatum sensu stricto, H. ohiense, H. suramericanum, and H. mississippiense), but there is no consensus today on these new species [79, 80]. Some histoplasmin skin reports and case reports showed that histoplasmosis is distributed more extensively than historically thought and probably has ecological niches also in Italy and Europe [81, 82].
Histoplasmosis is transmitted through conidia inhalation from soil containing bird or bat guano [83]. It is usually not transmitted from person to person, even if transmission through solid organ transplant is reported [84].
After inhalation, about 90% of individuals exposed remain asymptomatic or develop self-limited symptoms. Pulmonary histoplasmosis can be acute, subacute, and chronic with cavitations or nodules. The incubation period is short for the acute disease (2 weeks). Dissemination is described typically in HIV/AIDS patients in: skin and soft tissues, CNS, bone marrow and lymph nodes. Physical examination usually reveals lymphadenopathy, hepatomegaly, and splenomegaly [83]. Histoplasmosis is an AIDS-defining illness [85]. The progressive disseminated form has high mortality. Skin involvement implicates diffuse maculopapular eruption and ulceration in advanced lesions [84].
The culture of clinical specimens is routinely used for the diagnosis. Growth in Sabouraud dextrose agar culture usually takes up from 2 to 8 weeks [86]. Cultures of bone marrow, blood and respiratory samples are usefull in disseminated infection and in chronic cavitary pulmonary disease, respectively. CSF culture is often negative [84]. Histopathological evidence of H. capsulatum is one of the diagnostic criteria of proven histoplasmosis [87]. The yeast cells are ovoidal (size 2–4 μm), usually intracellular and showing narrow-based budding with Gomori methenamine silver (Fig. 3a) or periodic–acid Schiff stains. Histoplasmosis typically presents granulomatous inflammation (Fig. 3b), which may be caseating or non caseating [84]. Serological tests are available and show the highest sensitivity in subacute and chronic histoplasmosis [86]. Histoplasmosis antigen testing is mostly done on urine, but it is available (even if not validated) also on serum, bronchoalveolar lavage, and CSF. It’s useful also for monitoring the effectiveness of the therapy. The sensitivity is high in HIV/AIDS patients and in the disseminated disease (antigen detected in 95% of cases) [84]. For CNS histoplasmosis diagnosis the best combination is made by serology and CSF antigen tests [88]. Urine test for histoplasmosis antigen is indicated in patients with CD4 count < 100 cells/µL in areas where histoplasmosis is endemic, and it must be repeated every year [89]. Histoplasmosis antigen is not available worldwide (also in Italy). Cross-reactions occur with other endemic mycoses both for serologic tests and antigens [84, 86]. Serum β-d-glucan is not reliable for histoplasmosis diagnosis [31]. PCR based tests are useful (even if they are still not validated) [84].
Chronic pulmonary and disseminated histoplasmosis are fatal if untreated [84]. International guidelines are available [90]. The first choice for treatment is itraconazole (600 mg/day for 3 days, then 200–400 mg/day for 12 months) for mild/moderate acute pulmonary histoplasmosis and for chronic cavitary pulmonary. For severe pulmonary disease intravenous steroids are recommended [90]. Liposomal amphotericin B is indicated in CNS disease, severe/disseminated forms and in immunocompromised patients (3–5 mg/kg/day) for 2 weeks, followed by itraconazole for at least 12 months. For immunocompromised patients itraconazole should be continued until restoration of cellular immunity and demonstration of clinical cure (secondary prophylaxis) [91]. Primary prophylaxis with itraconazole 200 mg/day is indicated for HIV patients with CD4 < 150 cells/μL living in endemic areas [84].
Systematic reviews have been conducted about cases of histoplasmosis in Italy [4, 6,7,8]. According to our systematic research, 105 cases of histoplasmosis have been described in Italy (Table 2). Death occurs in 20 patients (19%). For 28 patients no history of travels outside Italy was described. For all the other subjects a country of infection's acquisition was signaled. Most cases have been acquired in Ecuador (n = 20) [92,93,94,95] and in general in central and south America [96, 97]. Immunocompromised patients were 49:42 with HIV/AIDS, two with leukemia [98], one with breast cancer [99], one has Crohn disease [94], one sarcoidosis [94], one rheumatoid arthritis [100] and only one was a lung transplant recipient [101]. Among them, 14 deaths were reported (28%). Almost half of the patients (n = 48) presented disseminated histoplasmosis. Most of them had AIDS as a major risk factor. Unusual clinical presentations have also been reported: one case of endophthalmitis occurred in an Italian 64 years old man with diabetes who lived in Brazil for several years [102], and one of adrenal incidentaloma [103] in an Italian 74-year-old man who worked in Pakistan as a well-driller for 2 years. Two different clusters have been investigated. In 1997 Nasta et al. described the first one. Four Italian spelunkers, returning from Perù, presented mild acute pulmonary disease with hepatosplenomegaly with complete resolution after ketoconazole treatment [104]. The second consists of 17 members of a naturalistic expedition to Ecuador. All subjects were immunocompetent, only one presented disseminated histoplasmosis, while the other suffered from mild acute pulmonary disease. All of them recovered. Only seven patients required antifungal therapy [92].
Discussion
We found out from 1914 to nowadays: 105 cases of histoplasmosis, 15 of paracoccidioidomycosis, 10 of coccidioidomycosis, 10 of blastomycosis and 3 of talaromycosis reported in Italy (Fig. 4). The most reported infection in Italy is histoplasmosis and this probably reflects its global distribution [79]. Typically, cases of endemic mycoses in non endemic countries are described in travelers, expatriates and migrants [1]. The understanding of the epidemiology of such diseases is still in progress [105].
In Italy a national surveillance system of endemic mycoses is missing. Moreover, the diagnosis of these mycoses is often difficult because clinical experience of physicians and diagnostic tests both are lacking (except for some reference centers). Thus, our data probably don’t reflect the real epidemiology of endemic mycoses in the country. In Spain, recently, Molina-Morant et al. reviewed the literature about endemic mycoses in the country between 1997 and 2014. There were 286 cases of histoplasmosis, 94 of coccidioidomycosis and 25 of paracoccidioidomycosis [106].
The period between the last time spent in an endemic area and the time of diagnosis sometimes is very long and could last years [62, 74, 107]. This latency period usually is longer for migrants or expatriates rather than travelers [4]. This reminds us how important it is to accurately collect the entire traveling history of the patient.
Among 143 cases of fungal infections reported here, death occurs in 21 cases (20 histoplasmosis, and one paracoccidioidomycosis). Mortality rate was 14.7% (21 deaths/143 cases). Immunosuppression is a major risk factor for getting these mycoses and for more severe outcomes [8]. Immunocompromised patients were 54 and among them 46 had HIV/AIDS. Mortality rate was 26% in immunocompromised patients (14 deaths/54 cases). Other reported conditions of immunosuppression were: cancer, inflammatory bowel diseases, sarcoidosis, rheumatologic disorders. We collect only one case of histoplasmosis in a lung transplant recipient [101]. However, this topic is emerging worldwide, considering the increasing use of immunosuppressive drugs for many diseases. In 2019 the American Society of Transplantation published their guidelines on diagnosis, prevention and management of blastomycosis, histoplasmosis, and coccidioidomycosis, that are endemic in USA, in the pre‐ and post- transplant period [108].
For histoplasmosis (n = 28), coccidioidomycosis (n = 4), and blastomycosis (n = 6), cases in people who never traveled or have links to endemic regions have been reported. So, these cases could be considered as autochthonous. In particular, evidence about the presence of Histoplasma spp in Italy has been documented by isolation in soil [109], cases in animals [110, 111] and histoplasmin reactivity surveys [81, 98]. No similar studies have been conducted in Italy for coccidioidomycosis, and blastomycosis. Nevertheless, species identification for the above mentioned isolations from soil or animals only relied on morphological features, and only one of these possible autochthonous cases was diagnosed by PCR-based tests.
In non-endemic countries the diagnosis of endemic mycoses could be challenging. Even if the culture remains the gold standard, PCR based tests detected the fungus in 8 cases (4 of these with negative cultures of clinical samples). Most of the described cases were diagnosed by histology and/or cultures. Serology and antigen testing have been less diriment.
Conclusions
The increasing trend of international travels, migration flows alongside the increasing number of persons living with immunosuppression conditions (e.g. solid organ transplants) has led to an increase of imported cases of endemic mycoses in non endemic countries such as Italy. A story of travels and immunosuppression should lead clinicians to consider endemic mycoses in differential diagnosis of systemic diseases.
References
Bonifaz A, Vázquez-González D, Perusquía-Ortiz AM. Endemic systemic mycoses: coccidioidomycosis, histoplasmosis, paracoccidioidomycosis and blastomycosis. J Dtsch Dermatol Ges. 2011;9:705–14 (quiz 715).
Gauthier GM. Fungal dimorphism and virulence: molecular mechanisms for temperature adaptation, immune evasion, and in vivo survival. Mediat Inflamm. 2017;2017:8491383.
Thompson GR 3rd, Le T, Chindamporn A, et al. Global guideline for the diagnosis and management of the endemic mycoses: an initiative of the European Confederation of Medical Mycology in cooperation with the International Society for Human and Animal Mycology. Lancet Infect Dis. 2021;21:e364–74.
Antinori S, Giacomelli A, Corbellino M, et al. Histoplasmosis diagnosed in Europe and Israel: a case report and systematic review of the literature from 2005 to 2020. J Fungi. 2021. https://doi.org/10.3390/jof7060481.
Ashraf N, Kubat RC, Poplin V, et al. Re-drawing the maps for endemic mycoses. Mycopathologia. 2020;185:843–65.
Antinori S, Magni C, Nebuloni M, et al. Histoplasmosis among human immunodeficiency virus-infected people in Europe: report of 4 cases and review of the literature. Medicine. 2006;85:22–36.
Farina C, Rizzi M, Ricci L, et al. Imported and autochthonous histoplasmosis in Italy: new cases and old problems. Rev Iberoam Micol. 2005;22:169–71.
Farina C, Gnecchi F, Michetti G, et al. Imported and autochthonous histoplasmosis in Bergamo province, Northern Italy. Scand J Infect Dis. 2000;32:271–4.
Wagner G, Moertl D, Glechner A, et al. Paracoccidioidomycosis diagnosed in Europe—a systematic literature review. J Fungi. 2021. https://doi.org/10.3390/jof7020157.
Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
Cao C, Xi L, Chaturvedi V. Talaromycosis (Penicilliosis) due to Talaromyces (Penicillium) marneffei: insights into the clinical trends of a major fungal disease 60 years after the discovery of the pathogen. Mycopathologia. 2019;184:709–20.
Ustianowski AP, Sieu TPM, Day JN. Penicillium marneffei infection in HIV. Curr Opin Infect Dis. 2008;21:31–6.
Sirisanthana V, Sirisanthana T. Disseminated Penicillium marneffei infection in human immunodeficiency virus-infected children. Pediatr Infect Dis J. 1995;14:935–40.
Kawila R, Chaiwarith R, Supparatpinyo K. Clinical and laboratory characteristics of penicilliosis marneffei among patients with and without HIV infection in Northern Thailand: a retrospective study. BMC Infect Dis. 2013;13:464.
Supparatpinyo K, Khamwan C, Baosoung V, et al. Disseminated Penicillium marneffei infection in Southeast Asia. Lancet. 1994;344:110–3.
Guidelines for Prevention and Treatment of Opportunistic Infections in HIV-Infected Adults and Adolescents Recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America; 2009
Viviani MA, Tortorano AM, Rizzardini G, et al. Treatment and serological studies of an Italian case of penicilliosis marneffei contracted in Thailand by a drug addict infected with the human immunodeficiency virus. Eur J Epidemiol. 1993;9:79–85.
Antinori S, Gianelli E, Bonaccorso C, et al. Disseminated Penicillium marneffei infection in an HIV-positive Italian patient and a review of cases reported outside endemic regions. J Travel Med. 2006;13:181–8.
Basile G, Piccica M, Vellere I, et al. Disseminated talaromyces infection in an AIDS patient. Clin Microbiol Infect. 2022;28:64–5.
Dukik K, Muñoz JF, Jiang Y, et al. Novel taxa of thermally dimorphic systemic pathogens in the Ajellomycetaceae (Onygenales). Mycoses. 2017. https://doi.org/10.1111/myc.12601.
Brown EM, McTaggart LR, Zhang SX, et al. Phylogenetic analysis reveals a cryptic species Blastomyces gilchristii, sp. nov. within the human pathogenic fungus Blastomyces dermatitidis. PLoS ONE. 2013. https://doi.org/10.1371/journal.pone.0059237.
Schwartz IS, Wiederhold NP, Hanson KE, et al. Blastomyces helicus, a new dimorphic fungus causing fatal pulmonary and systemic disease in humans and animals in Western Canada and the United States. Clin Infect Dis. 2019. https://doi.org/10.1093/cid/ciy483.
Baily GG, Robertson VJ, Neill P, et al. Blastomycosis in Africa: clinical features, diagnosis, and treatment. Clin Infect Dis. 1991;13:1005–8.
Randhawa HS, Chowdhary A, Kathuria S, et al. Blastomycosis in India: report of an imported case and current status. Med Mycol. 2013. https://doi.org/10.3109/13693786.2012.685960.
Codifava M, Guerra A, Rossi G, et al. Unusual osseous presentation of blastomycosis in an immigrant child: a challenge for European pediatricians. Ital J Pediatr. 2012. https://doi.org/10.1186/1824-7288-38-69.
Saccente M, Woods GL. Clinical and laboratory update on blastomycosis. Clin Microbiol Rev. 2010;23:367–81.
Shelnutt LM, Kaneene JB, Carneiro PAM, et al. Prevalence, distribution, and risk factors for canine blastomycosis in Michigan, USA. Med Mycol. 2019;58:609–16.
Klein BS, Vergeront JM, Davis JP. Epidemiologic aspects of blastomycosis, the enigmatic systemic mycosis. Semin Respir Infect. 1986;1:29–39.
Bradsher RW Jr. The endemic mimic: blastomycosis an illness often misdiagnosed. Trans Am Clin Climatol Assoc. 2014;125:188–202 (discussion 202–3).
Mazi PB, Rauseo AM, Spec A. Blastomycosis. Infect Dis Clin North Am. 2021;35:515–30.
Girouard G, Lachance C, Pelletier R. Observations on (1–3)-beta-D-glucan detection as a diagnostic tool in endemic mycosis caused by Histoplasma or Blastomyces. J Med Microbiol. 2007;56:1001–2.
Limper AH, Knox KS, Sarosi GA, et al. An official American thoracic society statement: treatment of fungal infections in adult pulmonary and critical care patients. Am J Respir Crit Care Med. 2011;183:96–128.
Chapman SW, Dismukes WE, Proia LA, et al. Clinical practice guidelines for the management of blastomycosis: 2008 update by the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:1801–12.
Ietto G, Baj A, Parise C, et al. Blastomycosis of the psoas muscles. IDCases. 2021;24:e01156.
Cavalot AL, Cravero L, Valente G, et al. Blastomycosis of the cervicofacial area: a review of the literature and case report. Acta Otorhinolaryngol Ital. 1992;12:605–10.
Sgobbi S, Bellando-Randone P. Systemic North American blastomycosis. Chir Organi Mov. 1978;64:439–46.
Florenzano G, Zini F. Experimental observations on the etiology of a granulomatous pulmonary blastomycosis. Sperimentale. 1950;100:139–56.
Rivasi F, Nanetti A, Cesinaro AM, et al. Histopathological evidence of North American blastomycosis in Italy: report of two cases. APMIS. 2000;108:273–5.
Chasen W. Two cases of facial blastomycosis. Riv Ital Stomatol. 1951;6:1040–7.
Rimondi AP, Bianchini E, Barucchello G, et al. Addison’s disease caused by adrenal blastomycosis: a case report with fine needle aspiration (FNA) cytology. Cytopathology. 1995;6:277–9.
Ta F, Jw W, Oa P. South American blastomycosis or paracoccidioidycosis: the mycosis of Lutz, Splendore, and Almeida. AMA Arch Derm Syphilol. 1954. https://doi.org/10.1001/archderm.1954.01540200026003.
Turissini DA, Gomez OM, Teixeira MM, et al. Species boundaries in the human pathogen Paracoccidioides. Fungal Genet Biol. 2017;106:9–25.
Mavengere H, Mattox K, Teixeira MM, et al. Genomes reflect high levels of species divergence and little interspecific gene flow. MBio. 2020. https://doi.org/10.1128/mBio.01999-20.
Martinez R. New trends in paracoccidioidomycosis epidemiology. J Fungi. 2017. https://doi.org/10.3390/jof3010001.
Queiroz-Telles F, Escuissato DL. Pulmonary paracoccidioidomycosis. Semin Respir Crit Care Med. 2011;32:764–74.
de Queiroz-Telles FV, Peçanha Pietrobom PM, Rosa Júnior M, et al. New insights on pulmonary paracoccidioidomycosis. Semin Respir Crit Care Med. 2020;41:53–68.
Ferreira CS, de Castro Ribeiro EM, de Miranda GA, et al. Current strategies for diagnosis of paracoccidioidomycosis and prospects of methods based on gold nanoparticles. Future Microbiol. 2016;11:973–85.
Teles FRR, Martins ML. Laboratorial diagnosis of paracoccidioidomycosis and new insights for the future of fungal diagnosis. Talanta. 2011;85:2254–64.
de Azevedo Melo AS, de Castro Lima Santos DW, Lima SL, et al. Evaluation of (1 → 3)-β-D-glucan assay for diagnosing paracoccidioidomycosis. Mycoses. 2020;63:38–42.
Shikanai-Yasuda MA, Mendes RP, Colombo AL, et al. Brazilian guidelines for the clinical management of paracoccidioidomycosis. Epidemiol Serv Saude. 2018;27:e0500001.
Borges SRC, da Silva GMS, da Chambela MC, et al. Itraconazole vs. trimethoprim-sulfamethoxazole: a comparative cohort study of 200 patients with paracoccidioidomycosis. Med Mycol. 2014;52:303–10.
Dalla Favera GB. Per la conoscenza della cosidetta blastomicosi cutanea (con un’osservazione personale di oidiomicosi (Gilchrist, Bushke) zimonematosi (de Beurmann et Gougerot). Giorn Ital Mal Ven Pelle. 1914;55:650–729.
Farris G. Report on a case of paracoccidioidomycosis (so-called Brazilian blastomycosis). G Ital Dermatol. 1955;96:321–58.
Solaroli C, Alol F, Becchis G, Zina A, Pippione M. Paracoceidioidomycosis description of a case. G Ital Dermatol Venereol. 1998;133:51–4.
Finzi FF, Bubola D, Lasagni A. Blastomicosi sudamericana. Ann Ital Dermatol Clin Sper. 1980;34:277–85.
Bertaccini G. Contributo allo studio della cosi detta blastomicosa sud-americana. Giorn Ital Dermatol Sifil. 1934;75:783–828.
Pecoraro C, Pinto A, Tortora G, et al. A case of South American blastomycosis of the bones and the lungs. Radiol Med. 1998;95:521–3.
Fulciniti F, Troncone G, Fazioli F, et al. Osteomyelitis by Paracoccidioides brasiliensis (South American blastomycosis): cytologic diagnosis on fine-needle aspiration biopsy smears: a case report. Diagn Cytopathol. 1996;15:442–6.
Borgia G, Reynaud L, Cerini R, et al. A case of paracoccidioidomycosis: experience with long-term therapy. Infection. 2000;28:119–20.
Cuomo A, Capra R, Di Gregorio A, Garavaldi G. On one case of South American blastomycosis with peculiar evolution. Riv Patol Clin Tuberc Pneumol. 1985;56:453–72.
Molese A, Pane A. South American blastomycosis (Lutz–Splendore–Almeida disease). Acta Med Ital Mal Infett Parassit. 1957;12:275–98.
Benoldi D, Alinovi A, Pezzarossa E, et al. Paracoccidioidomycosis (South American blastomycosis): a report of an imported case previously diagnosed as tuberculosis. Eur J Epidemiol. 1985;1:150–2.
Scarpa C, Nini G, Gualdi G. Clinico-radiological contribution to the study of paracoccidioidomycosis. Minerva Dermatol. 1965;40:413–21.
Schiraldi O, Grimaldi N. Granulomatosi paracoccidioide. Policlinico. 1963;70:65–84.
Chaturvedi S, Victor TR, Marathe A, et al. Real-time PCR assay for detection and differentiation of Coccidioides immitis and Coccidioides posadasii from culture and clinical specimens. PLoS Negl Trop Dis. 2021. https://doi.org/10.1371/journal.pntd.0009765.
Johnson RH, Sharma R, Kuran R, et al. Coccidioidomycosis: a review. J Investig Med. 2021;69:316–23.
McCotter OZ, Benedict K, Engelthaler DM, et al. Update on the Epidemiology of coccidioidomycosis in the United States. Med Mycol. 2019;57:S30-40.
Sutton DA. Diagnosis of coccidioidomycosis by culture: safety considerations, traditional methods, and susceptibility testing. Ann N Y Acad Sci. 2007;1111:315–25.
Zangeneh TT, Malo J, Luraschi-Monjagatta C, et al. Positive (1–3) B-d-glucan and cross reactivity of fungal assays in coccidioidomycosis. Med Mycol. 2015;53:171–3.
Galgiani JN, Ampel NM, Blair JE, et al. 2016 Infectious Diseases Society of America (IDSA) clinical practice guideline for the treatment of coccidioidomycosis. Clin Infect Dis. 2016;63:e112–46.
D’Avino A, Di Giambenedetto S, Fabbiani M, et al. Coccidioidomycosis of cervical lymph nodes in an HIV-infected patient with immunologic reconstitution on potent HAART: a rare observation in a nonendemic area. Diagn Microbiol Infect Dis. 2012;72:185–7.
Gobbi F, Angheben A, Farina C, et al. Coccidioidomycosis: first imported case in Italy. J Travel Med. 2012;19:192–4.
Sotgiu G, Corbelli G. Osservazione dei primi due casi di istoplasmosi in Italia e di un caso di coccidioidomicosi. Bull Sci Med. 1955;127:85–92.
Tortorano AM, Carminati G, Tosoni A, et al. Primary cutaneous coccidioidomycosis in an Italian Nun working in South America and review of published literature. Mycopathologia. 2015;180:229–35.
Corpolongo A, Giancola ML, Ghirga P, et al. An imported case of acute pulmonary coccidioidomycosis in an Italian traveller. Infection. 2014;42:921–4.
Scanarini M, Rotilio A, Rigobello L, et al. Primary intrasellar coccidioidomycosis simulating a pituitary adenoma. Neurosurgery. 1991;28:748–51.
Vilardo S, Cavalcanti U, Mancuso E. On a case of coccidioidomycos. Gazz Int Med Chir. 1964;68:2307–20.
Rodrigues AM, Beale MA, Hagen F, et al. The global epidemiology of emerging Histoplasma species in recent years. Stud Mycol. 2020;97:100095.
Taylor ML, del Rocío Reyes-Montes M, Estrada-Bárcenas DA, et al. Considerations about the geographic distribution of histoplasma species. Appl Environ Microbiol. 2022. https://doi.org/10.1128/aem.02010-21.
Sepúlveda VE, Márquez R, Turissini DA, et al. Genome sequences reveal cryptic speciation in the human pathogen Histoplasma capsulatum. MBio. 2017;8(6):e01339-e1417. https://doi.org/10.1128/mBio.01339-17.
Bahr NC, Antinori S, Joseph Wheat L, et al. Histoplasmosis infections worldwide: thinking outside of the Ohio River Valley. Curr Trop Med Rep. 2015;2:70–80.
Confalonieri M, Nanetti A, Gandola L, et al. Histoplasmosis capsulati in Italy: autochthonous or imported? Eur J Epidemiol. 1994;10:435–9.
Araúz AB, Papineni P. Histoplasmosis. Infect Dis Clin North Am. 2021;35:471–91.
Kauffman C, Miceli M. Histoplasmosis and blastomycosis in solid organ transplant recipients. J Fungi. 2015;1:94–106.
Zanotti P, Chirico C, Gulletta M, et al. Disseminated Histoplasmosis as AIDS-presentation. Case report and comprehensive review of current literature. Mediterr J Hematol Infect Dis. 2018;10:e2018040.
Azar MM, Hage CA. Laboratory diagnostics for histoplasmosis. J Clin Microbiol. 2017;55:1612–20.
Donnelly JP, Chen SC, Kauffman CA, et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2019;71:1367–76.
Bloch KC, Myint T, Raymond-Guillen L, et al. Improvement in diagnosis of histoplasma meningitis by combined testing for histoplasma antigen and immunoglobulin G and immunoglobulin M anti-histoplasma antibody in cerebrospinal fluid. Clin Infect Dis. 2017;66:89–94.
Saag MS. HIV Infection—screening, diagnosis, and treatment. Reply. N Engl J Med. 2021;385:1344.
Wheat LJ, Joseph Wheat L, Freifeld AG, et al. Clinical Practice Guidelines for the Management of Patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis. 2007;45:807–25.
Ryom L, De Miguel R, Cotter AG, et al. Major revision version 11.0 of the European AIDS clinical society guidelines 2021. HIV Med. 2022. https://doi.org/10.1111/hiv.13268.
Staffolani S, Riccardi N, Farina C, et al. Acute histoplasmosis in travelers: a retrospective study in an Italian referral center for tropical diseases. Pathog Glob Health. 2020;114:40–5.
Delfino E, Di Biagio A, Chandrapatham K, et al. Disseminated histoplasmosis with mucocutaneous immune reconstitution inflammatory syndrome in an HIV-infected patient. AIDS Res Hum Retroviruses. 2015;31:274–5.
Visonà A, Danieli D, Dante S, et al. Bilateral nodular pulmonary histoplasmosis: cytohistological correlation. Pathologica. 1991;83:197–200.
Bartoloni A, et al. Progressive disseminated histoplasmosis (PDH) in an art naïve migrant with advanced infection. Trop Med Int Health. 2011;16:163–4.
Papalini C, Belfiori B, Martino G, et al. An Italian Case of Disseminated Histoplasmosis Associated with HIV. Case Rep Infect Dis. 2019;2019:7403878.
Amadori F, Doria R, Gemignani G, et al. Histoplasmosis: the multiple sides of an uncommon disease. Infez Med. 2015;23:61–8.
Sotgiu G, Mantovani A, Mazzoni A. Histoplasmosis in Europe. Mycopathol Mycol Appl. 1970;40(1–2):53–74.
Galetta D, Pelosi G, Nebuloni M, et al. Challenging diagnosis of an unusual solitary pulmonary nodule. Thorac Cardiovasc Surg. 2007;55:123–5.
Romano C, Castelli A, Laurini L, et al. Case report. Primary cutaneous histoplasmosis in an immunosuppressed patient. Mycoses. 2000;43:151–4.
Righi E, Lugano M, Assi M, et al. Histoplasmosis in a lung transplant recipient from a nonendemic area. Transpl Int. 2014;27:e99-101.
Grancini A, Colosimo M, Mapelli C, et al. A case of Histoplasma capsulatum endophthalmitis diagnosed in Italy. Travel Med Infect Dis. 2013;11:256–8.
Lio S, Cibin M, Marcello R, et al. Adrenal bilateral incidentaloma by reactivated histoplasmosis. J Endocrinol Invest. 2000;23:476–9.
Nasta P, Donisi A, Cattane A, Chiodera A, Casari S. Acute histoplasmosis in spelunkers returning from Mato Grosso. Peru J Trav Med. 1997;4:176–8.
Antinori S, Ridolfo AL, Casalini G, Giacomelli A. Endemic mycoses: geographical distribution is still a work in progress. Lancet Infect Dis. 2022;22:451.
Molina-Morant D, Sánchez-Montalvá A, Salvador F, et al. Imported endemic mycoses in Spain: evolution of hospitalized cases, clinical characteristics and correlation with migratory movements, 1997–2014. PLoS Negl Trop Dis. 2018;12:e0006245.
Rivasi F, Casali B, Nanetti A, et al. Histoplasma capsulatum var. capsulatum occurring in an HIV-positive Ghanaian immigrant to Italy. Identification of H. capsulatum DNA by PCR from paraffin sample. APMIS. 2001;109:721–5.
Miller R, Assi M. AST Infectious Diseases Community of Practice. Endemic fungal infections in solid organ transplant recipients-Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transpl. 2019;33:e13553.
Sotgiu G, Mazzoni A, Mantovani A, Ajello L, Palmer J. Histoplasma capsulatum: occurrence in soil from the Emilia Romagna region of Italy. Science. 1965;147:624–6.
Mantovani A, Mazzoni A, Ajello L. Histoplasmosis in Italy Isolation of Histoplasma capsulatum from dogs in the province of Bologna. Sabouraudia. 1968;6:163–4.
Reginato A, Giannuzzi P, Ricciardi M, et al. Extradural spinal cord lesion in a dog: first case study of canine neurological histoplasmosis in Italy. Vet Microbiol. 2014;170:451–5.
Corbelli G, Mazzoni A, Allegri L. Two additional cases of histoplasmosis as observed in Bologna Medical Clinic. Minerva Med. 1957;48:3823–36.
Zavoli G. Istoplasmosi oro-faringea. Arch Ital Otol. 1957;68:374.
Allegri L, Bottiglioni E. Nuova segnalazione di un caso di istoplasmosi polmonare autoctona. Mal Infez. 1958;4:135–7.
Costa A, Biressi PC, Della Beffa A, Solero C. Il primo caso clinico di istoplasmosi osservato in Piemonte. Giorn Accad Med. 1959;122:53–63.
Salfelder K, Reyes de Liscano T, Romanovich J, Moncador F. Uber einen fall von lungenhistoplasmom mit möglicher infektion in Italien. Mycosen. 1963;6:29–34.
Papa B, Del Genio A. Su un caso di istoplasmosi a manifestazione cutanea. Arch Chir. 1965;2:257–158.
Mesolella C, Amorelli A. On a case of laryngeal histoplasmosis. Arch Ital Laringol. 1966;74:273–93.
Pellegrino A. Considerazioni clinico-epidemiologiche su un caso sporadico di istoplasmosi polmonare. Giorn It Mal Inf Par. 1977;29:318–9.
Zanini R, De Longis P, De Stefano G, Soscia F, Mascioli G, Perroni L. Osservazioni cliniche su un caso di istoplasmosi muco-cutanea. Giorn It Mal Inf Par. 1987;39:968–9.
Masini T, Ghidoni P, Riviera L. Mycoses in immunocompromised patients. An histopathologic study of 61 cases. Boll Ist Sieroter Milan. 1988;67:232–40.
Vaj P, Dragogna T, Milani R, et al. Pulmonary histoplasmosis. Presentation of a case. Radiol Med. 1989;78:393–5.
Tinelli M, Michelone G, Cavanna C. Recurrent Histoplasma capsulatum pneumonia: a case report. Microbiologica. 1992;15:89–93.
Biglino A, De Rosa G, Lipani F. Upper lobe infiltrate with cough, fever, fatigue. Eur Respir J. 1992;5:1021–2.
Gori S, Scasso A, Paladini A. Histoplasmosis due to Histoplasma capsulatum in an Italian HIV positive patient returning from the Americas. J Mycol Méd. 1993;4:239–41.
Manfredi R, Mazzoni A, Nanetti A, et al. Histoplasmosis capsulati and duboisii in Europe: the impact of the HIV pandemic, travel and immigration. Eur J Epidemiol. 1994;10:675–81.
Gargiulo M, Crispo C, Conte M, Forte G, Gnarini N, Sangiovanni V, Chirianni A, Progressive disseminated histoplasmosis in black male subject from Ivory Coast. Abstract 308. In: 5th European conference on clinical aspects and treatment of HIV-infection, Copenhagen; 1995
Conte A, De Longis P, Bordi E, De Mori P, Grillo LR. Istoplasmosi disseminata in un paziente con infezione da HIV. Giorn It AIDS. 1996;7:55–8.
Vullo V, Mastroianni CM, Ferone U, et al. Central nervous system involvement as a relapse of disseminated histoplasmosis in an Italian AIDS patient. J Infect. 1997;35:83–4.
Antinori S, Galimberti L, Bonaccorso C, et al. A case of fatal disseminated histoplasmosis of autochthonous origin in an Italian AIDS patient. Eur J Clin Microbiol Infect Dis. 1997;16:545–6.
Angius AG, Viviani MA, Muratori S, et al. Disseminated histoplasmosis presenting with cutaneous lesions in a patient with acquired immunodeficiency syndrome. J Eur Acad Dermatol Venereol. 1998;10:182–5.
Pometta R, Trovato C, Viviani MA, et al. Chronic pulmonary histoplasmosis in a patient with a recent history of tuberculosis and persistent round lung lesions. Eur J Clin Microbiol Infect Dis. 1999;18:229–31.
Faggi E, Tortoli E, Bartoloni A, et al. Late diagnosis of histoplasmosis in a Brazilian patient with acquired immunodeficiency syndrome. Clin Microbiol Infect. 2001;7:48–9.
D’Antuono A, Andalò F, Raone B. Cutaneous lesions of disseminated histoplasmosis in a HIV positive patient. Ann Ital Dermatol Allergol Clin Sper. 2000;54(1):33–5.
Antinori S, Ridolfo AL, Corbellino M, et al. Disseminated histoplasmosis in patients with AIDS. 2 case reports. Recenti Prog Med. 2000;91:362–4.
Mignogna MD, Fedele S, Lo Russo L, et al. A case of oral localized histoplasmosis in an immunocompetent patient. Eur J Clin Microbiol Infect Dis. 2001;20:753–5.
Rizzi EB, Schininà V, Bellussi A, et al. Pulmonary mycosis in AIDS. Eur J Radiol. 2001;37:42–6.
Calza L, Manfredi R, Donzelli C, et al. Disseminated histoplasmosis with atypical cutaneous lesions in an Italian HIV-infected patient: another autochtonous case. HIV Med. 2003;4:145–8.
Lo Cascio G, Ligozzi M, Maccacaro L, et al. Diagnostic aspects of cutaneous lesions due to Histoplasma capsulatum in African AIDS patients in nonendemic areas. Eur J Clin Microbiol Infect Dis. 2003;22:637–8.
Faggian F, Lanzafame M, Lattuada E, et al. Histoplasmosis in two human immunodeficiency virus-positive immigrants to Italy: clinical features and management in the highly active antiretroviral therapy era. South Med J. 2004;97:398–400.
Garavelli PL, Rosa F, Brustia D, et al. Disseminated histoplasmosis in a HIV seropositive patient with Kaposi sarcoma. Recenti Prog Med. 2005;96:492.
Inojosa W, Rossi MC, Laurino L, et al. Progressive disseminated histoplasmosis among human immunodeficiency virus-infected patients from West-Africa: report of four imported cases in Italy. Infez Med. 2011;19:49–55.
Fortuna G, Mignogna MD. Oral histoplasmosis of a healthy man in a non-endemic area. Infection. 2011;39:497–8.
Scarlata F, Imburgia C, Trizzino M, et al. Leprosy-like cutaneous presentation of Histoplasma capsulatum infection in an African HIV+ patient. Infez Med. 2012;20:211–3.
Ardizzoni A, Baschieri MC, Manca L, et al. The mycoarray as an aid for the diagnosis of an imported case of histoplasmosis in an Italian traveler returning from Brazil. J Travel Med. 2013;20:336–9.
Bonsignore A, Orcioni GF, Barranco R, et al. Fatal disseminated histoplasmosis presenting as FUO in an immunocompetent Italian host. Leg Med. 2017;25:66–70.
Asperges E, Cavanna C, Mollaschi EMG. A case report of disseminated histoplasmosis in AIDS diagnosed through peripheral blood smear. Curr HIV Res. 2021;19(5):457–9.
Acknowledgements
We thank Chiara Savio (Department of Infectious—Tropical Diseases and Microbiology, IRCCS Sacro Cuore—Don Calabria Hospital, Negrar di Valpolicella, Verona, Italy) for images concession. We thank Dr. Luca Tirloni for his kind help. We thank the AMCLI (Associazione Microbiologi Clinici Italiani) Committee for Mycology members: Gianluigi Lombardi; Stefano Andreoni; Elisabetta Blasi; Claudio Farina; Paolo Fazii, Silvana Sanna; Laura Trovato; Giuliana Lo Cascio.
Funding
Open access funding provided by Università degli Studi di Trieste within the CRUI-CARE Agreement. A.A.’s work was partly funded by the Italian Ministry of Health “Fondi Ricerca Corrente” to IRCCS Sacro Cuore—Don Calabria Hospital—Linea 1.
Author information
Authors and Affiliations
Consortia
Contributions
Conceptualization, SDB and LP; methodology, AA and LP; investigation, VZ, RP, FDA; writing, review and editing, VZ, RP, FDA, SDB, LP, AA, CF, GL; supervision, MC, FL, RL. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None to declare.
Additional information
Handling Editor: J.-P. Bouchara.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zerbato, V., Di Bella, S., Pol, R. et al. Endemic Systemic Mycoses in Italy: A Systematic Review of Literature and a Practical Update. Mycopathologia 188, 307–334 (2023). https://doi.org/10.1007/s11046-023-00735-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-023-00735-z