Abstract
Background
Deficiency of vitamin E results in several neurological and age-related disorders in humans. Utilization of maize mutants with favourable vte4-allele led to the development of several α-tocopherol (vitamin E) rich (16–19 µg/g) maize hybrids worldwide. However, the degradation of tocopherols during post-harvest storage substantially affects the efficacy of these genotypes.
Methods and results
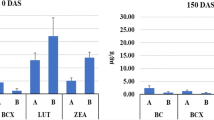
We studied the role of lipoxygenase enzyme and Lipoxygenase 3 (LOX3) gene on the degradation of tocopherols at monthly intervals under traditional storage up to six months in two vte4-based contrasting-tocopherol retention maize inbreds viz. HKI323-PVE and HKI193-1-PVE. The analysis revealed significant degradation of tocopherols across storage intervals in both the inbreds. Lower retention of α-tocopherol was noticed in HKI193-1-PVE. HKI323-PVE with the higher retention of α-tocopherol showed lower lipoxygenase activity throughout the storage intervals. LOX3 gene expression was higher (~ 1.5-fold) in HKI193-1-PVE compared to HKI323-PVE across the storage intervals. Both lipoxygenase activity and LOX3 expression peaked at 120 days after storage (DAS) in both genotypes. Further, a similar trend was observed for LOX3 expression and lipoxygenase activity. The α-tocopherol exhibited a significantly negative correlation with lipoxygenase enzyme and expression of LOX3 across the storage intervals.
Conclusions
HKI323-PVE with high tocopherol retention, low -lipoxygenase activity, and -LOX3 gene expression can act as a potential donor in the vitamin E biofortification program. Protein-protein association network analysis also indicated the independent effect of vte4 and LOX genes. This is the first comprehensive report analyzing the expression of the LOX3 gene and deciphering its vital role in the retention of α-tocopherol in biofortified maize varieties under traditional storage.




Similar content being viewed by others
Data Availability
All data needed to support the conclusions are included in this article. Additional data related to this paper can be requested from the corresponding author.
References
Hossain F, Zunjare RU, Muthusamy V, Bhat JS, Mehta BK, Sharma D, Talukder ZA, Chhabra R, Katral A, Dutta S, Chand G, Bhatt V, Mishra SJ, Gain N, Kasana R, Ikkurti G, Duo H (2022a) Biofortification of Maize for Nutritional Security. In: Kumar S, Dikshit HK, Mishra GP, Singh A (eds) Biofortification of staple crops. Springer, Singapore, pp 147–174. https://doi.org/10.1007/978-981-16-3280-8_6
Abigail THY, Anna J, Elizabeth WC, Jane M, Gary F, Mary H (2023) Developing a consensus to support health and social care professionals and patients manage nutrition in the context of COVID-19 recovery. J Hum Nutr Diet. https://doi.org/10.1111/jhn.13163
Prasanna BM, Palacios-Rojas N, Hossain F, Muthusamy V, Menkir A, Dhliwayo T, Ndhlela T, San Vicente F, Nair SK, Vivek BS, Zhang X (2020) Molecular breeding for nutritionally enriched maize: status and prospects. Front Genet 10:1392
Hossain F, Zunjare RU, Muthusamy V, Kumar A, Madhavan J, Ikkurti G, Katral A, Talukder ZA, Chhabra RC, Chand G, Bhatt V, Gul I, Mishra SJ, Duo H, Dutta S, Gain N, Chauhan P, Maman S, Reddappa SB, Kasana RK (2023) Genetic improvement of specialty corn for nutritional quality traits. In: Wani SH, Dar ZA, Singh GP (eds) Maize improvement. Springer, Cham. https://doi.org/10.1007/978-3-031-21640-4_11
Muzhingi T, Palacios-Rojas N, Miranda A, Cabrera ML, Yeum KJ, Tang G (2017) Genetic variation of carotenoids, vitamin E and phenolic compounds in provitamin A biofortified maize. J Sci Food Agric 97:793–801
Das AK, Gowda MM, Muthusamy V, Zunjare RU, Chauhan HS, Baveja A, Bhatt V, Chand G, Bhat JS, Guleria SK, Saha S, Gupta HS, Hossain F (2021) Development of maize hybrids with enhanced vitamin-e, vitamin-a, lysine, and tryptophan through molecular breeding. Front Plant Sci 20:1427. https://doi.org/10.3389/fpls.2021.659381
Erenstein O, Jaleta M, Sonder K, Mottaleb K, Prasanna BM (2022) Global maize production, consumption and trade: trends and R&D implications. Food Secur 1–25
Baveja A, Muthusamy V, Panda KK, Zunjare RU, Das AK, Chhabra R, Mishra SJ, Mehta BK, Saha S, Hossain F (2021) Development of multinutrient-rich biofortified sweet corn hybrids through genomics-assisted selection of shrunken2, opaque2, lcyE and crtRB1 genes. J Appl Genet 62:419–429. https://doi.org/10.1007/s13353-021-00633-4
Duo H, Hossain F, Muthusamy V, Zunjare RU, Goswami R, Chand G, Mishra SJ, Chhabra R, Gowda MM, Pal S, Baveja A (2021) Development of sub-tropically adapted diverse provitamin-A rich maize inbreds through marker-assisted pedigree selection, their characterization and utilization in hybrid breeding. PLoS ONE 16:e0245497. https://doi.org/10.1371/journal.pone.0245497
Das AK, Muthusamy V, Zunjare RU, Chauhan HS, Sharma PK, Bhat JS, Hossain F (2019) Genetic variability-, genotype× environment interactions-and combining ability-analyses of kernel tocopherols among maize genotypes possessing novel allele of γ-tocopherol methyl transferase (ZmVTE4). J Cereal Sci 86:1–8. https://doi.org/10.1016/j.jcs.2018.12.018
Mene-Saffrane L, Pellaud S (2017) Current strategies for vitamin E biofortification of crops. Curr Opin Biotechnol 44:189–197
De Moura F, Miloff A, Boy E (2015) Retention of provitamin A carotenoids in staple crops targeted for biofortification in Africa: cassava, maize and sweet potato. Crit Rev Food Sci Nutr 55:1246–1269
Leenhardt F, Lyan B, Rock E, Boussard A, Potus J, Chanliaud E, Remesy C (2006) Wheat lipoxygenase activity induces greater loss of carotenoids than vitamin E during bread making. J Agric Food Chem 54:1710–1715
Martins PF, Batistella CB, Maciel-Filho R, Wolf-Maciel MR (2006) Comparison of two different strategies for tocopherols enrichment using a molecular distillation process. Ind Eng Chem Res 45:753–758
Diplock AT (1985) Vitamin E. Fat-soluble Vitamins. Diplock AT (Ed.), Heineman, London, 145–224
Choi J, Chon JK, Kim S, Shin W (2008) Conformational flexibility in mammalian 15S-lipoxygenase: reinterpretation of the crystallographic data. Proteins 70:1023–1032
Taleon V, Mugode L, Cabrera-Soto L, Palacios-Rojas N (2017) Carotenoid retention in biofortified maize using different post-harvest storage and packaging methods. Food Chem 232:60–66
Yu C, Hu Q, Wang H, Deng Z (2020) Comparison of 11 rice bran stabilization methods by analyzing lipase activities. J Food Process Preser v 44(4) Article e14370. https://doi.org/10.1111/jfpp.14370
Ogunola OF, Hawkins LK, Mylroie E, Kolomiets MV, Borrego E, Tang JD et al (2017) Characterization of the maize lipoxygenase gene family in relation to aflatoxin accumulation resistance. PLoS ONE 12(7):e0181265
Poca E, Rabinovitch-Chable H, Cook-Moreau J, Pages M, Rigaud M (1990) Lipoxygenases from Zea mays L. Purification and physicochemical characteristics. Biochim Biophys Acta 1045:107–114
Casey R (1997) Lipoxygenases and breadmaking. In: Angelino SAGF, Hamer RJ, van Hartingsfeld W, Heidekamp F, van derLugt JP,(eds). In: Proceedings of the first European symposium on enzymes and grain processing, pp 188–194
Wu Z, Robinson DS, Hughes RK, Casey R, Hardy D, West SI (1999) Co-oxidation of β-carotene catalyzed by soybean and recombinant pea lipoxygenase. J Agric Food Chem 47(12):4899–4906
Bollinedi H, Singh N, Gopala Krishnan S, Vinod KK, Bhowmick PK, Nagarajan M, Ellur RK, Singh AK (2022) A novel LOX3-null allele (lox3-b) originated in the aromatic basmati rice cultivars imparts storage stability to rice bran. Food Chem 369:130887. https://doi.org/10.1016/j.foodchem.2021.130887
Maman S (2021) Analysis of genetic variability for lipoxygenase activity and validation of lipoxygenase3 (LOX3) gene affecting retention of kernel tocopherols in maize during storage. M.Sc. dissertation, ICAR-Indian Agricultural Research Institute, New Delhi, India
Maman S, Hossain F, Katral A, Zunjare R, Gain N, Shashidhar BR, Kasana R, Sekhar J, Neeraja C, Yadava D, Muthusamy V (2023) Influence of storage duration on retention of kernel tocopherols in vte4-based biofortified maize genotypes. J Food Compos Anal 123:105626. https://doi.org/10.1016/j.jfca.2023.105626
Saha S, Walia S, Kundu A, Pathak N (2013) Effect of mobile phase on resolution of the isomers and homologues of tocopherols on a triacontyl stationary phase. Anal Bioanal Chem 405(28):9285–9295
Das AK, Jaiswal SK, Muthusamy V, Zunjare RU, Chauhan HS, Chand G, Saha S, Hossain F (2018) Molecular diversity and genetic variability of kernel tocopherols among maize inbreds possessing favourable haplotypes of γ-tocopherol methyl transferase (ZmVTE4). J Plant Biochem Biotechnol 28(3):253–262. https://doi.org/10.1007/s13562-018-0470-x
Niu L, Li D, Liu C, Liu F (2015) Comparison of lipoxygenase activity characteristics in aqueous extracts from milk-stage sweet corn and waxy corn. Food Sci Biotechnol 24:867–873
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Dutta S, Muthusamy V, Hossain F, Baveja A, Chhabra R, Jha SK, Yadava DK, Zunjare RU (2020) Analysis of genetic variability for retention of kernel carotenoids in sub-tropically adapted biofortified maize under different storage conditions. J Cereal Sci 93:102987. https://doi.org/10.1016/j.jcs.2020.102987
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork P, Jensen LJ (2019) STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 47:D607–613. https://doi.org/10.1093/nar/gky1131
Hossain F, Muthusamy V, Bhat JS, Zunjare RU, Kumar S, Prakash NR, Mehta BK (2022) Maize Breeding. In “Fundamentals of Field Crop Breeding”, D. K. Yadava (eds.). Nat. Singap. pp: 221–258. https://doi.org/10.1007/978-981-16-9257-4_4
Feng F, Wang Q, Liang C, Yang R, Li X (2015) Enhancement of tocopherols in sweet corn by marker-assisted backcrossing of ZmVTE4. Euphytica, 206: 513–521. https://doi.org/10.1007/s10681-015-1519-8
Xiao Y, Yu Y, Li G, Xie L, Guo X, Li J, Li YM, Hu J (2020) Genome-wide association study of vitamin E in sweet corn kernels. Crop J 8:341–350
Knecht K, Sandfuchs K, Kulling SE, Bunzel D (2015) Tocopherol and tocotrienols analysis in raw and cooked vegetables: a validated method with emphasis on sample preparation. Food Chem 169:20–27
Hakkansson B, Jagerstad M (1990) The effect of thermal inactivation of lipoxygenase on the stability of vitamin E in wheat. J Cereal Sci 12:177–185. https://doi.org/10.1016/S0733-5210(09)80099-4
Velisek J (2002) Chemie potravin 1. OSSIS, Tabor, pp 117–162
Katral A, Muthusamy V, Zunjare RU, Chhabra R, Maman S, Yadava DK, Hossain F (2022) Allelic variation in Zmfatb gene defines variability for fatty acids composition among diverse maize genotypes. Front Nutr 9:845255. https://doi.org/10.3389/fnut.2022.845255
Katral A, Hossain F, Gopinath I, Chand G, Mehta BK, Kamboj MC, Zunjare RU, Yadava DK, Muthusamy V (2023) Genetic dissection of embryo size and weight related traits for enhancement of kernel oil in maize. Plant Physiol Biochem 197:107668. https://doi.org/10.1016/j.plaphy.2023.107668
Boyes S, Perera C, Young H (1992) Kiwifruit lipoxygenase: preparation and characteristics. J Food Sci 57:1390–1394. https://doi.org/10.1111/j.1365-2621.1992.tb06866.x
Christensen SA, Nemchenko A, Borrego E, Murray I, Sobhy IS, Bosak L, DeBlasio S, Erb M, Robert CAM, Vaughn KA, Herrfurth C, Tumlinson J, Feussner I, Jackson D, Turlings TCJ, Engelberth J, Nansen C, Meeley R, Kolomiets MV (2013) The maize lipoxygenase, ZmLOX10, mediates green leaf volatile, jasmonate, and herbivore induced plant volatile production for defense against insect attack. TPJ 74:59–73
Acknowledgements
First Author is thankful to Indian Council of Agricultural Research (ICAR) for providing Junior Research Fellowship (JRF) during Master’s degree programme.
Funding
Authors acknowledge the Indian Council of Agricultural Research (ICAR) for providing financial support for the study through Consortia Research Platform on Biofortification in Selected Crops for Nutritional Security-Maize.
Author information
Authors and Affiliations
Contributions
SM: Investigation, Formal analysis, Roles/Writing - original draft; VM: Conceptualization, Resources, Supervision, Roles/Writing - original draft, Writing - review & editing; AK: Methodology, Software, Roles/Writing - original draft; RC: Methodology; NG: Methodology; SBR: Methodology; SD: Methodology; AUS: Resources; RUZ: Software; CNN: Conceptualization, Project administration; DKY: Conceptualization; FH: Conceptualization, Funding acquisition, Project administration, Resources, Supervision, Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no conflict of interest exists.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11033_2023_8820_MOESM2_ESM.tif
Supplementary Material 2. Fig. S2. Heat map of gene co-occurrence (a) and co-expression (b) among VTE4 and LOX proteins. cl364_1 = VTE4; LOX = LOX1; LOC542495 = LOX3; LOX4 = LOX4; LOX5 = LOX5; LOX6 = LOX6; L0 × 7 = LOX7; LOX8 = LOX8; Lox9 = LOX9; LOX10 = LOX10; LOX11 = LOX11; LOX12 = LOX12; GRMZM5G822593_P01 = LOX13
11033_2023_8820_MOESM3_ESM.docx
Supplementary Material 3. Table S1 The details of the vte4 and LOX genes and their proteins used for the protein-protein association studies
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maman, S., Muthusamy, V., Katral, A. et al. Low expression of lipoxygenase 3 (LOX3) enhances the retention of kernel tocopherols in maize during storage. Mol Biol Rep 50, 9283–9294 (2023). https://doi.org/10.1007/s11033-023-08820-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08820-8