Abstract
Background
Bacterial diseases are a huge threat to the production of tomatoes. During infection intervals, pathogens affect biochemical, oxidant and molecular properties of tomato. Therefore, it is necessary to study the antioxidant enzymes, oxidation state and genes involved during bacterial infection in tomato.
Methods and results
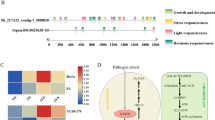
Different bioinformatic analyses were performed to conduct homology, gene promoter analysis and determined protein structure. Antioxidant, MDA and H2O2 response was measured in Falcon, Rio grande and Sazlica tomato cultivars. In this study, RNA Polymerase II (RNAP) C-Terminal Domain Phosphatase-like 3 (SlCPL-3) gene was identified and characterized. It contained 11 exons, and encoded for two protein domains i.e., CPDCs and BRCT. SOPMA and Phyre2, online bioinformatic tools were used to predict secondary structure. For the identification of protein pockets CASTp web-based tool was used. Netphos and Pondr was used for prediction of phosphorylation sites and protein disordered regions. Promoter analysis revealed that the SlCPL-3 is involved in defense-related mechanisms. We further amplified two different regions of SlCPL-3 and sequenced them. It showed homology respective to the reference tomato genome. Our results showed that SlCPL-3 gene was triggered during bacterial stress. SlCPL-3 expression was upregulated in response to bacterial stress during different time intervals. Rio grande showed a high level of SICPL-3 gene expression after 72 hpi. Biochemical and gene expression analysis showed that under biotic stress Rio grande cultivar is more sensitive to Pst DC 3000 bacteria.
Conclusion
This study lays a solid foundation for the functional characterization of SlCPL-3 gene in tomato cultivars. All these findings would be beneficial for further analysis of SlCPL-3 gene and may be helpful for the development of resilient tomato cultivars.




Similar content being viewed by others
Data Availability
The data will be made available on reasonable request to corresponding author.
Abbreviations
- PCR:
-
Polymerase Chain Reaction
- LB media:
-
Lysogeny Broth media
- BRCT:
-
Breast Cancer C Terminus
- CPDC:
-
Catalytic domain of C- terminal domain-like phosphatases
- CDKCs:
-
Cyclin dependent kinases
- Hpi:
-
Hours post infiltration
- H2O2 :
-
Hydrogen peroxide
- Bp:
-
Base pair
References
Balmant KM, Parker J, Yoo MJ et al (2015) Redox proteomics of tomato in response to Pseudomonas syringae infection. Hortic Res. https://doi.org/10.1038/hortres.2015.43. 2:
Quinet M, Angosto T, Yuste-Lisbona FJ et al (2019) Tomato Fruit Development and Metabolism. Front. Plant Sci. 10
Anzalone A, Mosca A, Dimaria G et al (2022) Soil and Soilless Tomato Cultivation promote different microbial Communities that provide New Models for Future Crop Interventions. Int J Mol Sci 23. https://doi.org/10.3390/ijms23158820
Arslan Ş, Arısoy H, Karakayacı Z (2022) The Situation of Regional Concentration of Tomato Foreign Trade in Turkey. Turkish J Agric - Food Sci Technol 10:280–289. https://doi.org/10.24925/turjaf.v10i2.280-289.4767
Li Y, Wang H, Zhang Y, Martin C (2018) Can the world’s favorite fruit, tomato, provide an effective biosynthetic chassis for high-value metabolites? Plant Cell Rep 37:1443–1450
Zhao J, Sauvage C, Zhao J et al (2019) Meta-analysis of genome-wide association studies provides insights into genetic control of tomato flavor. Nat Commun 10. https://doi.org/10.1038/s41467-019-09462-w
Cerda R, Avelino J, Gary C et al (2017) Primary and secondary yield losses caused by pests and diseases: Assessment and modeling in coffee. PLoS ONE 12. https://doi.org/10.1371/journal.pone.0169133
Buttimer C, McAuliffe O, Ross RP et al (2017) Bacteriophages and bacterial plant diseases. Front Microbiol 8
Thomas NC, Hendrich CG, Gill US et al (2020) The Immune receptor Roq1 confers resistance to the bacterial pathogens Xanthomonas, Pseudomonas syringae, and Ralstonia in Tomato. Front Plant Sci 11. https://doi.org/10.3389/fpls.2020.00463
Hamdoun S, Liu Z, Gill M et al (2013) Dynamics of defense responses and cell fate change during Arabidopsis-Pseudomonas syringae interactions. PLoS ONE 8. https://doi.org/10.1371/journal.pone.0083219
Chassot C, Nawrath C, Métraux JP (2007) Cuticular defects lead to full immunity to a major plant pathogen. Plant J 49:972–980. https://doi.org/10.1111/j.1365-313X.2006.03017.x
Lewis LA, Polanski K, de Torres-Zabala M et al (2015) Transcriptional dynamics driving MAMP-triggered immunity and pathogen effector-mediated immunosuppression in Arabidopsis leaves following infection with Pseudomonas syringae pv tomato DC3000. Plant Cell 27:3038–3064. https://doi.org/10.1105/tpc.15.00471
Derksen H, Rampitsch C, Daayf F (2013) Signaling cross-talk in plant disease resistance. Plant Sci 207:79–87
Bowler C, Fluhr R (2000) The role of calcium and activated oxygens as signals for controlling cross-tolerance. Trends Plant Sci 5:241–246. https://doi.org/10.1016/S1360-1385(00)01628-9
Lamb C, Dixon RA (1997) The oxidative burst in plant disease resistance. Annu Rev Plant Biol 48:251–275. https://doi.org/10.1146/annurev.arplant.48.1.251
Passardi F, Cosio C, Penel C, Dunand C (2005) Peroxidases have more functions than a swiss army knife. Plant Cell Rep 24:255–265
Watanabe S, Sato M, Sawada Y et al (2018) Arabidopsis molybdenum cofactor sulfurase ABA3 contributes to anthocyanin accumulation and oxidative stress tolerance in ABA-dependent and independent ways. Sci Rep 8. https://doi.org/10.1038/s41598-018-34862-1
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91:179–194
Nowogórska A, Patykowski J (2015) Selected reactive oxygen species and antioxidant enzymes in common bean after Pseudomonas syringae pv. Phaseolicola and Botrytis cinerea infection. Acta Physiol Plant 37. https://doi.org/10.1007/s11738-014-1725-3
Qiu JL, Zhou L, Yun BW et al (2008) Arabidopsis mitogen-activated protein kinase kinases MKK1 and MKK2 have overlapping functions in defense signaling mediated by MEKK1, MPK4, and MKS1. Plant Physiol 148:212–222. https://doi.org/10.1104/pp.108.120006
AbuQamar S, Chai MF, Luo H et al (2008) Tomato protein kinase 1b mediates signaling of plant responses to necrotrophic fungi and insect herbivory. Plant Cell 20:1964–1983. https://doi.org/10.1105/tpc.108.059477
Hirose Y, Manley JL (2000) RNA polymerase II and the integration of nuclear events. Genes Dev 14:1415–1429. https://doi.org/10.1101/GAD.14.12.1415
Koiwa H, Barb AW, Xiong L et al (2002) C-terminal domain phosphatase-like family members (AtCPLs) differentially regulate Arabidopsis thaliana abiotic stress signaling, growth, and development. Proc Natl Acad Sci U S A 99:10893–10898. https://doi.org/10.1073/pnas.112276199
Koiwa H, Bressan RA, Hasegawa PM (2006) Identification of plant stress-responsive determinants in arabidopsis by large-scale forward genetic screens. In: Journal of Experimental Botany. pp 1119–1128
Koiwa H, Hausmann S, Bang WY et al (2004) Arabidopsis C-terminal domain phosphatase-like 1 and 2 are essential Ser-5-specific C-terminal domain phosphatases. Proc Natl Acad Sci U S A 101:14539–14544. https://doi.org/10.1073/pnas.0403174101
Koiwa H (2006) Phosphorylation of RNA polymerase II C-terminal domain and plant osmotic-stress responses. In: Abiotic Stress Tolerance in Plants. Springer Netherlands, pp 47–57
Li F, Cheng C, Cui F et al (2014) Modulation of RNA polymerase II phosphorylation downstream of pathogen perception orchestrates plant immunity. Cell Host Microbe 16:748–758. https://doi.org/10.1016/j.chom.2014.10.018
Geourjon C, Deléage G (1995) Sopma: significant improvements in protein secondary structure prediction by consensus prediction from multiple alignments. Bioinformatics 11:681–684. https://doi.org/10.1093/bioinformatics/11.6.681
Kelley LA, Mezulis S, Yates CM et al (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10:845–858. https://doi.org/10.1038/nprot.2015.053
Tian W, Chen C, Lei X et al (2018) CASTp 3.0: computed atlas of surface topography of proteins. Nucleic Acids Res 46:W363–W367. https://doi.org/10.1093/nar/gky473
Li X, Sun Z, Shao S et al (2015) Tomato-Pseudomonas syringae interactions under elevated CO2 concentration: the role of stomata. J Exp Bot 66:307–316. https://doi.org/10.1093/jxb/eru420
MARKLUND S, MARKLUND G (1974) Involvement of the Superoxide Anion Radical in the autoxidation of Pyrogallol and a convenient assay for Superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Diao M, Ma L, Wang J et al (2014) Selenium promotes the growth and photosynthesis of Tomato Seedlings under Salt stress by enhancing chloroplast antioxidant Defense System. J Plant Growth Regul 33:671–682. https://doi.org/10.1007/s00344-014-9416-2
Bustin SA, Benes V, Garson JA et al (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622. https://doi.org/10.1373/clinchem.2008.112797
Yuan JS, Reed A, Chen F, Stewart CN (2006) Statistical analysis of real-time PCR data. BMC Bioinformatics 7. https://doi.org/10.1186/1471-2105-7-85
Stank A, Kokh DB, Fuller JC, Wade RC (2016) Protein binding Pocket Dynamics. Acc Chem Res 49:809–815. https://doi.org/10.1021/acs.accounts.5b00516
Bang W, Kim S, Ueda A et al (2006) Arabidopsis carboxyl-terminal domain phosphatase-like isoforms share common catalytic and interaction domains but have distinct in planta functions. Plant Physiol 142:586–594. https://doi.org/10.1104/pp.106.084939
Hussain A, Yun BW, Kim JH et al (2019) Novel and conserved functions of S-nitrosoglutathione reductase in tomato. J Exp Bot 70:4877–4886. https://doi.org/10.1093/JXB/ERZ234
Ray S, Mondal S, Chowdhury S, Kundu S (2015) Differential responses of resistant and susceptible tomato varieties to inoculation with Alternaria solani. Physiol Mol Plant Pathol 90:78–88. https://doi.org/10.1016/j.pmpp.2015.04.002
Muday GK, Brown-Harding H (2018) Nervous system-like signaling in plant defense herbivory induces rapid long-distance calcium signals through glutamate-l ike receptors. Science 361:1068–1069 (80-.)
Taverne YJ, Merkus D, Bogers AJ et al (2018) Reactive oxygen species: radical factors in the evolution of Animal Life: a molecular timescale from Earth’s earliest history to the rise of complex life. https://doi.org/10.1002/bies.201700158. BioEssays 40:
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Lobna H, Aymen EM, Hajer R et al (2017) Biochemical and plant nutrient alterations induced by Meloidogyne javanica and Fusarium oxysporum f.Sp.radicis lycopersici co-infection on tomato cultivars with differing level of resistance to M. javanica. Eur J Plant Pathol 148:463–472. https://doi.org/10.1007/s10658-016-1104-6
Vanderspool MC, Kaplan DT, McCollum TG, Wodzinski RJ (1994) Partial characterization of cytosolic superoxide dismutase activity in the interaction of Meloidogyne incognita with two cultivars of Glycine max. J Nematol 26:422–429
Zacheo G, Bleve-Zacheo T (1988) Involvement of superoxide dismutases and superoxide radicals in the susceptibility and resistance of tomato plants to Meloidogyne incognita attack. Physiol Mol Plant Pathol 32:313–322. https://doi.org/10.1016/S0885-5765(88)80026-2
Debona D, Rodrigues F, Rios JA, Nascimento KJT (2012) Biochemical changes in the leaves of wheat plants infected by Pyricularia oryzae. Phytopathology 102:1121–1129. https://doi.org/10.1094/PHYTO-06-12-0125-R
Shu P, Zhang S, Li Y et al Over-expression of SlWRKY46 in tomato plants increases susceptibility to Botrytis cinerea by modulating ROS homeostasis and SA and JA signaling. Elsevier
Yao N, Greenberg JT (2006) Arabidopsis ACCELERATED CELL DEATH2 modulates programmed cell death. Plant Cell 18:397–411. https://doi.org/10.1105/tpc.105.036251
Meena M, Zehra A, Dubey MK et al (2016) Comparative evaluation of biochemical changes in tomato (Lycopersicon esculentum Mill.) Infected by alternaria alternata and its toxic metabolites (TeA, AOH, and AME). Front Plant Sci 7. https://doi.org/10.3389/fpls.2016.01408
Gadjev I, Stone JM, Gechev TS (2008) Chap. 3: programmed cell death in plants. New Insights into Redox Regulation and the role of Hydrogen Peroxide. Int Rev Cell Mol Biol 270:87–144
Tran BQ, Jung S (2020) Modulation of chloroplast components and defense responses during programmed cell death in tobacco infected with Pseudomonas syringae. Biochem Biophys Res Commun 528:753–759. https://doi.org/10.1016/j.bbrc.2020.05.086
Silva LC, Debona D, Aucique-Pérez CE et al (2020) Physiological and antioxidant insights into common bean resistance to common bacterial blight. Physiol Mol Plant Pathol 111. https://doi.org/10.1016/j.pmpp.2020.101505
Acknowledgements
This study is part of the Ph.D. thesis work of Faisal Saeed who is doctoral candidate working under the supervision of Ufuk Demirel and Allah Bakhsh. The authors acknowledge the Ayhan Şahenk Foundation for providing fellowship during the doctoral study of FS. Authors are also grateful to Dr. Eminur Elçi, Associate Professor, Department of Plant Production and Technologies, Nigde Omer Halisdemir University for providing seeds of tomato cultivars.
Author information
Authors and Affiliations
Contributions
EA, AB and FS conceived the idea. FS performed the experiments, collected data and wrote the initial draft of manuscript. AB, EA and UD corrected the manuscript. MHH helped data interpretation. UD and AB supervised the overall study, and all authors approved the final draft.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest among authors.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saeed, F., Hashmi, M.H., Aksoy, E. et al. Identification and characterization of RNA polymerase II (RNAP) C-Terminal domain phosphatase-like 3 (SlCPL3) in tomato under biotic stress. Mol Biol Rep 50, 6783–6793 (2023). https://doi.org/10.1007/s11033-023-08564-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08564-5