Abstract
Background
By the time our study was completed, the chloroplast genomes of Syringa oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis had not been sequenced, and their genetic background was not clear.
The research content
In this study, the chloroplast genomes of Syringa oblata, S. pubescents subsp. Microphylla, S. reticulate subsp. Amurensis, and five other species of Syringa were sequenced for a comparative genomics analysis, inverted repeat (IR) boundary analysis, collinearity analysis, codon preference analysis and a nucleotide variability analysis. Differences in the complete chloroplast genomes of 30 species of Oleaceae were compared with that of S. oblata as the reference species, and Ginkgo biloba was used as the out group to construct the phylogenetic tree.
Results
The results showed that the chloroplast genomes of S. oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis were similar to those of other angiosperms and showed a typical four-segment structure, with full lengths of 155,569, 160,491, 155,419, and protein codes of 88, 95, and 87, respectively. Because the IR boundary of S. pubescents subsp. Microphylla was significantly expanded to the large single copy (LSC) region, resulting in complete replication of some genes in the IR region, the LSC region of S. pubescents subsp. Microphylla was significantly shorter than those of S. oblate and S. reticulate subsp. Amurensis. Similar to most higher plants, these three species have a preference for their codons ending with A/T.
Conclusions
We consider the genus Syringa to be a synphyletic group. The nucleotide variability and phylogenetic analyses showed that Syringa differentiated before Ligustrum and Ligustrum developed from Syringa. We propose removing the existing section division and directly dividing Syringa into five series.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Syringa plants occupy an important position in Chinese gardens. Syringa oblata, Syringa pubescents subsp. Microphylla, and Syringa reticulate subsp. Amurensis belong to Syringa of Oleaceae [1]. According to the relative length of corolla tubes and sepals, syringa is classified into Sect. Syringa and Sect. Ligustrina. Accord to the different leaf shape and inflorescence, Sect. Syringa be divided into Ser. Pinnatifoliae Rehd, Ser. Pubescentes (Schneid.) Lingelsh, Ser. Syringa and Ser. Villosae (Schneid.) Rehd. However, this classification is controversial, and whether Sect. Syringa should be an independent section or a parallel series with the Ser. Syringa is the main point of controversy.
Chloroplasts are the photosynthetic organelles of green plants, and have functions of synthesizing starch, fatty acids, pigments, and proteins [2]. Chloroplasts are generally considered the origin of endosymbiosis. Photosynthesizing prokaryotic cyanobacteria were absorbed into cells by primitive eukaryotes with phagocytic ability, and gradually transitioned from parasitism to symbiosis, and became a eukaryote organelle that allowed solar energy to be used autonomously, and the transformation from heterotrophic to autotrophic occurred. The chloroplast has a complete genetic system called the chloroplast genome. The chloroplast genome of gymnosperms is generally inherited from the paternal line [3], while the chloroplast genome of most angiosperms is inherited from the maternal line. About 20% of the genome may be inherited from both parents or from the paternal line [4,5,6].
The sequence and structure of the chloroplast genome are relatively conserved. The chloroplast genome of most plants has a double-stranded ring structure, including four regions, as two inverted repeat regions (IRa/IRb), one large single copy region (LSC) and one small single copy region (SSC) in four parts. Genome length is relatively fixed at 12–18 kb [7]. As a higher organelle of plants, the chloroplast genome has a stable structure that is highly conserved, with a large number of copies that can be easily extracted and purified. A series of characteristics determine the important position and significance of the chloroplast genome in plant phylogeny, species identification, and genetic transformation. The chloroplast genome can be used as important research material to study species phylogeny, which plays an important role in determining the genetic relationships among related species. Applying the chloroplast genome at the population level provides clues to study the timing of differentiation and intraspecific intensity. Analyses with nuclear genome data provide a more comprehensive understanding of plant pollen transmission, migration pathways, and species evolutionary dynamics.
In this study, we sequenced the complete chloroplast genomes of S. oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis and conducted comparative genomics of eight species of Oleaceae. We analyzed and combined the complete chloroplast genomes, LSC regions, SSC regions, IR regions, and introns of 30 species of Oleaceae published at the NCBI to construct a phylogenetic tree, and explored the phylogenetic relationships among the three species and their phylogeny in Oleaceae.
Methods
Plant materials
The plant materials required for this study were collected from the campus of Northeast Forestry University (45°43′ 16.83″ N, 126°38′ 2.04″ E) in July 2020. The fresh leaves of the three plants were quickly frozen in liquid nitrogen after collection, and then stored in a refrigerator at − 40 ℃. DNA extraction was carried out, and the extracted DNA was sent to Nanjing Jisihuiyuan Company for subsequent sequencing. The sequencing was submitted to NCBI database. The serial numbers of Syringa oblata, S. reticulate subsp Amurensis and S. pubescents subsp Microphylla is MT872639.1 MT872640.1 MT872641.1. The complete chloroplast genome sequences of the other five syringa species were obtained by NCBI database.
Genome assembly and annotation
This study adopted the whole genome shotgun strategy to construct libraries with different insert fragments [8] using second-generation sequencing technology (next-generation sequencing), based on the HiSeq sequencing platform, to construct the libraries using paired-end (PaIRed-end, PE) sequencing [9]. After DNA extraction, purification, library construction and sequencing. The off-machine data were saved in the PaIRed-end FASTQ format, and the off-machine data were further filtered to remove the reads with connectors and the low quality in the sequencing data to ensure the quality of the subsequent information analysis [10]. Spades 3.12 was used to de-assemble the sequencing data with the adapter sequence removed to obtain the final assembly sequence [11]. OGDRAW (https://chlorobox.mpimp-golm.mpg.de/OGDraw.html) was used to make a chloroplast genome map [12].
Genome feature analysis and comparative genome analytical method
Repetitive sequences are important genetic markers and are closely related to the origin and evolution of a species. Repetitive sequences have been divided into scattered repetition and tandem repetition types. Scattered repetitive sequences are repetitive sequences that are scattered in the genome. The scattered repetitions were analyzed using Reputer software [13], there is a special form of tandem repetition, which is also called SSR. SSRs often have natural polymorphisms, and cpSSR is the key component of a chloroplast genome repeat sequence analysis. The simple repetitive sequences were identified using MISA tools (parameters: 1-10 2-5 3-4 4-3 5-3 6-3) [14]. Codon preference was analyzed using the EMBOSS [15] (The European Molecular Biology Open Software Suite) and CodonW online programs, and the loss of genetic diversity reflected in the chloroplast genome was enhanced and showed a lower level of genetic diversity. Nucleotide diversity (π) was used to evaluate the level of population genetic diversity, using DnaSP software [16] to determine the local collinearity between genomes through multiple sequence alignment, and to perform similarity and a reanalysis of row, inversion, and other phenomena. The Mauve tool was used for the collinearity analysis. RAxML software was used to construct the maximum likelihood tree for the chloroplast genome [17].
Results
Genetic analysis
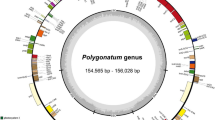
Similar to other angiosperms, the chloroplast genome structures of the eight species of Syringa listed were all closed circular double-stranded DNA, each containing four partitions, one LSC region, and one SSC region with two IRs (Fig. 1). The length of seven genomes, except that of S. pubescens subsp. Microphylla, was about 15.5 kb, and GC content was 37.9%. The lengths of the LSC, SSC, and IR regions were 8.6, 1.7, and 2.5 kb, respectively, and GC contents were 36%, 32%, and 43% respectively. The total number of encoded genes was 130–132. S. oblate and S. reticulate subsp. Amurensis showed highly consistent chloroplast genome characteristics. The total length of the chloroplast genome of the two species was about 15.5 kb, the length of each partition was similar, and the numbers of tRNAs and rRNAs were 37 and 8, respectively. Only one protein-coding gene difference was detected in the chloroplast genomes of these two species (Table S1). The chloroplast genome of S. oblata contained 88 protein-coding genes, which had one more acetyl-CoA carboxylase gene (accD) than S. reticulate subsp. Amurensis. The chloroplast genome of S. pubescens subsp. Microphylla was significantly longer than that of the other seven species at 160,491 bp, as well as the length of the four partitions. The largest difference was found in the length of the LSC region of S. pubescens subsp. Microphylla, which was 82,890 bp, while the length of the LSC region of the other seven species was about 86 kb, which was significantly shorter than the other listed species. There was little difference in the length of the SSC regions of these species. The IR region of S. pubescens subsp. Microphylla was 29,958 bp, which was longer than that of the other seven species. The IR regions of the other seven species were 25–26 kb. The shortening of the LSC region and expansion of the IR region of S. pubescens subsp. Microphylla caused rpl14, rpl16, rpl22, rps19, rps3, rps8, infA, and other genes originally located in the LSC region to fall into the IR region. Thus, resulting in 95 protein-coding genes and 140 total genes. The gene types in the chloroplast genome of S. pubescens subsp. Microphylla were the same as those in S. oblata. Only quantitative differences in the three types of genes were detected, such as large ribosomal subunits, small ribosomal subunits, and translational initiation factors. The chloroplast genome of S. oblata contained only one copy of the rpl14, Rpl16, rpl22, rps19, rps3, rps8, and infA genes, whereas S. pubescens subsp. Microphylla contained two. These differences appeared between trnH-GUG and ycf2, and no other differences were detected in genes at other positions (Table S2).
Expansion and contraction of the IR zone
Although the IR region of the chloroplast genome is the most conserved region, the contraction and expansion of its border region is a common phenomenon in the evolution of a chloroplast genome. This is also one of the most important factors for the length of the chloroplast genome. Among the eight species of Syringa, the borders of the LSC and IRb regions of seven species, except S. pubescens subsp. Microphylla, fell between the two genes, rps19 and rpl2. The rps11, rpl36, and infA genes were found near the boundary of the LSC and IRb regions in S. pubescens subsp. Microphylla, which was different from the other seven species. The rps19 gene of S. oblata and S. reticulate subsp. Amurensis was located on the boundary between the LSC and IRb regions, and both extended 2 bp into the IRb region. The ycf1 and ndhF genes were found near the borders of the SSC and IRb regions of S. oblata, S. pubescens subsp. Microphylla, and S. reticulate subsp. Amurensis. The lengths of the ycf1 gene extending into the SSC region were 74, 305, and 99 bp, respectively. The ndhF gene of S. pubescens subsp. Microphylla and S. reticulate subsp. Amurensis fell completely in the SSC region, and the ndhF gene of S. oblata extended 23 bp into the IRb region, becoming a pseudogene. The rpl2 and trnH genes were found near the boundary between the LSC and IRa regions of the remaining seven species, except S. pubescens subsp. Microphylla, and they were close to the boundary between the LSC and IRa regions. The trnH gene of Syringa wolfii was 14 bp from the boundary between the LSC and Ira regions, and the remaining six species exhibited a distance of 13 bp. Three genes, such as rps8, infA, and rpl36, were detected at the boundary between the LSC and IRa regions of S. pubescens subsp. Microphylla. All three genes were located in the IRa region, and rpl36 was 1 bp from the boundary. The trnH gene was also found in the LSC region of S. pubescens subsp. Microphylla but it was distant from the boundary between the LSC and Ira regions (Fig. 2).
Codon preference analysis
26,620, 27,428, and 25,723 codons were found in the complete chloroplast genomes of S. oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis, respectively. The most frequently used amino acid was Leu, and the smallest amino acid was Cys. All other amino acids have two or more codons, except Try, which has only one codon, and Arg, Leu and Ser have six codons. The codons of the eight species tended to end in A/U, which is the same as the codon preference of other angiosperm chloroplast genomes [18] (Fig. 3).
Repeat sequence analysis
Repetitive sequences are important genetic markers and are closely related to the origin and evolution of a species. Interspersed and tandem repetitive sequences are the two main types of repetitive sequences. Interspersed repetitive sequences generally include four types, namely forward repeats, reverse repeats, palindrome sequences, and complement sequences. There are differences in the numbers and types of scattered repeating sequences in different species. The total number of interspersed repetitive sequences of S. pubescents subsp. Microphylla (n = 164) and S. reticulate subsp. Amurensis (n = 163) was far more than the other listed species, and only 46 were found in S. oblata. Among the eight species of Syringa, only complementary sequences were found in the chloroplast genomes of S. microphylla and S. yunnanensis. The chloroplast genome of S. reticulate subsp. Amurensis contained only forward (n = 135) and palindromic sequences (n = 29), excluding IRs and complementary sequences. All four scattered repeats (111, 32, 10, and 10) were found in S. pubescents subsp. Microphylla. No complementary sequences were detected in the chloroplast genome of S. oblata. Twenty forward repeats and 25 palindromic sequences were observed, which contained only one IR (Table S3).
A special form of tandem repeat is a simple tandem repeat, also called a simple sequence repeat (SSR). SSRs often have natural polymorphisms. cpSSR is the key content of a chloroplast genome repeat sequence analysis. The types and numbers of SSR sites in S. oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis were studied. The results showed that the number of SSRs varied among the different species. Mononucleotide repeats account for the majority of SSRs, and these SSRs all appear in non-coding regions or protein-coding genes, and generally do not appear in tRNA or rRNA (Table S4). The numbers of mononucleotide SSRs in S. oblata, S. pubescents subsp. Microphylla, S. reticulate subsp. Amurensis were 35, 36 and 47, respectively. None of these three species contained pentanucleotide SSRs, and only S. pubescents subsp. Microphylla contained a hexanucleotides SSR. The SSRs of the three species were located in the protein coding or non-coding regions, and no SSR sites were found on tRNA or rRNA (Table S5). Among the eight species of Syringa, the SSRs were primarily located in the LSC region, with fewer distributed in the SSC and IR regions. Some species, such as S. oblata, S. pinnatifolia, and S. vulgaris, have no SSRs in the IR region. Totals of 43, 40, 52 SSRs were found in the LSC region, and four SSR sites were found in the SSC regions of S. oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis. No SSR was found in the IR of S. oblata, and one and five SSRs were found in the IRs of S. pubescents subsp. Microphylla and S. reticulate subsp. Amurensis, respectively (Fig. 4). The complex nucleotide SSRs of the three species were all located in the non-coding region of the LSC region, and two trinucleotide ((TTA)4(ATA)4) SSRs of S. reticulate subsp. Amurensis only appeared in the IRa/IRb region. The trinucleotide SSRs of S. oblate and S. pubescents subsp. Microphylla occurred in the LSC region, and the four hexanucleotide SSRs of S. pubescents subsp. Microphylla all appeared in the IRa/IRb region. No hexanucleotides were detected in the chloroplast genomes of the other two species. SSRs mainly appear in several genes, such as matK, rps16, atpF, rpoC2, ycf3, atpB, rps12, ycf1, ycf2, and rpl16. No SSRs were found in the ycf1, ycf2, and rpl16 genomes of lilac. No SSRs were found in the ycf2 and rpl16 genes of S. reticulate subsp. Amurensis.
Analysis of variations in the chloroplast genome
Many variations are found in chloroplast genome sequences. The variations in the genome are mainly divided into three categories: 1. single nucleotide variations, (usually called single nucleotide polymorphisms, that is, the difference in a single DNA base, referred to as a SNP); SNPs only involve the variation of a single base, which can be caused by transition or transversion of a single base, or by the insertion or deletion of bases; 2. small indels (insertion and deletion); that is, insertion or deletion of a small sequence < 50 bp at a certain position in the genome; 3. large structural variations (SVs), including those with lengths ≥ 50 bp, insertion or deletion of long fragments, shift inversion, copy number variations, and some more complicated forms of variation. The second and third types of variation are also called SVs to distinguish them from SNP variation. The analytical results are shown in Table S6 using S. oblata as the reference species. The maximum number of SNPs in S. reticulate subsp. Amurensis was 1,642, with 311 indels. In total, 372 indels were present in S. pubescens subsp. Microphylla, with 1588 SNPs, indicating that the two are quite different from the chloroplast genome of S. oblata, and their genetic relationship is relatively distant; the numbers of SNPs and indels in S. vulgaris were 171 and 49 respectively. In traditional taxonomy, S. reticulate subsp. Amurensis belongs to Sect. Ligustrina, whereas the other listed species belong to Sect. Syringa. S. vulgaris and S. oblata belong to Ser. Syringa, and S. pubescens subsp. Microphylla belongs to Ser. Pubescentes (Schneid.) Lingelsh. The results of variations in the chloroplast genome verified this traditional classification result (Table S6).
Collinearity analysis
A collinearity analysis is performed to determine the local collinearity blocks between genomes through multiple sequence comparisons, and analyze the similarities, rearrangements, and inversions of the collinearity blocks to illustrate the events that occur during species evolution. Blocks of the same color connected by the same line represent similar gene segments of different species. Organisms increase the length of gene segments to follow changes in the evolutionary process. The collinearity analysis of eight species of Syringa is shown in the Fig. 4, considering S. oblata as the benchmark species. Only one gene segment of S. persica was rearranged, and the gene segments of the other seven Syringa species increased or decreased significantly compared with those of S. oblata (Fig. 5).
Chloroplast variability analysis
Using S. oblata as the benchmark species, the Vista tool was used to conduct a genome-wide comparative analysis of 28 Oleaceae species. The results showed that the variation in the non-coding region was greater than that in the coding region, the SC region was larger than the IR region, and the RNA part of the gene was the most conserved. The genes were divided into three categories based on variations in the different genes from different species. The first parts of the genes were quite different among some species. For example, the accD, ycf1, and ycf2 genes of some species were quite different from those of lilac. The second parts of the genes were also quite different in all of the listed species, such as the ndhD and psaC genes. The third part was the more conserved genes, such as pasA, pasB, and other genes, which differed highly among the listed species. The first and second types of genes can be used for the development of genetic barcodes (Fig. 6).
Phylogenetic analysis
We use Ginkgo biloba as the out group and constructed a phylogenetic tree with the chloroplast genomes, CDS regions, introns, LSC regions, and IR region of S. oblata, S. pubescents subsp. Microphylla, S. reticulate subsp. Amurensis and 26 other species of Oleaceae published at the NCBI. The five phylogenetic trees all showed that Syringa differentiated before Ligustrum, indicating that Ligustrum was derived from Syringa (Fig. 7).
The phylogenetic tree of the CDS region was similar to the phylogenetic tree of the complete chloroplast genome, as both show that S. persica and S. pinnatifolia were sequentially separated from the whole of Oleaceae, indicating that the two are evolutionarily primitive. Ligustrum and some Syringa aggregated into one large branch, and the lower branches belonged to Ligustrum and Ser. Villosae (Schneid.) Rehd of Sect. Syringa, including Syringa wolfii. Syringa yunnanensis and Ser. Pubescentes (Schneid.) Lingelsh, juxtaposed with S. reticulata subsp. Amurensis, which belonged to Sect. Ligustrina Rupr. In other branches, Ser. Syringa represented by S. vulgaris and S. oblata was on the same branch with Fraxinus, Forsythia, and Osmanthus, and the relationship was distant from Ser. Pubescentes (Schneid.) Lingelsh represented by S. pubescens subsp. Microphylla, although the two both belonged to Sect. Syringa in the existing classification of Syringa.
The results of the evolutionary tree of introns and the LSC regions were similar. Forsythia was the first to differentiate. Syringa and Ligustrum aggregated on one branch, and several other genera aggregated on a large branch. Among the Syringa and Ligustrum branches, Syringa differentiated earlier than Ligustrum. The sections and series in Syringa were aggregated separately. Sect. Syringa was divided into groups before Sect. Ligustrina. The differentiation order of Sect. Syringa is Ser. Syringa, Ser. Pinnatifoliae Rehd, and Ser. Villosae (Schneid.) Rehd. Ser. Pubescentes (Schneid.) Lingelsh. In the IR zone of the evolutionary tree, Forsythia first differentiated and Syringa and Ligustrum converged into one branch. Ser. Pinnatifoliae Rehd and Ser. Syringa converged into one branch juxtaposed with other lines of Syringa and Ligustrum. In the lower branch, Ligustrum and Sect. Ligustrina were juxtaposed with Ser. Villosae (Schneid.) Rehd and Ser. Pubescentes (Schneid.) Lingelsh on one branch.
The five-part phylogenetic tree shows that Ligustrum has the closest relationship with Syringa in Oleaceae, and it maybe evolved from Syringa, which is older than Ligustrum. The existing systematic classification system within Syringa has certain problems, and the results support converting the genus Syringa into five series.
Discussion
The chloroplast genomes of different species differed in length and the number of encoded proteins, but the range of variation was rather small. The chloroplast genome of most angiosperms is relatively fixed at 12–17 kb in length and is divided into four partitions. The IR and SSC regions are relatively conserved, and the LSC region is the most variable. The number of genes encoding proteins was relatively fixed at about 85; four rRNAs were present, and GC% content was relatively fixed at about 38%. Studies have shown that some species of Amaryllidaceae [19] and Rosaceae [20] have similar genetic characteristics, such as the length of the chloroplast genome and the number of encoded proteins. The IR region is the most conserved region in the plant chloroplast genome. The length, structure, and boundary between the IR and SC regions are all highly conserved. Expansion and contraction of the IR region are important factors that change the length of the plant chloroplast genome, and they are generally divided into two types. Expansion and contraction of the IR region of most species are reflected in the small deviation in the IR/SC boundary within a few fixed genes. This deviation may cause some genes to become pseudogenes. Labiatae [21] and Vitis [22, 23] have φycf1 and φrps19 pseudogenes in the IR/SC boundary, and the IR/SSC boundary was more conserved than the IR/LSC boundary, indicating that the main direction of expansion of the IR region is towards the LSC area. The LSC/IRb boundary generally fell on or near rps19, rpl2, or rpl22. IRb/SSC generally fell on ycf1 or between ycf1 and ndhF, SSC/IRa generally fell on ycf1, and the IRa/LSC boundary generally fell on or near rps19, rpl2, rpl12, and trnH. The IRb/SSC boundary of Ricinus [24], Gossypium [25], Gossypium [22] and other species falls on or near the ycf1 gene, and the SSC/IRa boundary falls on the ycf1 gene. The IR/LSC boundaries of the chloroplast genomes of the above are different. The lcs/IRb boundary of Ricinus falls on rpl22, and the IRa/LSC boundary falls on or near rpl22 and trnH. The Gossypium and Vitis lcs/IRb boundary falls on rps19, and the IRa/LSC boundary falls between rps19 and trnH. In this case, expansion of the IR region usually only changes from a few to dozens of base pairs, and does not cause a change in the number of genes. The other is the IR boundary crossing to other genes. For example, expansion and contraction of the IR region of S. oblate and S. reticulate subsp. Amurensis allow the IR boundary to change tens or even a few bps on a few genes, while this region in S. pubescents subsp. Microphylla expanded to the LSC region into the rpl36 gene. Several genes originally located in the LSC region were included in the IR region, resulting in complete replication of the gene, and the total number of genes in the chloroplast genome changed accordingly.
cpSSR molecular markers were first developed by Powell in 1995 as a phylogenetic hotspot [26]. The density of the cpSSRs contained in different regions of the chloroplast genome is different because of the uneven distribution of molecular markers among different taxa. Other characteristics have become excellent material for phylogenetic research. SSRs contain multiple types of repetitive sequences, including single nucleotide, dinucleotide, trinucleotide, tetranucleotide, pentanucleotide, hexanucleotide, and complex nucleotide repetitive sequences. The results of published plant chloroplast genome analyses show that cpSSRs with a single nucleotide repeat sequence of more than eight bases are more common and more numerous in the non-coding regions of most plants. Jakobsson and Ebert reported that CpSSRs with a single-base repeat sequence of fewer than seven bases cannot be used to distinguish between species [27, 28]. The number of cpSSRs contained in different groups of species was also quite different. Studies have shown that some species of Nymphaeaceae and Cuscuta contain fewer cpSSRs, while some species of Cruciferae contain more cpSSRs [29].
Traditional systematic classification is based on the morphological characteristics of Syringa. Rehder is divided into two subgenera under Syringa (Sect. Syringa and Sect. Ligustrina) [30]. The current classification divides the genus Syringa into two subgenera, namely Sect. Syringa and Sect. Ligustrina, among which Sect. Syringa consists of Ser. Pinnatifoliae Rehd, Ser. Pubescentes (Schneid.) Lingelsh, Ser. Syringa, and Ser. Villosae (Schneid.) Rehd. Chen Jinyong [31] reported shape and multivariate analyses that the genus Syringa is divided into two section s, two series, 12 species, and 13 subspecies, among which the Sect. Ligustrina contains one species, three subspecies, and Sect. Syringa contains a terminal inflorescence system and a lateral inflorescence system and includes five species and five subspecies and six species and 5 subspecies, respectively. He Miao and Zhuo Lihuan selected 11 species of Syringa from northeastern China. After a phylogenetic analysis based on 25 morphological characteristics, Sect. Ligustrina belonged to Sect. Syringa, the division of the section was removed, and Syringa was directly divided into five series, namely Ser. Pinnatifoliae Rehd. Ser. Pubescentes (Schneid.) Lingelsh. Ser. Syringa, Ser. Villosae (Schneid.) Rehd, and Ser. Ligustrina Rupr. [32]. Gao Yan et al. studied the relationship between the leaf epidermis characteristics of seven wild species of Syringa and the environmental adaptation mechanism and systematics, and inferred that the relationships among Ser. Pinnatifoliae Rehd. Ser. Pubescentes (Schneid.) Lingelsh. Ser. Villosae (Schneid.) Rehd. are closer [33]. The results of Kim’s restriction fragment length polymorphism technology using cpDNA and Qin Xiangkun’s isozyme technology support division of the genus into two sections [34, 35]; Gao Hongxiao et al. used amplified fragment length polymorphism analysis to remove the division of the sections, and reported that Syringa should be directly divided into four series [36]. Li et al. reached a similar conclusion by studying the ITS and ETS of rDNA [37]. In the present study, the phylogenetic results were similar to the result of He Miao and Zhuo Li huan, and directly support dividing Syringa into five series.
Conclusion
In this study, the complete chloroplast genome sequences and annotations of S. oblata, S. pubescents subsp. Microphylla, and S. reticulate subsp. Amurensis were described and compared with five other species of Syringa. A comparative genomics analysis, IR region boundary analysis, collinearity analysis, and codon preference analysis were performed. Using S. oblata as the reference species, 30 species of Oleaceae were used as materials to analyze variability in the entire chloroplast genome. A phylogenetic tree was constructed using G. biloba as the out group, as well as the CDS regions, IR regions, LSC regions, and introns. The results support the existing classification of Oleaceae, sort out the phylogenetic relationships among the genera in Oleaceae, and sort out the group divisions within Syringa.
Data accessibility
The authors declare that we have submitted the sequence information to NCBI with the numbers MT872639.1 MT872640.1 MT872641.1. We have released the above data.
Abbreviations
- LSC:
-
Large single copy regions
- SSC:
-
Small single copy regions
- IR:
-
Inverted repeat regions
- CDS:
-
Coding sequence
- SSR:
-
Simple sequence repeat
- cpSSR:
-
Chloroplast simple sequence repeat
- mono:
-
Mononucleotide repeats
- di:
-
Dinucleotide repeats
- tri:
-
Trinucleotide repeats
- tetra:
-
Tetranucleotide repeats
- penta:
-
Pentanucleotide repeats
- hexa:
-
Hexanucleotides repeats
- complex:
-
Complexnucleotide repeats
- SNP:
-
Single nucleotide polymorphism;
- InDel:
-
Insertion or deletion of small DNA sequences < 50 bp
- Ts:
-
Number of SNPS converted;
- Tv:
-
Number of switched SNPS
References
Editorial Board of Flora of China (1993) Chinese Academy of Sciences Flora of China. Science Press, Beijing 61:63–81
Hagemann R (2004) The sexual inheritance of plant organelles. In: Daniell H, Chase C (eds) Molecular biology and biotechnology of plant organelles. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-3166-3_4
Hu YC, Zhang Q, Rao GY (2008) Occurrence of plastids in the sperm cells of caprifoliaceae: biparental plastid inheritance in angiosperms is unilaterally derived from maternal inheritance. Plant Cell Physiol 49.6:958–968
Zhang Q, Sodmergen (2010) Why does biparental plastid inheritance revive in angiosperms? J Plant Res 123(2):201–206
Corriveau J (1998) Rapid screening method to detect potential biparental inheritance of plastid DNA and results for over 200 angiosperms species. Am J Bot 75(10):1443–58
Lilly JW, Havey MJ, Jiang JJ (2001) Cytogenomic analyses reveal the structural plasticity of the chloroplast genome in higher plants. Plant Cell 13(2):245–254
Zhang T, Fang Y, Wang X et al (2012) The complete chloroplast and mitochondrial genome sequences of Boea hygrometrica: insights into the evolution of plant organellar genomes. PLoS ONE 7(1):e30531
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410
Blake JD, Cohen FE (2001) PaIRwise sequence alignment below the twilight zone. J Mol Biol 307(2):721–735
Coil D, Jospin G, Darling AE (2014) A5-MiSeq: an updated pipeline to assemble microbial genomes from Illumina MiSeq data. Bioinformatics 31(4):587–589
Mikkel S, Stinus L, Ludovic O (2016) Adapterremoval v2.2016: rapid adapter trimming, identification, and read merging. BMC Res Notes 9:88
Liu C et al (2012) An integrated web server for the annotation, visualization, analysis, and GenBank submission of completely sequenced chloroplast genome sequences. BMC Genomics 13:715
Kurtz S, Schleiermacher C (1999) REPuter: fast computation of maximal repeats in complete genomes. Bioinformatics 5:426–427
Langmead B, Trapnell C, Pop M, Salzberg SL (2019) Ultrafast and memory-efcient alignment of short DNA sequences to the human genome. Genome Biol 10:R25
Olson SA (2002) Emboss opens up sequence analysis. Brief Bioinform 1:87–91
Librado P, Rozas J (2009) DnaSPv5: sofware for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313
Clegg MT, Gaut BS, Learn GH et al (1994) Rates and patterns of chloroplast DNA evolution. Proc Nat Acad Sci 91(15):6795–6801
Zhang F, Wang T, Shu X, Wang N, Zhuang W, Wang Z (2020) Complete chloroplast genomes and comparative analyses of L. chinensis, L. anhuiensis, and L. aurea (Amaryllidaceae). Int J Mol Sci 21(16):5729
Xue S, Shi T, Luo W, Ni X, Gao Z (2019) Comparative analysis of the complete chloroplast genome among Prunus mume, P. armeniaca, and P. salicina. Horticult Res. https://doi.org/10.1038/s41438-019-0171-1
Shen LQ (2018) Complete chloroplast genomes of three Lamiaceae medicinal plants: comparativeand evolutionary analysis in Lamiaceae. Zhejiang University, Hangzhou
Yang Y, Jiao J, Fan X, Zhang Y, Liu C (2019) Complete chloroplast genome sequence and characteristics analysis of vitis ficifolia. Acta Horticult Sin 46(4):635–48
Wang YH (2018) Chloroplast genome sequences of Vitis structural organization and phylogeny analysis. Huazhong Agricultural University, Wuhan
Muraguri S, Xu W, Chapman M et al (2020) Intraspecific variation within castor bean (Ricinus communis L.) based on chloroplast genomes. Ind Crops Prod 155:179
Feng K (2103) Chloroplast genome sequence of ten species of Gossypium: structural organization and Phylogeny analysis. Chinese Academy of Agricultural Sciences, Beijing
Powell W, Morgante M, Mcdevitt R, Vendramin J (1995) Polymorphic simple sequence repeat regions in chloroplast genomes: applications to the population genetics of pines. Proc Natl Acad Sci 92:7759–7763
Jakobsson M, Sll T, Lind-Halldén C, Halldén C (2007) Evolution of chloroplast mononucleotide microsatellites in Arabidopsis thaliana. Theor Appl Genet 114(2):223–235
Ebert D, Peakall R (2009) A new set of universal de novo sequencing primers for extensive coverage of non-coding chloroplast DNA: new opportunities for phylogenetic studies and cpSSR discovery. Mol Ecol Resour 9:777–783
Li B, Liu HX (2012) Research advances in chloroplast simple sequence repeat (cpSSR). J Anhui Agricult 13:32–3343
Rehder (1945) Notes on some cultivated trees and shrubs. J Arnold Arbor 26:67–78
Chen JY, Zhang ZS, Hong DY (2008) Taxonomic revision of Syringa pinetorum complex (Oleaceae). J Syst Evolut 46(1):93–95
He M, Zhuo LH (2007) A cladistic analysis of Syringa from Northeastern China based on the morphologic characters. For Sci Technol 32(2):60–64
Gao Y, Cui HX, Shi L, Qu YY (2008) Characteristics of leaf epidermis of Syringa and the association with their environmental adaptation and systematics. Acta Bot Boreal-Occident Sin 03:475–484
Ki-Joong K, Robert KJ (1998) A chloroplast DNA phylogeny of lilacs (Syringa, Oleaceae): Plast omegroups show a strong correlation with crossing groups. Am J Bot 85(9):1338–1351
Qin XK (1996) The use of peroxidases in the systematics of Oleaceae. Aeta Botaulca Yunnanica 18(2):159–166
Gao HX, Yang K, Liu JB (2001) Analysis of the phylogenetic relationship of Syringa by AFLP technique. J China Agricult Univ 16(05):50–54
Li J, Alexander JH, Zhang D (2002) Paraphyletic Syringa (Oleaceae): evidence from sequences of nu clear ribosomal DNA ITS and E TS regions. Syst Bot 27(3):592–597
Funding
We gratefully acknowledge financial support for this research from Key Laboratory of Saline-alkali Vegetation Ecology Restoration (Northeast Forestry University), Ministry of Education (0924210201).
Author information
Authors and Affiliations
Contributions
This study was designed by CY and JS. Collection and identification of field material were performed by CY and QG. Paper writing and Data analysis were performed by KW and HZ. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This experiment does not involve human and animal experiments. The plant materials used in this study were collected from the campus of NORTHEAST Forestry University, and the collection process was in accordance with the regulations of the university and relevant local laws and regulations.
Consent to participate
This experiment does not involve human and animal experiments.
Consent for publication
All data and manuscripts are published with the consent of all authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, C., Wang, K., Zhang, H. et al. Analysis of the chloroplast genome and phylogenetic evolution of three species of Syringa. Mol Biol Rep 50, 665–677 (2023). https://doi.org/10.1007/s11033-022-08004-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-08004-w