Abstract
Background
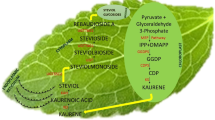
Stevia rebaudiana is a medicinal herb that accumulates non-caloric sweeteners called steviol glycosides (SGs) which are approximately 300 times sweeter than sucrose. This study used alginate (ALG) as an elicitor to increase steviol glycosides accumulation and elucidate gene transcription in the steviol glycosides biosynthesis pathway.
Methods and Results
To minimize the grassy taste associated with stevia sweeteners, plantlets were grown in complete darkness. ALG was applied to stevia plants grown in suspension culture with a Murashige and Skoog (MS) medium to determine its effect on SGs’ content and the transcription profile of SG-related genes using the HPLC and RT-qPCR methods, respectively. Treatment with alginate did not significantly affect plantlet growth parameters such as shoot number, dry and fresh weight. Rebaudioside A (Reb A) content increased approximately sixfold in the presence of 1g L-1 alginate and KS, KAH, and UGT74G1 genes showed significant up-regulation. When the concentration was increased to 2g L-1, the transcription of KO and UGT76G1, responsible for the conversion of stevioside to Reb A, was increased about twofold.
Conclusions
The current study proposes that adding alginate to the MS suspension medium can increase Reb A levels by altering the SG biosynthesize pathway’s transcription profile. The present experiment provides new insights into the biochemical and transcriptional response mechanisms of suspension-cultured stevia plants to alginate.




Similar content being viewed by others
References
Rippe JM, Angelopoulos TJ (2016) Relationship between Added Sugars Consumption and Chronic Disease Risk Factors: Current Understanding. Nutrients 8(11):697
Yadav SK, Guleria P (2012) Steviol Glycosides from Stevia: Biosynthesis Pathway Review and their Application in Foods and Medicine. Crit Rev Food Sci Nutr 52(11):988–998. https://doi.org/10.1080/10408398.2010.519447
Gardana C, Simonetti P, Canzi E, Zanchi R, Pietta P (2003) Metabolism of Stevioside and Rebaudioside A from Stevia rebaudiana Extracts by Human Microflora. J Agric Food Chem 51(22):6618–6622. https://doi.org/10.1021/jf0303619
Prakash I, Markosyan A, Bunders C (2014) Development of Next Generation Stevia Sweetener: Rebaudioside M. Foods 3(1):162–175. https://doi.org/10.3390/foods3010162
Kumar YA, S S, D. D SAP (2011) A review on the improvement of stevia [Stevia rebaudiana (Bertoni). Can J Plant Sci 91(1):1–27. https://doi.org/10.4141/cjps10086
Tadhani MB, Patel VH, Subhash R (2007) In vitro antioxidant activities of Stevia rebaudiana leaves and callus. J Food Compos Anal 20(3):323–329. https://doi.org/10.1016/j.jfca.2006.08.004
Hsieh M-H, Chan P, Sue Y-M, Liu J-C, Liang TH, Huang T-Y, Tomlinson B, Chow MSS, Kao P-F, Chen Y-J (2003) Efficacy and tolerability of oral stevioside in patients with mild essential hypertension: A two-year, randomized, placebo-controlled study. Clin Ther 25(11):2797–2808. https://doi.org/10.1016/S0149-2918(03)80334-X
Gregersen S, Jeppesen PB, Holst JJ, Hermansen K (2004) Antihyperglycemic effects of stevioside in type 2 diabetic subjects. Metabolism 53(1):73–76. https://doi.org/10.1016/j.metabol.2003.07.013
Ritu M, Nandini J (2016) Nutritional composition of Stevia rebaudiana, a sweet herb, and its hypoglycaemic and hypolipidaemic effect on patients with non-insulin dependent diabetes mellitus. J Sci Food Agric 96(12):4231–4234. https://doi.org/10.1002/jsfa.7627
Brandle JE, Rosa N (1992) Heritability for yield, leaf:stem ratio and stevioside content estimated from a landrace cultivar of Stevia rebaudiana. Can J Plant Sci 72(4):1263–1266. https://doi.org/10.4141/cjps92-159
McGarvey DJ, Croteau R (1995) Terpenoid metabolism. Plant Cell 7(7):1015–1026. https://doi.org/10.1105/tpc.7.7.1015
Brandle JE, Telmer PG (2007) Steviol glycoside biosynthesis. Phytochemistry 68(14):1855–1863. https://doi.org/10.1016/j.phytochem.2007.02.010
Etienne H, Berthouly M (2002) Temporary immersion systems in plant micropropagation. Planr Cell Tissue Organ Cult 69(3):215–231. https://doi.org/10.1023/A:1015668610465
Akimoto C, Aoyagi H, Tanaka H (1999) Endogenous elicitor-like effects of alginate on physiological activities of plant cells. Appl Microbiol Biotechnol 52(3):429–436. https://doi.org/10.1007/s002530051542
Xu S, Zhou W, Pottinger S, Baldwin IT (2015) Herbivore associated elicitor-induced defences are highly specific among closely related Nicotiana species. BMC Plant Biol 15(1):2. https://doi.org/10.1186/s12870-014-0406-0
Gilleta F, Roisin C, Fliniaux MA, Jacquin–Dubreuil A, Barbotin JN, Nava–Saucedo JE (2000) Immobilization of Nicotiana tabacum plant cell suspensions within calcium alginate gel beads for the production of enhanced amounts of scopolin. Enzym Microb Technol 26(2):229–234. https://doi.org/10.1016/S0141-0229(99)00138-6
Bayraktar M, Naziri E, Akgun IH, Karabey F, Ilhan E, Akyol B, Bedir E, Gurel A (2016) Elicitor induced stevioside production, in vitro shoot growth, and biomass accumulation in micropropagated Stevia rebaudiana. Plant Cell Tissue and Organ Culture (PCTOC) 127(2):289–300. https://doi.org/10.1007/s11240-016-1049-7
Thakur K, Ashrita, Sood A, Kumar P, Kumar D, Warghat AR (2021) : Steviol glycoside accumulation and expression profiling of biosynthetic pathway genes in elicited in vitro cultures of Stevia rebaudiana. In Vitro Cellular & Developmental Biology - Plant 57(2):214–224. 214–224 https://doi.org/10.1007/s11627-020-10151-3
Gerami M, Majidian P, Ghorbanpour A, Alipour Z (2020) Stevia rebaudiana Bertoni responses to salt stress and chitosan elicitor. Physiol Mol Biology Plants 26(5):965–974. https://doi.org/10.1007/s12298-020-00788-0
Cai Z, Kastell A, Mewis I, Knorr D, Smetanska I (2012) Polysaccharide elicitors enhance anthocyanin and phenolic acid accumulation in cell suspension cultures of Vitis vinifera. Plant Cell Tissue and Organ Culture (PCTOC) 108(3):401–409. https://doi.org/10.1007/s11240-011-0051-3
Thilip C, Mehaboob VM, Varutharaju K, Faizal K, Raja P, Aslam A, Shajahan A (2019) Elicitation of withaferin-A in hairy root culture of Withania somnifera (L.) Dunal using natural polysaccharides. Biologia 74(8):961–968
Jakinovich W, Moon C, Choi Y-H, Kinghorn AD (1990) Evaluation of Plant Extracts for Sweetness Using the Mongolian Gerbil. J Nat Prod 53(1):190–195. https://doi.org/10.1021/np50067a030
Hearn LK, Subedi PP (2009) Determining levels of steviol glycosides in the leaves of Stevia rebaudiana by near infrared reflectance spectroscopy. J Food Compos Anal 22(2):165–168. https://doi.org/10.1016/j.jfca.2008.10.008
Feng L, Gao M-J, Hou R-Y, Hu X-Y, Zhang L, Wan X-C, Wei S (2014) Determination of quality constituents in the young leaves of albino tea cultivars. Food Chem 155:98–104. https://doi.org/10.1016/j.foodchem.2014.01.044
Josse E-M, Halliday KJ (2008) Skotomorphogenesis: The Dark Side of Light Signalling. Curr Biol 18(24):R1144–R1146. https://doi.org/10.1016/j.cub.2008.10.034. R1144-R1146 DOI
Iqbal Z, Iqbal MS, Hashem A, Abd_Allah EF, Ansari MI (2021) Plant Defense Responses to Biotic Stress and Its Interplay With Fluctuating Dark/Light Conditions. Front Plant Sci 12(297). https://doi.org/10.3389/fpls.2021.631810
Murashige T, Skoog F (1962) A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Modi A, Litoriya N, Prajapati V, Rafalia R, Narayanan S (2014) Transcriptional profiling of genes involved in steviol glycoside biosynthesis in Stevia rebaudiana bertoni during plant hardening. Dev Dyn 243(9):1067–1073. https://doi.org/10.1002/dvdy.24157
Livak KJ, Schmittgen TD (2001) Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2 – ∆∆CT Method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Mamta RP, Pathania V, Gulati A, Singh B, Bhanwra RK, Tewari R (2010) Stimulatory effect of phosphate-solubilizing bacteria on plant growth, stevioside and rebaudioside-A contents of Stevia rebaudiana Bertoni. Appl Soil Ecol 46(2):222–229. https://doi.org/10.1016/j.apsoil.2010.08.008
Hajihashemi S, Geuns JMC, Ehsanpour AA (2013) Gene transcription of steviol glycoside biosynthesis in Stevia rebaudiana Bertoni under polyethylene glycol, paclobutrazol and gibberellic acid treatments in vitro. Acta Physiol Plant 35(6):2009–2014 2009–2014. https://doi.org/10.1007/s11738-013-1226-9
Lucho SR, do Amaral MN, Auler PA, Bianchi VJ, Ferrer M, Calderón AA, Braga EJB (2019) Salt Stress-Induced Changes in In Vitro Cultured Stevia rebaudiana Bertoni: Effect on Metabolite Contents, Antioxidant Capacity and Expression of Steviol Glycosides-Related Biosynthetic Genes. J Plant Growth Regul 38(4):1341–1353. https://doi.org/10.1007/s00344-019-09937-6
Lucho SR, do Amaral MN, Milech C, Ferrer M, Calderón AA, Bianchi VJ, Braga EJB (2018) Elicitor-Induced Transcriptional Changes of Genes of the Steviol Glycoside Biosynthesis Pathway in Stevia rebaudiana Bertoni. J Plant Growth Regul 37(3):971–985. https://doi.org/10.1007/s00344-018-9795-x
de Hoon MJ, Imoto S, Nolan J, Miyano S (2004) Open source clustering software. Bioinformatics 20(9):1453–1454. https://doi.org/10.1093/bioinformatics/bth078
Kharchouf S, Bouchador A, Drioiche A, Khiya Z, El Hilali F, Zair T (2017) : Étude phytochimique et évaluation de l’activité antioxydante de Stevia rebaudiana. Phytothérapie https://doi.org/10.1007/s10298-017-1163-7
Aoyagi H, Okada M, Akimoto C, Katsuyama H, Yoshida S, Kusakabe I, Tanaka H (1996) Promotion effect of alginate on chitinase production by Wasabia japonica. Biotechnol Tech 10(9):649–654. https://doi.org/10.1007/BF00168473
Dwivedi S, Alam A, Shekhawat GS (2016) Antioxidant response of Stevia rebaudiana (Bertoni) Bertoni (Angiosperms; Asteraceae) during developing phase of suspension cell culture. Plant Sci Today 3(2):115–123. https://doi.org/10.14719/pst.2016.3.2.227
Gupta P, Sharma S, Saxena S (2015) Biomass Yield and Steviol Glycoside Production in Callus and Suspension Culture of Stevia rebaudiana Treated with Proline and Polyethylene Glycol. Appl Biochem Biotechnol 176(3):863–874. https://doi.org/10.1007/s12010-015-1616-0
Mathur S, Shekhawat GS (2013) Establishment and characterization of Stevia rebaudiana (Bertoni) cell suspension culture: an in vitro approach for production of stevioside. Acta Physiol Plant 35(3):931–939. https://doi.org/10.1007/s11738-012-1136-2
Liu H, Zhang Y-H, Yin H, Wang W-X, Zhao X-M, Du Y-G (2013) Alginate oligosaccharides enhanced Triticum aestivum L. tolerance to drought stress. Plant Physiol Biochem 62:33–40. https://doi.org/10.1016/j.plaphy.2012.10.012
Zhang C, Howlader P, Liu T, Sun X, Jia X, Zhao X, Shen P, Qin Y, Wang W, Yin H (2019) Alginate Oligosaccharide (AOS) induced resistance to Pst DC3000 via salicylic acid-mediated signaling pathway in Arabidopsis thaliana. Carbohydr Polym 225:115221. https://doi.org/10.1016/j.carbpol.2019.115221
Kumar H, Kaul K, Bajpai-Gupta S, Kaul VK, Kumar S (2012) A comprehensive analysis of fifteen genes of steviol glycosides biosynthesis pathway in Stevia rebaudiana (Bertoni). Gene 492(1):276–284. https://doi.org/10.1016/j.gene.2011.10.015
Richman A, Swanson A, Humphrey T, Chapman R, McGarvey B, Pocs R, Brandle J (2005) Functional genomics uncovers three glucosyltransferases involved in the synthesis of the major sweet glucosides of Stevia rebaudiana. Plant J 41(1):56–67. https://doi.org/10.1111/j.1365-313X.2004.02275.x
Zheng J, Zhuang Y, Mao H-Z, Jang I-C (2019) Overexpression of SrDXS1 and SrKAH enhances steviol glycosides content in transgenic Stevia plants. BMC Plant Biol 19(1):1. https://doi.org/10.1186/s12870-018-1600-2
Singh G, Singh G, Singh P, Parmar R, Paul N, Vashist R, Swarnkar MK, Kumar A, Singh S, Singh AK et al (2017) Molecular dissection of transcriptional reprogramming of steviol glycosides synthesis in leaf tissue during developmental phase transitions in Stevia rebaudiana Bert. Sci Rep 7(1):11835. https://doi.org/10.1038/s41598-017-12025-y
Acknowledgements
The author would like to thank the phytochemistry department of Medicinal Plants and Drugs Research Institute of Shahid Beheshti University, Tehran, IRAN, for staff collaboration.
Funding
This research has not received any financial support.
Author information
Authors and Affiliations
Contributions
Alireza S. Tehranian carried out the experiment, laboratory analyses, statistical analysis and wrote the manuscript. Hossein Askari conceived the original idea and supervised the project. Hassan Rezadoost performed the HPLC analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Compliance with ethical standards
The research does not involve human participants.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tehranian, A.S., Askari, H. & Rezadoost, H. The effect of alginate as an elicitor on transcription of steviol glycosides biosynthesis pathway related key genes and sweeteners content in in vitro cultured Stevia rebaudiana. Mol Biol Rep 50, 2283–2291 (2023). https://doi.org/10.1007/s11033-022-07906-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07906-z