Abstract
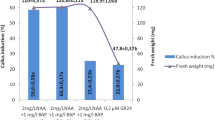
Enhanced production of steviol glycosides (SGs) was observed in callus and suspension culture of Stevia rebaudiana treated with proline and polyethylene glycol (PEG). To study their effect, yellow-green and compact calli obtained from in vitro raised Stevia leaves were sub-cultured on MS medium supplemented with 2.0 mg l−1 NAA and different concentrations of proline (2.5–10 mM) and PEG (2.5–10 %) for 2 weeks, and incubated at 24 ± 1 °C and 22.4 μmol m−2 s−1 light intensity provided by white fluorescent tubes for 16 h. Callus and suspension culture biomass (i.e. both fresh and dry weight content) was increased with 5 mM proline and 5 % PEG, while at further higher concentrations, they got reduced. Further, quantification of SGs content in callus (collected at 15th day) and suspension culture (collected at 10th and 15th day) treated with and without elicitors was analysed by HPLC. It was observed that chemical stress enhanced the production of SGs significantly. In callus, the content of SGs increased from 0.27 (control) to 1.09 and 1.83 % with 7.5 mM proline and 5 % PEG, respectively, which was about 4.0 and 7.0 times higher than control. However, in the case of suspension culture, the same concentrations of proline and polyethylene glycol enhanced the SG content from 1.36 (control) to 5.03 and 6.38 %, respectively, on 10th day which were 3.7 times and 4.7 times higher than control.


Similar content being viewed by others
References
Aazami, M. A., Torabi, M., & Jalili, E. (2010). In vitro response of promising tomato genotypes for tolerance to osmotic stress. African Journal of Biotechnology, 9(26), 4014–4017.
Ahmad, M. S. A., Javed, F., & Ashraf, M. (2007). Iso-osmotic effect of NaCl and PEG on growth, cations and free proline accumulation in callus tissue of two indica rice (Oryza sativa L.) genotypes. Plant Growth Regulation, 53, 53–63.
Almansouri, M., Kinet, J. M., & Lutts, S. (2001). Effect of salt and osmotic stresses on germination in durum wheat (Triticum durum Desf.). Plant and Soil, 231, 243–254.
Bhagwath, S. G., & Hjortso, M. A. (2000). Statistical analysis of elicitation strategies for thiarubrine: a production in hairy root cultures of Ambrosia artemisiifolia. Journal of Biotechnology, 80(2), 159–167.
Bohnert, H. J., & Shen, B. (1999). Transformation and compatible solutes. Scientia Horticulturae, 78, 237–260.
Bondarev, N., Reshetnyak, O., & Nosov, A. (2001). Peculiarities of diterpenoid steviol glycoside production in in vitro cultures of Stevia rebaudiana Bertoni. Plant Science, 161, 155–163.
Bonfill, M., Mangas, S., Moyano, E., Cusido, R. M., & Palazon, J. (2011). Production of centellosides and phytosterols in cell suspension cultures of Centella asiatica. Plant Cell Tissue and Organ Culture, 104, 61–67.
Canter, P. H., Thomas, H., & Ernst, E. (2005). Bringing medicinal plants into cultivation: opportunities and challenges for biotechnology. Trends Biotechnology, 23, 180–185.
Chan, L. K., See, K. S., Hoon, L. P., & Lim, B. P. (2008). Effect of plant growth regulators and subculture frequency on callus culture and the establishment of Melastoma malabathricum cell suspension culture for the production of pigments. Biotechnology, 7(4), 678–685.
Dey, A., Kundu, S., Bandyopadhya, A., & Bhattacharjee, A. (2013). Efficient micropropagation and chlorocholine chloride induced Stevioside production of Stevia rebaudiana Bertoni, Comptes Rendus. Biologies, 336, 17–28.
Geuns, J. M. C. (2003). Molecules of interest stevioside. Phytochemistry, 64, 913–921.
Gupta, P., Sharma, S., & Saxena, S. (2010). Callusing in Stevia rebaudiana (natural sweetener) for steviol glycoside production. International Journal of Agricultural and Biological Sciences, 1(1), 30–34.
Gupta, P., Sharma, S., & Saxena, S. (2014). Effect of salts (NaCl and Na2CO3) on callus and suspension culture of Stevia rebaudiana for steviol glycoside (natural sweetener) production. Applied Biochemistry and Biotechnology, Part A: Enzyme Engineering and Biotechnology, 172, 2894–2906.
Jacomini, E., Bertani, A., & Mapelli, S. (1988). Accumulation of polyethylene glycol 6000 and its effects on water content and carbohydrate level in water-stressed tomato plants. Canadian Journal of Botany, 66(5), 970–973.
Jaitak, V., Kaul, K., Kaul, V. K., Singh, V., Singh, B. (2011). Stevia rebaudiana-A natural substitute for sugar. In R. J. Singh (Ed.), Genetic resources, chromosome engineering, and crop improvement, medicinal plants. vol. 6, (pp. 885–910). CRC Press.
Janarthanam, B., Gopalakrishnan, M., & Sekar, T. (2010). Secondary metabolite production in callus cultures of Stevia rebaudiana Bertoni. Bangladesh Journal of Science and Industrial Research, 45(3), 243–248.
Kim, I., Yang, M., Lee, O., & Kang, S. (2011). The antioxidant activity and the bioactive compound content of Stevia rebaudiana water extracts. LWT - Food Science and Technology, 44, 1328–1332.
Lawlor, D. W. (1970). Absorption of polyethylene glycols by plants and their effects on plant growth. New Phytololist, 69, 501–514.
Lee-Parsons, C. W. T., & Rogce, A. J. (2006). Precursor limitations in methyl jasmonate-induced Catharanthus roseus cell cultures. Plant Cell Report, 25, 607–612.
Liu, J. Y., Guo, Z. G., & Zeng, Z. L. (2007). Improved accumulation of phenylethanoid glycosides by precursor feeding to suspension culture of Cistanche salsa. Biochemical Engineering Journal, 33(1), 88–93.
Masoumian, M., Arbakariya, A., Syahida, A., & Maziah, M. (2011). Flavonoids production in Hydrocotyle bonariensis callus tissues. Journal of Medicinal Plants Research, 5(9), 1564–1574.
Melis, M. S., Rocha, S. T., & Augusto, A. (2009). Steviol effect, a glycoside of Stevia rebaudiana, on glucose clearances in rats. Brazilian Journal of Biology, 69(2), 371–374.
Munir, N., & Aftab, F. (2009). The role of polyethylene glycol (PEG) pretreatment in improving sugarcane’s salt (NaCl) tolerance. Turkish Journal of Botany, 33, 407–415.
Navari-Izzo, M., & Rascio, N. (1999). Plant response to water deficit conditions. In M. Pessarakli (Ed.), Handbook of plant and crop stress (pp. 231–270). New York: Marcel Dekker Inc.
Okuma, E., Soeda, K., Tada, M., & Murata, Y. (2000). Exogenous proline mitigates the inhibition of growth of Nicotiana tabacum cultured cells under saline conditions. Soil Science and Plant Nutrition, 46, 257–263.
Ouyang, J., Wang, X. D., Zhao, B., & Wang, Y. C. (2005). Enhanced production of phenylethanoid glycosides by precursor feeding to cell culture of Cistanche deserticola. Process Biochemistry, 40, 3480–3484.
Patade, V. Y., Bhargava, S., & Suprasanna, P. (2012). Effects of NaCl and iso-osmotic PEG stress on growth, osmolytes accumulation and antioxidant defense in cultured sugarcane cells. Plant Cell Tissue and Organ Culture, 108, 279–286.
Rao, R. S., & Ravishankar, G. A. (2002). Plant cell cultures: chemical factories of secondary metabolites. Biotechnology Advances, 20(2), 101–153.
Riedel, H., Akumo, D. N., Thaw Saw, N. M. M., Kutuk, Onur., Neubauer, P., & Smetanska, I. (2012) Elicitation and precursor feeding influence phenolic acids composition in Vitis vinifera suspension culture. African Journal of Biotechnology, 11(12), 3000–3008.
Roberts, M. W., & Wright, J. T. (2012). Non-nutritive, low caloric substitutes for food sugars: clinical implications for addressing the incidence of dental caries and overweight/obesity. International Journal of Dentistry, 1–8.
Sakthivelu, G., Akitha, D. M. K., Giridhar, P., Rajasekaran, T., Ravishankar, G. A., & Nedev, T. (2008). Druoght-induced alterations in growth, osmotic potential and in vitro regeneration of soyabean cultivars. General Applied Plant Physiology, 34(1-2), 103–112.
Sato, K., Nakayama, M., & Shigeta, J. (1996). Culturing conditions affecting the production of anthocyanin in suspended cell cultures of strawberry. Plant Science, 113, 91–98.
See, S. K., Bhatt, A., & Keng, C. L. (2011). Effect of sucrose and methyl jasmonate on biomass and anthocyanin production in cell suspension culture of Melastoma malabathricum (Melastomaceae). Revista Biologia Tropical, 59(2), 597–606.
Shinde, A. N., Malpathak, N., & Fulzele, D. P. (2010). Determination of isoflavone content and antioxidant activity in Psoralea corylifolia L. callus cultures. Food Chemistry, 118, 128–132.
Siddiqui, M. H., Mohammad, F., Khan, M. N., Al-Whaibi, M. H., & Bahkali, A. H. A. (2010). Nitrogen in relation to photosynthetic capacity and accumulation of osmoprotectant and nutrients in brassica genotypes grown under salt stress. Agricultural Sciences in China, 9, 671–680.
Verbruggen, N., & Hermans, C. (2008). Proline accumulation in plants: a review. Amino Acids, 35, 753–759.
Whitmer, S., Heijden, V. R., & Verpoorte, R. (2002). Effect of precursor feeding on alkaloid accumulation by a tryptophan decarboxylase overexpressing transgenic cell line T22 of Catharanthus roseus. Journal of Biotechnology, 96(2), 193–203.
Xu, J., Yin, H. X., & Li, X. (2009). Protective effects of proline against cadmium toxicity in micropropagated hyperaccumulator Solanum nigrum L. Plant Cell Report, 28, 325–333.
Zia, M., Mannan, A., & Chaudhary, M. F. (2007). Effect of growth regulators and amino acids on Artemisinin production in the callus of Artemisia absinthium. Pakistan Journal of Botany, 39(3), 799–805.
Acknowledgments
The author would like to thank the Science and Society Division (now called SEED) of the Department of Science and Technology, India, and Ministry of Human Resource Development, India, for the financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 84 kb)
Rights and permissions
About this article
Cite this article
Gupta, P., Sharma, S. & Saxena, S. Biomass Yield and Steviol Glycoside Production in Callus and Suspension Culture of Stevia rebaudiana Treated with Proline and Polyethylene Glycol. Appl Biochem Biotechnol 176, 863–874 (2015). https://doi.org/10.1007/s12010-015-1616-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1616-0