Abstract
Background
HCC is among the most common cancer. Ganoderma lucidum (G.lucidum) has been essential in preventing and treating cancer. The Nrf2 signaling cascade is a cell protective mechanism against further damage, such as cancer development. This signaling pathway upregulates the cytoprotective genes and is vital in eliminating xenobiotics and reactive oxygen. This study aimed to show the potential cytotoxic activity of G. lucidum aqueous extract in HCC.
Methods and results
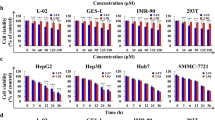
MTT assay was used to detect cell viability. Nrf2-related proteins were measured by western blotting, and the flow cytometry method assayed cell population in different cycle phases. Cell viability was 49% and 47% following G. lucidum extract at 100 µg/ml at 24 and 48 h treatments, respectively. G. lucidum extract (aqueous, 100 or 50 µg/ml) treatments for 24, 48, or 72 h were able to significantly change the cytoplasmic/nuclear amount of Nrf2 and HO-1, NQO1 protein levels. Moreover, at both concentrations, arrest of the G0/G1 cell cycle was stimulated in HCC.
Conclusions
The activation of the Nrf2 signaling pathways seems to be among the mechanisms underlining the protective and therapeutic action of G. lucidum against HCC.




Similar content being viewed by others
Data availability
All data are available from the corresponding author on reasonable request.
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- HBV:
-
Hepatitis B virus
- HCV:
-
Hepatitis C virus
- Keapl:
-
The Kelch-like ECH-associated protein
- Nrf2:
-
NF-E2-related factor 2
- ARE:
-
Antioxidant response element
- NQO1:
-
NAD(P)H quinone oxidoreductase 1
- HO1:
-
Heme oxygenase-1
- GLP:
-
Ganoderma lucidum polysaccharides
- IL:
-
Interleukin
References
Alves-Bezerra M, Cohen DE (2017) Triglyceride metabolism in the liver. Compr Physiol 8:1–8
Bakiri L, Hamacher R, Graña O et al (2017) Liver carcinogenesis by FOS-dependent inflammation and cholesterol dysregulation. J Exp Med 214:1387–1409
Ferlay J, Colombet M, Soerjomataram I et al (2021) Cancer statistics for the year 2020: an overview. Int J Cancer 149:778–789
Philips CA, Rajesh S, Nair DC et al (2021) Hepatocellular carcinoma in 2021: an exhaustive update. Cureus 13:e19274
Kole C, Charalampakis N, Tsakatikas S et al (2020) Immunotherapy for hepatocellular carcinoma: a 2021 update. Cancers 12:2859
Wang YL, Wu J, Li RX et al (2021) A double-edged sword: the Kelch-like ECH-associated protein 1-nuclear factor erythroid-derived 2-related factor 2-antioxidant response element pathway targeted pharmacological modulation in nonalcoholic fatty liver disease. Curr Opin Pharmacol 60:281–290
Vomund S, Schäfer A, Parnham MJ et al (2017) Nrf2, the master regulator of anti-oxidative responses. Int J Mol Sci 18(12):2772
Cui T, Lai Y, Janicki JS et al (2016) Nuclear factor erythroid-2 related factor 2 (Nrf2)-mediated protein quality control in cardiomyocytes. Front Biosci (Landmark edition) 21:192–202
Cykowiak M, Krajka-Kuźniak V (2022) Role of Nrf2 in pancreatic cancer. Antioxidants 11:98
Lee JH, Khor TO, Shu L et al (2013) Dietary phytochemicals and cancer prevention: Nrf2 signaling, epigenetics, and cell death mechanisms in blocking cancer initiation and progression. Pharmacol Ther 137:153–171
Jaganjac M, Milkovic L, Sunjic SB et al (2020) The NRF2, thioredoxin, and glutathione system in tumorigenesis and anticancer therapies. Antioxidants 9(11):1151
Pouremamali F, Pouremamali A, Dadashpour M et al (2022) An update of Nrf2 activators and inhibitors in cancer prevention/promotion. Cell Commun Signal 20:100
Sporn MB, Liby KT (2012) NRF2 and cancer: the good, the bad and the importance of context. Nat Rev Cancer 12:564–571
Venturella G, Ferraro V, Cirlincione F et al (2021) Medicinal mushrooms: bioactive compounds, use, and clinical trials. Int J Mol Sci 22:634
Jeitler M, Michalsen A, Frings D et al (2020) Significance of medicinal mushrooms in integrative oncology: a narrative review. Front Pharmacol 11:580656
Lee KH, Morris-Natschke SL, Yang X et al (2012) Recent progress of research on medicinal mushrooms, foods, and other herbal products used in traditional Chinese medicine. J Tradit Complement Med 2:84–95
El Sheikha AF (2022) Nutritional profile and health benefits of Ganoderma lucidum “Lingzhi, Reishi, or Mannentake” as functional foods: current scenario and future perspectives. Foods 11(7):1030
Papp V, Dima B, Wasser SP (2017) What is Ganoderma lucidum in the molecular era? Int J Med Mushrooms 19(7):575–593
Na K, Li K, Sang T et al (2017) Anticarcinogenic effects of water extract of sporoderm-broken spores of Ganoderma lucidum on colorectal cancer in vitro and in vivo. Int J Oncol 50:1541–1554
Yuen JW, Gohel MD (2005) Anticancer effects of Ganoderma lucidum: a review of scientific evidence. Nutr Cancer 53:11–17
Cao Y, Xu X, Liu S et al (2018) Ganoderma: a cancer immunotherapy review. Front Pharmacol 9:1217
Unlu A, Nayir E, Kirca O et al (2016) Ganoderma lucidum (Reishi Mushroom) and cancer. J buon 21:792–798
Ahmad MF (2018) Ganoderma lucidum: persuasive biologically active constituents and their health endorsement. Biomed Pharmacother 107:507–519
Sohretoglu D, Huang S (2018) Ganoderma lucidum polysaccharides as an anti-cancer agent. Anticancer Agents Med Chem 18:667–674
Roda E, Luca F, Locatelli CA et al (2020) From a medicinal mushroom blend a direct anticancer effect on triple-negative breast cancer: a preclinical study on lung metastases. Molecules 25(22):5400
Lin ZB, Zhang HN (2004) Anti-tumor and immunoregulatory activities of Ganoderma lucidum and its possible mechanisms. Acta Pharmacol Sin 25:1387–1395
Zhao R, Chen Q, He Y-m (2018) The effect of Ganoderma lucidum extract on immunological function and identify its anti-tumor immunostimulatory activity based on the biological network. Sci Rep 8:1–14
Chen S, Yong T, Zhang Y et al (2017) Anti-tumor and anti-angiogenic ergosterols from Ganoderma lucidum. Front Chem 5:85
Patel S, Goyal A (2012) Recent developments in mushrooms as anti-cancer therapeutics: a review. 3 Biotech 2:1–15
Liu YW, Gao JL, Guan J et al (2009) Evaluation of antiproliferative activities and action mechanisms of extracts from two species of Ganoderma on tumor cell lines. J Agric Food Chem 57:3087–3093
Liu X, Xu Y, Li Y et al (2020) Ganoderma lucidum fruiting body extracts inhibit colorectal cancer by inducing apoptosis, autophagy, and G0/G1 phase cell cycle arrest in vitro and in vivo. Am J Transl Res 12:2675–2684
Wu G, Qian Z, Guo J et al (2012) Ganoderma lucidum extract induces G1 cell cycle arrest, and apoptosis in human breast cancer cells. Am J Chin Med 40:631–642
Xu Z, Chen X, Zhong Z et al (2011) Ganoderma lucidum polysaccharides: immunomodulation and potential anti-tumor activities. Am J Chin Med 39:15–27
Ahmad MF (2020) Ganoderma lucidum: a rational pharmacological approach to surmount cancer. J Ethnopharmacol 260:113047
Meng J, Wang S-z, He J-z et al (2020) Ganoderic acid A is the effective ingredient of Ganoderma triterpenes in retarding renal cyst development in polycystic kidney disease. Acta Pharmacol Sin 41:782–790
Zakaria N, Mahdzir MA, Yusoff M et al (2018) Cytotoxic effects of Pinnatane A extracted from Walsura pinnata (Meliaceae) on human liver cancer cells. Molecules 23(11):2733
Nandi S, Chandra S, Sikder R et al (2019) Characterization and inception of a triterpenoid astrakurkurol, as a cytotoxic molecule on human hepatocellular carcinoma cells, Hep3B. J Agric Food Chem 67:7660–7673
Cao L, Jin H, Liang Q et al (2021) A new anti-tumor cytotoxic triterpene from Ganoderma lucidum. Nat Product Res. https://doi.org/10.1080/14786419.2021.1976175
Kaur V, Kumar M, Kumar A et al (2018) Pharmacotherapeutic potential of phytochemicals: implications in cancer chemoprevention and future perspectives. Biomed Pharmacother 97:564–586
Wu GS, Lu JJ, Guo JJ et al (2012) Ganoderic acid DM, a natural triterpenoid, induces DNA damage, G1 cell cycle arrest and apoptosis in human breast cancer cells. Fitoterapia 83:408–414
George BP, Chandran R, Abrahamse H (2021) Role of phytochemicals in cancer chemoprevention: insights. Antioxidants (Basel) 10(9):1455
Qin S, Hou DX (2016) Multiple regulations of Keap1/Nrf2 system by dietary phytochemicals. Mol Nutr Food Res 60:1731–1755
Lee S, Hu L (2020) Nrf2 activation through the inhibition of Keap1-Nrf2 protein-protein interaction. Med Chem Res 29:846–867
Esmaeilnejad B, Tavassoli M, Samiei A et al (2018) Evaluation of oxidative stress and antioxidant status, serum trace mineral levels and cholinesterases activity in cattle infected with Anaplasma marginale. Microb Pathog 123:402–409
Rani P, Lal MR, Maheshwari U et al (2015) Antioxidant potential of Lingzhi or Reishi Medicinal Mushroom, Ganoderma lucidum (higher basidiomycetes) cultivated on artocarpus heterophyllus sawdust substrate in India. Int J Med Mushrooms 17:1171–1177
Lee YH, Kim JH, Song CH et al (2016) Ethanol extract of Ganoderma lucidum augments cellular anti-oxidant defense through activation of Nrf2/HO-1. J Pharmacopuncture 19:59–69
Orrù C, Giordano S, Columbano A (2020) Nrf2 in neoplastic and non-neoplastic liver diseases. Cancers (Basel) 12:2932
Deshmukh P, Unni S, Krishnappa G et al (2017) The Keap1-Nrf2 pathway: promising therapeutic target to counteract ROS-mediated damage in cancers and neurodegenerative diseases. Biophys Rev 9:41–56
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295
Lee DY, Song MY, Kim EH (2021) Role of oxidative stress and Nrf2/KEAP1 signaling in colorectal cancer: mechanisms and therapeutic perspectives with phytochemicals. Antioxidants 10(5):74
Kladar NV, Gavarić NS, Božin BN (2016) Ganoderma: insights into anticancer effects. Eur J Cancer Prev 25:462–471
Li A, Shuai X, Jia Z et al (2015) Ganoderma lucidum polysaccharide extract inhibits hepatocellular carcinoma growth by downregulating regulatory T cells accumulation and function by inducing microRNA-125b. J Transl Med 13:100
Yu Y, Qian L, Du N et al (2017) Ganoderma lucidum polysaccharide enhances radiosensitivity of hepatocellular carcinoma cell line HepG2 through Akt signaling pathway. Exp Ther Med 14:5903–5907
Raghunath A, Sundarraj K, Arfuso F, et al. (2018) Dysregulation of Nrf2 in Hepatocellular Carcinoma: Role in Cancer Progression and Chemoresistance. Cancers (Basel) 10:
Yarmohammadi F, Rezaee R, Karimi G (2021) Natural compounds against doxorubicin-induced cardiotoxicity: a review on the involvement of Nrf2/ARE signaling pathway. Phytother Res 35:1163–1175
Hsieh TC, Wu JM (2011) Suppression of proliferation and oxidative stress by extracts of Ganoderma lucidum in the ovarian cancer cell line OVCAR-3. Int J Mol Med 28:1065–1069
Acknowledgements
None.
Funding
This work was supported financially by Ege Üniversitesi.
Author information
Authors and Affiliations
Contributions
RA, GA, TD, and AKKHAN: conceptualization, methodology, writing—original draft preparation. NR, SAH, GÖ, OSA, and AMA: writing—review and editing, validation, visualization. AA: conceptualization, formal analysis. SK: methodology, ınvestigation. AMÖ: supervision.
Corresponding authors
Ethics declarations
Conflict of interest
There is no scientific or medical committee membership or membership, consultancy, expertise, working status, or shareholding in any firm, which may be a potential conflict of interest of authors and/or family members concerning this work.
Ethical approval
The Ethical Committee of Ege Üniversitesi approved this study before the study began.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aslaminabad, R., Rahimianshahreza, N., Hosseini, S.A. et al. Regulation of Nrf2 and Nrf2-related proteins by ganoderma lucidum ın hepatocellular carcinoma. Mol Biol Rep 49, 9605–9612 (2022). https://doi.org/10.1007/s11033-022-07862-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07862-8