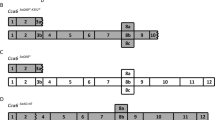
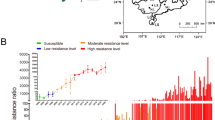
Abstract
Western flower thrips, Frankliniella occidentalis is an economically important agricultural pest. It causes damage by feeding and oviposition or indirectly by plant virus transmission. Australian F. occidentalis are resistant to many insecticides including spinosad and the related chemical spinetoram. Spinetoram resistance to F. occidentalis has been recently reported in three different Australian States, however, mechanisms conferring that resistance have not been investigated. To identify the mechanisms underlying resistance to spinetoram in F. occidentalis, we sequenced the genomic region of nicotinic acetylcholine receptor Foα6 in number of spinosad and spinetoram resistant field-populations. We found that a single nucleotide substitution (G to A) in exon 9 of the α6 subunit was present in resistant strains (G275E) and absent from susceptible. By examining field populations we consider the G275E mutation is the major cause of resistance to spinetoram in Australian F. occidentalis. We developed a real-time PCR diagnostic assay to quickly identify resistant alleles in field-populations. The method was used to test spinetoram resistant F. occidentalis collected from Australian cotton during the 2018–2019. Results show thrips tested carried the G275E mutation and the resistance allele was unusually widely distributed. The wide distribution of G275E mutation was not expected because spinetoram is not extensively used in Australian cotton. We speculate resistance may relate to extensive chemical use in crops nearby such as horticulture where thrips are often targeted for control. Our molecular diagnostic assay can provide timely and precise resistance frequency information that can support sustainable chemical use including spinetoram based IPM.


Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
He Z, Guo J-F, Reitz SR, Lei Z-R, Wu S-Y (2020) A global invasion by the thrip, Frankliniella occidentalis: current virus vector status and its management. Insect Sci 27(4):626–645. https://doi.org/10.1111/1744-7917.12721
Childers CC, Achor DS (1995) Thrips feeding and oviposition injuries to economic plants, subsequent damage and host responses to infestation. In: Parker BL, Skinner M, Lewis T (eds) Thrips biology and management, vol NATO ASI Series (Series A: Life Sciences). Springer, Boston, pp 31–51
Kirk WDJ, Terry LI (2003) The spread of the western flower thrips Frankliniella occidentalis (Pergande). Agric For Entomol 5(4):301–310. https://doi.org/10.1046/j.1461-9563.2003.00192.x
Malipatil MB, Postle AC, Osmelak JA, Hill M, Moran J (1993) 1st record of Frankliniella occidentalis (pergande) in Australia (thysanoptera, thripidae). J Aust Entomol Soc 32:378–378
Herron G, Gullick G (1998) Insecticide resistance in western flower thrips Frankliniella occidentalis (Pergande) in Australia. Paper presented at the Sixth Australian Applied Entomological Research Conference, 29th September–2nd October, The University of Queensland, Brisbane, Australia
Herron G, Gullick G (2001) Insecticide resistance in Australian populations of Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) causes the abandonment of pyrethroid chemicals for its control. Gen Appl Entomol 30:21–26
Herron GA, James TM (2005) Monitoring insecticide resistance in Australian Frankliniella occidentalis Pergande (Thysanoptera: Thripidae) detects fipronil and spinosad resistance. Aust J Entomol 44:299–303. https://doi.org/10.1111/j.1440-6055.2005.00478.x
Herron GA, James TM (2007) Insecticide resistance in Australian populations of western flower thrips Franliniella occidentalis (Pergande) (Thysanoptera: Thripidae). Gen Appl Entomol 36:1–6
Herron G, Langfield B (2011) An increase in spinosad resistance detected in Australian Frankliniella occidentalis (Pergandae) (Thipidae, Thysanoptera). Gen Appl Entomol 40:7–8
Herron GA, Gunning RV, Cottage ELA, Borzatta V, Gobbi C (2014) Spinosad resistance, esterase isoenzymes and temporal synergism in Frankliniella occidentalis (Pergande) in Australia. Pestic Biochem Physiol 114:32–37. https://doi.org/10.1016/j.pestbp.2014.07.006
AVPMA PUBCRIS database. ‘Success Neo Insecticide’ label (2019) https://www.infopest.com.au/. Accessed 14th Jan 2019
Sparks TC, Thompson GD, Kirst HA, Hertlein MB, Larson LL, Worden TV, Thibault ST (1998) Biological activity of the spinosyns, new fermentation derived insect control agents, on tobacco budworm (Lepidoptera: Noctuidae) larvae. J Econ Entomol 91(6):1277–1283. https://doi.org/10.1093/jee/91.6.1277
Mertz FP, Yao RC (1990) Saccharopolyspora spinosa sp. nov. isolated from soil collected in a sugar mill rum still. Int J Syst Evol Microbiol 40(1):34–39. https://doi.org/10.1099/00207713-40-1-34
Sparks TC, Crouse GD, Dripps JE, Anzeveno P, Martynow J, Deamicis CV, Gifford J (2008) Neural network-based QSAR and insecticide discovery: spinetoram. J Comput Aided Mol Des 22(6–7):393–401. https://doi.org/10.1007/s10822-008-9205-8
Dripps J, Olson B, Sparks T, Crouse G (2008) Spinetoram: how artificial intelligence combined natural fermentation with synthetic chemistry to produce a new spinosyn insecticide. Plant Health Prog. http://www.plantmanagementnetwork.org/pub/php/perspective/2008/spinetoram/
Langfield K, Woolley L, Learmonth S, Herron G (2018) Spinetoram resistance detected in western flower thrips Frankliniella occidentalis (Pergande) following a control failure. Gen Appl Entomol 46:43–45
Langfield K, Nguyen D, Annetts R, Herron G (2019) Spinetoram resistance detected in Australian western flower thrips Frankliniella occidentalis (pergande) from Queensland and Victoria. Gen Appl Entomol 47:13–16
Perry T, McKenzie JA, Batterham P (2007) A D alpha 6 knockout strain of Drosophila melahogaster confers a high level of resistance to spinosad. Insect Biochem Mol Biol 37(2):184–188. https://doi.org/10.1016/j.ibmb.2006.11.009
Hsu JC, Feng HT, Wu WJ, Geib SM, Mao CH, Vontas J (2012) Truncated transcripts of nicotinic acetylcholine subunit gene Bdα6 are associated with spinosad resistance in Bactrocera dorsalis. Insect Biochem Mol Biol 42(10):806–815. https://doi.org/10.1016/j.ibmb.2012.07.010
Rinkevich FD, Chen M, Shelton AM, Scott JG (2010) Transcripts of the nicotinic acetylcholine receptor subunit gene Pxylα6 with premature stop codons are associated with spinosad resistance in diamondback moth, Plutella xylostella. Invert Neurosci 10(1):25–33. https://doi.org/10.1007/s10158-010-0102-1
Ureña E, Guillem-Amat A, Couso-Ferrer F, Beroiz B, Perera N, López-Errasquín E, Castañera P, Ortego F, Hernández-Crespo P (2019) Multiple mutations in the nicotinic acetylcholine receptor Ccα6 gene associated with resistance to spinosad in medfly. Sci Rep 9(1):2961. https://doi.org/10.1038/s41598-019-38681-w
Wan YR, Yuan GD, He BQ, Xu BY, Xie W, Wang SL, Zhang YJ, Wu QJ, Zhou XG (2018) Focc alpha 6, a truncated nAChR subunit, positively correlates with spinosad resistance in the western flower thrips, Frankliniella occidentalis (Pergande). Insect Biochem Mol Biol 99:1–10. https://doi.org/10.1016/j.ibmb.2018.05.002
Puinean AM, Lansdell SJ, Collins T, Bielza P, Millar NS (2013) A nicotinic acetylcholine receptor transmembrane point mutation (G275E) associated with resistance to spinosad in Frankliniella occidentalis. J Neurochem 124(5):590–601. https://doi.org/10.1111/jnc.12029
Bao WX, Narai Y, Nakano A, Kaneda T, Murai T, Sonoda S (2014) Spinosad resistance of melon thrips, Thrips palmi, is conferred by G275E mutation in α6 subunit of nicotinic acetylcholine receptor and cytochrome P450 detoxification. Pestic Biochem Physiol 112:51–55. https://doi.org/10.1016/j.pestbp.2014.04.013
Silva WM, Berger M, Bass C, Williamson M, Moura DMN, Ribeiro LMS, Siqueira HAA (2016) Mutation (G275E) of the nicotinic acetylcholine receptor alpha 6 subunit is associated with high levels of resistance to spinosyns in Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Pestic Biochem Physiol 131:1–8. https://doi.org/10.1016/j.pestbp.2016.02.006
Hiruta E, Aizawa M, Nakano A, Sonoda S (2018) Nicotinic acetylcholine receptor α6 subunit mutation (G275V) found in a spinosad-resistant strain of the flower thrips, Frankliniella intonsa (Thysanoptera: Thripidae). J Pestic Sci 43(4):272–276. https://doi.org/10.1584/jpestics.D18-007
Mota-Sanchez D, Wise JC (2020) The arthropod pesticide resistance database. Michigan State University. On-line at: http://www.pesticideresistance.org. Accessed 15 July 2020
Abbott WS (1925) A method of computing the effectiveness of an insecticide. J Econ Entomol 18(2):265–267. https://doi.org/10.1093/jee/18.2.265a
Edwards AW (2008) G. H. Hardy (1908) and Hardy-Weinberg equilibrium. Genetics 179(3):1143–1150. https://doi.org/10.1534/genetics.104.92940
Bielza P, Quinto V, Contreras J, Torne M, Martin A, Espinosa PJ (2007) Resistance to spinosad in the western flower thrips, Frankliniella occidentalis (Pergande), in greenhouses of south-eastern Spain. Pest ManagSci 63(7):682–687. https://doi.org/10.1002/ps.1388
Bielza P, Quinto V, Fernandez E, Gravalos C, Contreras J (2007) Genetics of spinosad resistance in Frankliniella occidentalis (Thysanoptera: Thripidae). J Econ Entomol 100(3):916–920. https://doi.org/10.1603/0022-0493(2007)100[916:gosrif]2.0.co;2
Okuma DM, Bernardi D, Horikoshi RJ, Bernardi O, Silva AP, Omoto C (2018) Inheritance and fitness costs of Spodoptera frugiperda (Lepidoptera: Noctuidae) resistance to spinosad in Brazil. Pest Manag Sci 74(6):1441–1448. https://doi.org/10.1002/ps.4829
Chen Y, Bogema DR, Barchia IM, Herron GA (2014) Quantification of the pirimicarb resistance allele frequency in pooled cotton aphid (Aphis gossypii glover) samples by taqmansnp genotyping assay. PLoS One 9(3):e91104. https://doi.org/10.1371/journal.pone.0091104
Corteva Agrisceince™, Delegate® Insecticide. (2019) https://www.corteva.com.au/products-and-solutions/cropprotection/delegate.html. Accessed 17th Sept 2019
Acknowledgements
This work was funded in part by the Australian Cotton Research and Development Corporation (DAN1507) with in-kind contributions from the New South Wales Department of Primary Industries.
Funding
This work was funded by the Australian Cotton Research and Development Corporation (DAN1507) with in-kind contributions from the New South Wales Department of Primary Industries.
Author information
Authors and Affiliations
Contributions
YC, GH and DN conceived and designed the experiments. DN and RG performed the experiments. YC, DN and RG analysed the data. GH contributed reagents/materials/analysis-tools via Cotton Research and Development Corporation funded DAN1507. YC, DN and GH wrote the paper. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, Y., Nguyen, D.T., Gupta, R. et al. Mutation (G275E) of nAChR subunit Foα6 associated with spinetoram resistance in Australian western flower thrips, Frankliniella occidentalis (Pergande). Mol Biol Rep 48, 3155–3163 (2021). https://doi.org/10.1007/s11033-021-06372-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06372-3