Abstract
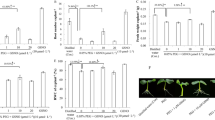
Although nitric oxide (NO) is a key regulatory molecule in plants, its function in plants under conditions of simulated acid rain (SAR) has not been fully established yet. In this study, exogenous sodium nitroprusside (SNP) at three different concentrations were applied to mung bean seedlings. Malondialdehyde (MDA), NO, hydrogen peroxide (H2O2), antioxidant enzyme activities, and nitrate reductases (NR) were measured. Real time PCR was used to measure the NR expression. Compared to the control, the NR activity and NO content under the pH 2 SAR decreased by 79% and 85.6% respectively. Meanwhile, the SAR treatment reduced the activities of superoxide dismutase (SOD), peroxidase (POD), ascorbate peroxidase (APX), while increased MDA content. Application of SNP could potentially reverse the adverse impact of SAR, depending on its concentration. For plants under the pH 2 SAR and 0.25 mM SNP condition, the activities of SOD, POD, APX increased by 123%, 291%, and 135.7% respectively, meanwhile, MDA concentration decreased by 43%, NR activities increased by 269%, and NO concentration increased by 123.6% compared with plants undergoing only pH 2 SAR. The relative expression of the NR1 gene was 2.69 times higher than that of pH 2 SAR alone. Overall, the application of 0.25 mM SNP eliminated reactive oxygen species (ROS) by stimulating antioxidant enzyme activities, reducing oxidative stress and mitigating the toxic effects of SAR on mung bean seedlings. This research provides a foundation for further research on the mechanism of NO on plants under SAR conditions.




Similar content being viewed by others
References
Abbasi T, Poornima P, Kannadasan T (2013) Acid rain: past, present, and future. Int J Environ Eng 5(3):229–272. https://doi.org/10.1504/IJEE.2013.054703
Qiao F, Zhang XM, Liu X, Chen J, Hu WJ, Liu TW, Liu JY, Zhu CQ, Kabir G, Zhu XY, Zheng HL (2018) Elevated nitrogen metabolism and nitric oxide production are involved in arabidopsis resistance to acid rain. Plant Physiol Biochem 127:238–247. https://doi.org/10.1016/j.plaphy.2018.03.025
Sun X, Wang Y, Li HY, Yang XQ, Sun L, Wang XF, Wang T, Wang WX (2016) Organic acids in cloud water and rainwater at a mountain site in acid rain areas of south China. Environ Sci Pollut Res 23(10):9529–9539. https://doi.org/10.1007/s11356-016-6038-1
Roseline X, Keshavkant S (2017) Acid rain-induced oxidative stress regulated metabolic interventions and their amelioration mechanisms in plants. Biologia 72(12):1387–1393. https://doi.org/10.1515/biolog-2017-0171
Erwann A, Marc G, Beatrice G, Gwendal C, Loic R (2013) Nitric oxide implication in the control of seed dormancy and germination. Front Plant Sci 4:346. https://doi.org/10.3389/fpls.2013.00346
Groß F, Durner J, Gaupels F (2013) Nitric oxide, antioxidants and prooxidants in plant defence responses. Front Plant Sci 4:419. https://doi.org/10.3389/fpls.2013.00419
Siddiqui MH, Al Whaibi MH, Basalah MO (2011) Role of nitric oxide in tolerance of plants to abiotic stress. Protoplasma 248(3):447–455. https://doi.org/10.1007/s00709-010-0206-9
Asgher M, Per TS, Masood A, Fatma M, Freschi L, Corpas FJ, Khan NA (2017) Nitric oxide signaling and its crosstalk with other plant growth regulators in plant responses to abiotic stress. Environ Sci Pollut Res 24(3):2273–2285. https://doi.org/10.1007/s11356-016-7947-8
Du YJ, Wei ML, Reddy KR, Liu ZP, Jin F (2014) Effect of acid rain pH on leaching behavior of cement stabilized lead-contaminated soil. J Hazard Mater 271:131–140. https://doi.org/10.1016/j.jhazmat.2014.02.002
Chen YE, Mao JJ, Sun LQ, Huang B, Ding CB, Gu Y, Liao JQ, Hu C, Zhang ZW, Yuan S, Yuan M (2018) Exogenous melatonin enhances salt stress tolerance in maize seedlings by improving antioxidant and photosynthetic capacity. Physiol Plant 164(3):349–363. https://doi.org/10.1111/ppl.12737
Chen YE, Yuan S, DuJ B, Xu MY, Zhang ZW, Lin HH (2009) Phosphorylation of photosynthetic antenna protein CP29 and photosystem II structure changes in monocotyledonous plants under environmental stresses. Biochemistry 48(41):9757–9763. https://doi.org/10.1021/bi901308x
Ren XQ, Zhu JZ, Liu HY, Xu X, Liang CJ (2018) Response of antioxidative system in rice (Oryza sativa) leaves to simulated acid rain stress. Ecotoxicol Environ Saf 148:851–856. https://doi.org/10.1016/j.ecoenv.2017.11.046
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151(1):59–66. https://doi.org/10.1016/S0168-9452(99)00197-1
Merewitz EB, Gianfagna T, Huang B (2011) Protein accumulation in leaves and roots associated with improved drought tolerance in creeping bentgrass expressing an ipt gene for cytokinin synthesis. J Exp Bot 62(15):5311–5333. https://doi.org/10.1093/jxb/err166
Ayadi M, Brini F, Masmoudi K (2019) Overexpression of a wheat aquaporin gene, TdPIP2;1, enhances salt and drought tolerance in transgenic durum wheat cv. Maali. Int J Mol Sci 20(10):2389. https://doi.org/10.3390/ijms20102389
Bailly C, Audigier C, Ladonne F, Wagner MH, Coste F, Corbineau F, Come D (2001) Changes in oligosaccharide content and antioxidant enzyme activities in developing bean seeds as related to acquisition of drying tolerance and seed quality. J Exp Bot 52(357):701–708. https://doi.org/10.1093/jexbot/52.357.701
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2-ΔΔCt method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Schroeder S, Kim SH, Lee S, Sterflinger K, Breuil C (2002) The β-tubulin gene is a useful target for PCR-based detection of an albino Ophiostoma piliferum used in biological control of sapstain. Eur J Plant Pathol 108:793–801. https://doi.org/10.1023/A:1020897018610
Sun JW, Hu HQ, Li YL, Wang LH, Zhou Q, Huang XH (2016) Effects and mechanism of acid rain on plant chloroplast ATP synthase. Environ Sci Pollut Res 23(18):18296–18306. https://doi.org/10.1007/s11356-016-7016-3
Wang T, Yang WH, Xie YF, Shi DW, Ma YG, Sun X (2017) Effects of exogenous nitric oxide on the photosynthetic characteristics of bamboo (Indocalamus barbatus McClure) seedlings under acid rain stress. Plant Growth Regul 82(1):69–78. https://doi.org/10.1007/s10725-016-0239-y
Karpets YV, Kolupaev YE, Vayner AA (2015) Functional interaction between nitric oxide and hydrogen peroxide during formation of wheat seedling induced heat resistance. Russ J Plant Physiol 62(1):65–70. https://doi.org/10.1134/S1021443714060090
Parvaiz A, Abdel LAA, Abeer H, Abd Allah EF, Salih G, Tran LP (2016) Nitric oxide mitigates salt stress by regulating levels of osmolytes and antioxidant enzymes in chickpea. Front Plant Sci 7:347. https://doi.org/10.3389/fpls.2016.00347
Sheokand S, Kumari A, Sawhney V (2008) Effect of nitric oxide and putrescine on antioxidative responses under NaCl stress in chickpea plants. Physiol Mol Biol Plants 14(4):355–362. https://doi.org/10.1007/s12298-008-0034-y
Arora D, Jain P, Singh N, Kaur H, Bhatla SC (2015) Mechanisms of nitric oxide crosstalk with reactive oxygen species scavenging enzymes during abiotic stress tolerance in plants. Free Radic Res 50(3):291–303. https://doi.org/10.3109/10715762.2015.1118473
Zhang CM, Shi SL (2018) Physiological and proteomic responses of contrasting alfalfa (Medicago sativa L.) varieties to PEG-Induced osmotic stress. Front Plant Sci 9:242. https://doi.org/10.3389/fpls.2018.00242
Chamizo Ampudia A, Sanz Luque E, Llamas A, Galvan A, Fernandez E (2017) Nitrate reductase regulates plant nitric oxide homeostasis. Trends Plant Sci 22(2):163–174. https://doi.org/10.1016/j.tplants.2016.12.001
Soares C, Sousa AD, Pinto A, Azenha M, Fidalgo F (2016) Effect of 24-epibrassinolide on ROS content, antioxidant system, lipid peroxidation and Ni uptake in Solanum nigrum L. under Ni stress. Environ Exp Bot 122:115–125. https://doi.org/10.1016/j.envexpbot.2015.09.010
Arora D, Jain P, Singh N, Kaur H, Bhatla SC (2015) Mechanisms of nitric oxide crosstalk with reactive oxygen species scavenging enzymes during abiotic stress tolerance in plants. Free Radic Res 52(8):826–839. https://doi.org/10.3109/10715762.2015.1118473
Yang HJ, Mu JY, Chen LC, Feng J, Zuo JR (2015) S-nitrosylation positively regulates ascorbate peroxidase activity during plant stress responses. Plant Physiol 167(4):1604–1615. https://doi.org/10.1104/pp.114.255216
Mengel A, Chaki M, Shekariesfahlan A, Lindermayr C (2013) Effect of nitric oxide on gene transcription-s-nitrosylation of nuclear proteins. Front Plant Sci 4:293. https://doi.org/10.3389/fpls.2013.00293
Wendehenne D, Durner J, Klessig DF (2004) Nitric oxide: a new player in plant signalling and defence responses. Curr Opin Plant Biol 7(4):449–455. https://doi.org/10.1016/j.pbi.2004.04.002
Lamattina L, García-Mata C, Graziano M, Pagnussat G (2003) Nitric oxide: the versatility of an extensive signal molecule. Annu Rev Plant Biol 54:109–136. https://doi.org/10.1146/annurev.arplant.54.031902.134752
Kaiser WM, Huber SC (2001) Post-translational regulation of nitrate reductase: mechanism, physiological relevance and environmental triggers. J Exp Bot 52(363):1981–1989. https://doi.org/10.1093/jexbot/52.363.1981
Xia BX, Sun ZG, Wang LH, Zhou Q, Huang XH (2017) Analysis of the combined effects of lanthanum and acid rain, and their mechanisms, on nitrate reductase transcription in plants. Ecotoxicol Environ Saf 138:170–178. https://doi.org/10.1016/j.ecoenv.2016.12.034
Silva LS, Alves MQ, Seabra AR, Carvalho HG (2019) Characterization of plant glutamine synthetase s-nitrosation. Nitric Oxide Biol Chem 88:73–86. https://doi.org/10.1016/j.niox.2019.04.006
Funding
Funding was provided by undergraduate innovation and entrepreneurship training program in China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiao, R., Zhang, M., Wei, Z. et al. Alleviative effects of nitric oxide on Vigna radiata seedlings under acidic rain stress. Mol Biol Rep 48, 2243–2251 (2021). https://doi.org/10.1007/s11033-021-06244-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06244-w