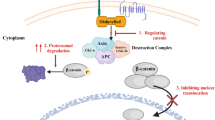
Abstract
Osteosarcoma is the most frequently occurring cancer in children as well as young adolescents and the metastatic forms worsen this condition to a further great extent. The metastatic dissemination of cancer cells is often acquired through a process of epithelial-mesenchymal transition (EMT). Since, phytochemicals have attracted intense interest in recent years due to their diverse pharmacological effects, in the present study, we investigated if berberine, a naturally occurring isoquinoline quaternary alkaloid, could modulate the EMT in osteosarcoma cells. Our experimental studies showed that berberine reduced cell viability, colony formation, wound healing ability and migration of osteosarcoma cells. Also, berberine significantly reduced the expression of matrix metalloproteinase (MMP)-2, suggesting its inhibitory action on the matrix metalloproteinases that are required for cancer cell invasion. The significant reduction in the expression of vimentin, N-cadherin, fibronectin and increased expression of E-cadherin further suggested its role in the inhibition of EMT in osteosarcoma cells. The downregulation of H3K27me3, as well as the decreased expression of the histone methyl transferase enzyme EZH2, further substantiated the fact that the plant alkaloid can be used as an epigenetic modulator in the treatment of osteosarcoma. In conclusion, our findings suggest that berberine inhibits proliferation and migration of osteosarcoma cells and most importantly reverses EMT along with modulation of key epigenetic regulators.






Similar content being viewed by others
References
Mirabello L, Troisi RJ, Savage SA (2009) International osteosarcoma incidence patterns in children and adolescents, middle ages and elderly persons. Int J Cancer 125(1):229–234
Mirabello L, Troisi RJ, Savage SA (2009) Osteosarcoma incidence and survival rates from 1973 to 2004: data from the Surveillance, Epidemiology, and End Results Program. Cancer: Interdiscip Int J Am Cancer Soc 115(7):1531–1543
Kaste SC, Pratt CB, Cain AM, Jones-Wallace DJ, Rao BN (1999) Metastases detected at the time of diagnosis of primary pediatric extremity osteosarcoma at diagnosis: imaging features. Cancer 86(8):1602–1608
Huang X, Zhao J, Bai J, Shen H, Zhang B, Deng L, Sun C, Liu Y, Zhang J, Zheng J (2019) Risk and clinicopathological features of osteosarcoma metastasis to the lung: a population-based study. J Bone Oncol 16:100230
Hunter KW, Crawford NP, Alsarraj J (2008) Mechanisms of metastasis. Breast Cancer Res 10, Article number: S2
Gonzalez DM, Medici D (2014) Signaling mechanisms of the epithelial-mesenchymal transition. Sci Signal 7(344):re8
Moreno-Bueno G, Portillo F, Cano A (2008) Transcriptional regulation of cell polarity in EMT and cancer. Oncogene 27(55):6958–6969
Serrano-Gomez SJ, Maziveyi M, Alahari SK (2016) Regulation of epithelial-mesenchymal transition through epigenetic and post-translational modifications. Mol Cancer 15(1), Article number: 18
Hu Y, Dai M, Zheng Y, Wu J, Yu B, Zhang H, Kong W, Wu H, Yu X (2018) Epigenetic suppression of E-cadherin expression by Snail2 during the metastasis of colorectal cancer. Clin Epigenet 10(1), Article number: 154
Zha L, Cao Q, Cui X, Li F, Liang H, Xue B, Shi H (2016) Epigenetic regulation of E-cadherin expression by the histone demethylase UTX in colon cancer cells. Med Oncol 33(3):21
Lv YF, Yan GN, Meng G, Zhang X, Guo QN (2015) Enhancer of zeste homolog 2 silencing inhibits tumor growth and lung metastasis in osteosarcoma. Sci Rep 5, Article number: 12999
Chou AJ, Geller DS, Gorlick R (2008) Therapy for osteosarcoma. Pediatric Drugs 10(5):315–327
Neergheen VS, Bahorun T, Taylor EW, Jen LS, Aruoma OI (2010) Targeting specific cell signaling transduction pathways by dietary and medicinal phytochemicals in cancer chemoprevention. Toxicology 278(2):229–241
Hosseini A, Ghorbani A (2015) Cancer therapy with phytochemicals: evidence from clinical studies. Avicenna J Phytomed 5(2):84–97
Vuddanda PR, Chakraborty S, Singh S (2010) Berberine: a potential phytochemical with multispectrum therapeutic activities. Expert Opin Investig Drugs 19(10):1297–1307
Cai C, Huang W, Zhou J, Wu Q, Huang Y, Zhao W, Zhang F, Wang Q, Zhang Y, Fang J (2019) An insight into the molecular mechanism of berberine towards multiple cancer types through systems pharmacology. Front Pharmacol 10:857
Sun Y, Xun K, Wang Y, Chen X (2009) A systematic review of the anticancer properties of berberine, a natural product from Chinese herbs. Anticancer Drugs 20(9):757–769
Jin H, Jin X, Cao B, Wang W (2017) Berberine affects osteosarcoma via downregulating the caspase-1/IL-1β signaling axis. Oncol Rep 37(2):729–736
Liu Z, Liu Q, Xu B, Wu J, Guo C, Zhu F, Yang Q, Gao G, Gong Y, Shao C (2009) Berberine induces p53-dependent cell cycle arrest and apoptosis of human osteosarcoma cells by inflicting DNA damage. Mutat Res 662(1–2):75–83
Zhu Y, Ma N, Li HX, Tian L, Ba YF, Hao B (2014) Berberine induces apoptosis and DNA damage in MG–63 human osteosarcoma cells. Mol Med Rep 10(4):1734–1738
Varshney R, Mishra R, Das N, Sircar D, Roy P (2019) A comparative analysis of various flavonoids in the regulation of obesity and diabetes: an in vitro and in vivo study. J Funct Foods 59:194–205
Varshney R, Gupta S, Roy P (2017) Cytoprotective effect of kaempferol against palmitic acid-induced pancreatic β-cell death through modulation of autophagy via AMPK/mTOR signaling pathway. Mol Cell Endocrinol 448:1–20
Varshney R, Varshney R, Mishra R, Gupta S, Sircar D, Roy P (2018) Kaempferol alleviates palmitic acid-induced lipid stores, endoplasmic reticulum stress and pancreatic β-cell dysfunction through AMPK/mTOR-mediated lipophagy. J Nutr Biochem 57:212–227
Loh CY, Chai JY, Tang TF, Wong WF, Sethi G, Shanmugam MK, Chong PP, Looi CY (2019) The E-cadherin and N-cadherin switch in epithelial-to-mesenchymal transition: signaling, therapeutic implications, and challenges. Cells 8(10):1118
Tsai JH, Yang J (2013) Epithelial–mesenchymal plasticity in carcinoma metastasis. Genes Dev 27(20):2192–2206
Yang G, Yuan J, Li K (2013) EMT transcription factors: implication in osteosarcoma. Med Oncol 30(4):697
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100(1):57–70
Machesky LM (2008) Lamellipodia and filopodia in metastasis and invasion. FEBS Lett 582(14):2102–2111
Izdebska M, Zielińska W, Grzanka D, Gagat M (2018) The role of actin dynamics and actin-binding proteins expression in epithelial-to-mesenchymal transition and its association with cancer progression and evaluation of possible therapeutic targets. BioMed Res Int 2018, Article ID 4578373
Chu SC, Yu CC, Hsu LS, Chen KS, Su MY, Chen PN (2014) Berberine reverses epithelial-to-mesenchymal transition and inhibits metastasis and tumor-induced angiogenesis in human cervical cancer cells. Mol Pharmacol 86(6):609–623
PosthumaDeBoer J, Witlox MA, Kaspers GJ, van Royen BJ (2011) Molecular alterations as target for therapy in metastatic osteosarcoma: a review of literature. Clin Exp Metas 28(5):493–503
Kleiner DE, Stetler-Stevenson WG (1999) Matrix metalloproteinases and metastasis. Cancer Chemother Pharmacol 43(1):S42–S51
Bjørnland K, Flatmark K, Pettersen S, Aaasen AO, Fodstad O, Melandsmo GM (2005) Matrix metalloproteinases participate in osteosarcoma invasion. J Surg Res 127(2):151–156
Dave B, Mittal V, Tan NM, Chang JC (2012) Epithelial-mesenchymal transition, cancer stem cells and treatment resistance. Breast Cancer Res 14(1), Article number: 202
Yang H, Zhang Y, Zhou Z, Jiang X, Shen A (2014) Transcription factor Snai1–1 induces osteosarcoma invasion and metastasis by inhibiting E–cadherin expression. Oncol Lett 8(1):193–197
Kim K, Lu Z, Hay ED (2002) Direct evidence for a role of β-catenin/LEF-1 signaling pathway in induction of EMT. Cell Biol Int 26(5):463–476
Chen C, Zhao M, Tian A, Zhang X, Yao Z, Ma X (2015) Aberrant activation of Wnt/β-catenin signaling drives proliferation of bone sarcoma cells. Oncotarget 6(19):17570–17583
Lin CH, Ji T, Chen CF, Hoang BH (2014) Wnt signaling in osteosarcoma. Adv Exp Med Biol 804:33–45
Gottardi CJ, Wong E, Gumbiner BM (2001) E-cadherin suppresses cellular transformation by inhibiting β-catenin signaling in an adhesion-independent manner. J Cell Biol 153(5):1049–1060
Feng H, Tillman H, Wu G, Davidoff AM, Yang J (2018) Frequent epigenetic alterations in polycomb repressive complex 2 in osteosarcoma cell lines. Oncotarget 9(43):27087–27091
Liu D, Meng X, Wu D, Qiu Z, Luo H (2019) A natural isoquinoline alkaloid with antitumor activity: studies of the biological activities of berberine. Front Pharmacol 10:9
Wang Z, Liu Y, Xue Y, Hu H, Ye J, Li X, Lu Z, Meng F, Liang S (2016) Berberine acts as a putative epigenetic modulator by affecting the histone code. Toxicol In Vitro 36:10–17
Chen S, Chen X, Li Y, Zhang F, Yang S, Mo KL, Mo X, Ding Y (2015) Inhibitory effect of berberine on zeste homolog 2 (Ezh2) enhancement in human esophageal cell lines. Trop J Pharm Res 14(9):1589–1595
Acknowledgements
The authors are highly grateful to Michigan Diagnostics, Royal Oak, MI, USA for the kind gift of immune-detection reagent for western blot analysis.
Funding
This study was funded by research grants from Ministry of Human Resource and Development, Government of India and Indian Institute of Technology Roorkee, Roorkee, as research fellowship to RM.
Author information
Authors and Affiliations
Contributions
RM: Conceptualization, Data curation. Investigation, Formal analysis, Validation, Writing—original draft. SN: Data curation, Formal analysis, Writing—original draft. RV: Data curation, Formal analysis, Writing—original draft. DS: Resources, and Visualization. PR: Project administration, Resources, Supervision, Conceptualization, Visualization, Validation, Writing—Review & Editing.
Corresponding author
Ethics declarations
Conflicts of interest
RM declares that she has no conflict of interest. SN declares that he has no conflict of interest. RV declares that she has no conflict of interest. DS declares that he has no conflict of interest. PR declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mishra, R., Nathani, S., Varshney, R. et al. Berberine reverses epithelial-mesenchymal transition and modulates histone methylation in osteosarcoma cells. Mol Biol Rep 47, 8499–8511 (2020). https://doi.org/10.1007/s11033-020-05892-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05892-8