Abstract
The Nomo1 gene mediates a wide range of biological processes of importance in embryonic development. Accordingly, constitutive perturbation of Nomo1 function may result in myriad developmental defects that trigger embryonic lethality. To extend our understanding of Nomo1 function in postnatal stages and in a tissue-specific manner, we generated a conditional knockout mouse model of Nomo1. To achieve this, we used clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 technology in C57Bl/6J mouse zygotes to generate a new mouse model in which exon 3 of the Nomo1 gene is specifically flanked (or floxed) by LoxP sites (Nomo1f/f). Nomo1f/f mouse embryonic fibroblasts were transduced with a Cre adenovirus and efficiently recombined between LoxP sites. Genomic and expression studies in Nomo1-transduced MEFs demonstrated that the Nomo1 exon 3 is ablated. Western blot assay showed that no protein or early truncated protein is produced. In vivo assay crossing Nomo1f/f mouse with a Msi1-CRE transgenic mouse corroborated the previous findings and it showed Nomo1 exon 3 deletion at msi1+ cell compartment. This short technical report demonstrates that CRISPR/Cas9 technology is a simple and easy method for creating conditional mouse models. The Nomo1f/f mouse will be useful to researchers who wish to explore the role of Nomo1 in any developmental stage or in a tissue-specific manner.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Human Genome Project (HPG) was an international, collaborative research program whose goal was the complete mapping of all the genes of human beings. The HPG revealed that there are probably about 20,500 human genes [7] and subsequently much effort has been expended on understanding their functions and on studying how gene mutations affect the occurrence of human diseases. Since human and mouse genes are 95% homologous, it soon became apparent that mouse genes could serve as tools for understanding human gene function. In this context, researchers and various consortia initially used gene targeting or gene trapping in mouse stem cells to knock out the corresponding murine homologous loci [1, 10]. Phenotypic characterization of these mutations is the most accurate and widely used method for elucidating in vivo gene functions and the genetics of diseases. Nevertheless, customary knockout methods can give rise to embryonic lethality from a gene mutation, which prevents researchers from studying the gene in postnatal stages [35]. Tissue-specific gene functions could remain unknown when the null mutation cannot be exclusively produced in a particular tissue. To overcome these obstacles, conditional gene knockout with the Cre-loxP recombination system [31] is widely used to eliminate a specific gene in a particular tissue at a certain development stage [15]. Many mouse stem cell clones have been developed by the International Knockout Mouse Consortium with putative conditional null mutations that are being used to generate conditional mice [1, 8]. However, the process of generating the mouse model from the ES cell clone is not free of problems: it is laborious, time-consuming, and can only be carried out in certain mouse strains. Fortunately, the recently developed clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 system has changed the gene targeting landscape, making the use of transgenesis and mutagenesis technology more feasible, not only in zebrafish or mice as was first reported [28], but in all organisms [14, 19, 32]. This report describes in detail the generation by CRISPR/Cas9 technology of a novel conditional Nomo1 mouse, which will allow Nomo1 gene function to be studied in a rigorous and specific way that avoids embryonic lethality.
The Nodal subfamily belongs to the group of TGFβ proteins that play a key role in the embryonic development of vertebrates. Nodal signaling is essential for the patterning of the early embryo during mesoderm and endoderm induction as well as the specification of left–right asymmetry [26, 27]. Gene expression studies showed that Nodal proteins act before gastrulation to specify the mesodermal and endodermal progenitors, and that the patterning depends on graded signaling with participation of Nodal inhibitors [5, 9, 29]. These inhibitors can directly bind and inhibit Nodal proteins or compete with them to bind to their coreceptors [3, 6]. Nomo (Nodal modulator) and its binding partner, Nicalin, are components of a novel protein complex involved in Nodal signaling. This complex has been revealed as an antagonist of Nodal signaling during mesendodermal patterning in zebrafish. The evolutionary conservation of Nicalin and Nomo in metazoans and plants indicates that the Nomo/Nicalin complex could have a similarly important role. Nomo/Nicalin transcripts were detected in all postnatal human tissues, and an overlapping subcellular distribution was found in endoplasmic reticulum membranes. In zebrafish embryos, Nomo expression was detected in the endoderm and anterior mesendoderm, and its overexpression was found to impair forebrain patterning [11]. Other studies showed that the Nomo1 gene positively regulates the expression of the master regulator and other key development genes involved in bone formation and cartilage development, and that it is essential for cartilage development in zebrafish [2]. The NOMO1 gene is also known to be substantially downregulated in human ventricular septal defect myocardium and, thus, may be an important molecular pathway in human heart development [34]. In humans, tumors from the early-onset subgroup of colorectal cancers showed a homozygous deletion in NOMO1 and were associated with a microsatellite-stable feature and better prognosis, although the molecular mechanism was not identified [21].
Despite the study of Nomo in embryogenesis and pattern expression in embryo and adult tissues, its biological functions in mammalian postnatal tissues remain unknown. Since Nomo is required to produce proper axial mesoderm patterning, bone and heart formation, complete Nomo inhibition could impair embryo development. To study its functions during postnatal stages we decided to create a new mouse model to conditionally interfere with its functions.
Material and methods
Ethics statement
This study followed Spanish and European Union guidelines for animal experimentation (RD 1201/05, RD 53/2013 and 86/609/CEE). The study was approved by the Bioethics Committee of the University of Salamanca.
In silico analysis and CRISPR/Cas9 system design
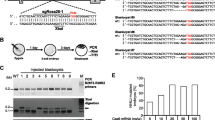
The mouse Nomo1 gene contains 31 coding exons that produce 5 different transcripts (annotated as Nomo1-201 to Nomo1-205). Nomo1-201, with a length of 4257 bp, is the only one capable of translating into a protein, comprising 1214 residues. Nomo1 exon 3 is the first exon with a residue overlapping the splicing site. In silico analysis showed that an early truncated Nomo1 protein could be produced when exon 3 is removed. Two sgRNAs (Nomo1-sgRNA1 and Nomo1-sgRNA2), targeting the coding sequence of Nomo1 exon 3 and the adjacent intron sequence, were designed with the Spanish National Biotechnology Centre (CNB)-CSIC web tool (http://bioinfogp.cnb.csic.es/tools/breakingcas/) (Fig. 1). Two complementary oligos corresponding to sgRNAs were designed, including two 4-bp overhang sequences (Nomo1 UP and LO oligos, Table 1).
Schematic representation of Nomo1 locus and targeting strategy. Detail of wild-type Nomo1 exon 3 with expected cleavage points of two sgRNAs and the long ssDNA template used for HDR, carrying two silent mutations in the PAM sequences of sgRNAs (Loxp sites are represented by red arrowhead). The flox allele represents the expected result of homology recombination between wt allele and the template. Finally, the delta allele arises from Cre recombinase action, eliminating exon 3 in mRNA and generating a stop codon in Nomo1 exon 4
As earlier papers described, a long single-strand oligonucleotide of DNA (long ssDNA) (Integrated DNA Technologies, IDT) was considered as donor for homologous recombination to create the conditional KO allele [17, 18, 24]. A Nomo1 long ssDNA containing LoxP sequences in the same orientation flanking exon 3 was designed, adding silent mutations that destroy PAM sequences. This had an expanded total length of 789 bp (Supplementary Fig. S1).
Generation of CRISPR/Cas9 reagents
Cas9 mRNA and sgRNA were generated by cloning into a plasmid vector and followed by in vitro transcription [12]. To clone the sgRNAs into the pX458 vector it was previously linearized with BpiI (NEB). The two complementary oligos corresponding to each Nomo1 sgRNA were denatured at 95 °C for 5 min, ramp-cooled to 25 °C over 45 min to allow annealing, and they were ligated with the linearized pX458 (Addgene plasmid # 48138) [25]. Competent cells were transformed with 2 μl of the ligated plasmid, and single colonies were grown before plasmid extraction using a QIAprep Spin Maxiprep kit (Qiagen). The correct insertion of the sgRNA sequences was confirmed by Sanger sequencing. sgRNA sequences were PCR-amplified from px458-based vector with primers carrying the T7 RNA polymerase promoter at the 5′ ends (Nomo1 T7 F and gRNA R, Table 1). PCR products were purified (NZYGelpure, NZYTech) and used as a template for in vitro T7 RNA polymerase transcription (MEGAshortscript T7 Transcription Kit, Thermo Fisher).
The Cas9 nuclease ORF, including NLS, was also PCR-amplified from pX458 vector [25] with primers carrying the T7 RNA polymerase promoter at the 5′ ends (T7 Cas9 F and R, Table 1). The Cas9 PCR product was purified and used as a template for in vitro transcription, 5′ capping (mMESSAGE mMACHINE T7 Transcription Kit, Thermo Fisher), and 3′ poly(A) tailing [Poly(A) Tailing Kit, Thermo Fisher]. Transcription products were purified with RNeasy Mini Kit (Qiagen) and eluted in nuclease-free EmbryoMax microinjection buffer (Millipore).
Nowadays, all this reagents and methodology can be replaced by injecting embryos with CRISPR ribonucleoproteins (RNPs), acquiring commercial sgRNAs and Cas9 protein [4, 13, 20, 24].
In vitro test to analyze Nomo1-sgRNAs and Cas9 activity
Ten surplus C57BL/6J zygotes from routine processes carried out in the Transgenesis Facility of University of Salamanca were microinjected with 20 ng/µl of Nomo1 sgRNAs, 20 ng/µl of Cas9 mRNA, and 20 ng/µl of Cas9 protein into the cytoplasm and pronucleus. Microinjected embryos were grown on KSOM with non-essential amino acids (Millipore), 37 °C and 5% CO2 atmosphere at the blastocyst stage. Genomic DNA from single blastocyst-staged embryos were extracted in 10 μl of lysis buffer (50 mM KCl, 10 mM Tris–HCl pH 8.5, 0.1% Triton X-100, and 4 mg/ml of proteinase K) at 55 °C overnight, then heated at 95 °C for 10 min. 2 μl of this DNA solution was used as a template for two rounds of PCR (30 cycles + 20 cycles) to amplify the target sequences using a specific primer for the Nomo1 target region (Nomo1 F and R oligos, Table 1). PCR products were purified using a High Pure PCR Product Purification Kit (Roche) and sequenced by the Sanger method using forward and reverse PCR primers.
Mice and embryo microinjection
Sixty-seven 1-cell-stage embryos from 6 superovulated C57BL/6J females were harvested and microinjected with 20 ng/µl of sgRNA, 20 ng/µl of Cas9 mRNA, 20 ng/µl of Cas9 protein and 25 ng/µl of long ssDNA template solution into the cytoplasm and pronucleus. Microinjected embryos were grown overnight on KSOM with non-essential amino acids (Millipore), 37 °C and 5% CO2 atmosphere. Fifty-six two-cell-stage embryos were recovered and transferred to two pseudopregnant females. Embryo donor females were euthanized by cervical dislocation and given humanitarian care in accordance with the standards of the Bioethical Committee of University of Salamanca and with Spanish and European Union guidelines for animal experimentation.
Msi1-Cre transgenic mice (MGI:5296021) [22] (see Supplementary Data) was used to express CRE protein at brain and intestinal crypt cells when they were crossed with the Nomo1 conditional model (Nomo1f/f F2). Cre expression location was tested crossing Msi1-Cre with a Rosa26 βgal Cre-reporter mouse [30] (Supplementary Fig. S2).
Mouse embryonic fibroblast extraction, culture and viral transduction
Mouse embryonic fibroblasts (MEFs) were extracted 13.5 days post coitum (dpc) following procedures described at Bio Protoc. 2013 Sep 20; 3(18): e908. MEFs were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) (Life Technologies) supplemented with 10% fetal bovine serum (FBS) and 1% of penicillin/streptomycin (Life Technologies) at 37 °C in a 5% CO2 atmosphere.
Cre recombinase was expressed in MEFs by virally transducing 300 MOI of Ad5CMVCre-eGFP virus (Viral Vector Core, University of Iowa). 1 × 10e5 MEFs were incubated overnight with the virus in the absence of FBS and 48 h after infection. DNA and RNA were extracted with a QIAamp DNA Micro Kit and an RNAeasy Mini Kit, respectively (Qiagen).
Analysis of Cas9-system targets sites
Mouse genomic DNA was extracted from tail biopsy though proteinase K digestion, followed by phenol/chloroform purification. Genomic DNA from MEFs was extracted using the QIAamp DNA Micro Kit (Qiagen) following the manufacturer’s protocol. To amplify target regions of mouse Nomo1 gene, PCR was performed with Nomo1 F and Nomo1 R oligos (Table 1). PCR products were purified using a High Pure PCR Product Purification Kit (Roche) and sequenced by the Sanger method using forward and reverse PCR primers.
To characterize the various alleles generated in mouse pups or in MEFs, PCR products were subcloned and transformed in bacteria. DNA from single clones was extracted with a QIAprep Spin Miniprep Kit (Qiagen) and Sanger-sequenced.
RT-PCR and quantitative PCR
To test Nomo1 mRNA expression, 150 ng of total mRNA from MEFs was in vitro-retrotranscribed using a SuperScript III First-Strand Synthesis Super Mix kit (Thermo Fisher). MEF cDNA was used as a template to PCR-amplify the specific sequence between exons 2 and 4 with oligos Nomo1Ex2F and Nomo1Ex4R (Table 1). PCR product was purified and Sanger-sequenced.
Nomo1 mRNA expression was quantified by qPCR (Applied Biosystems) using oligos targeting Nomo1 exon 5 Nomo1Ex5qPCRF 5 and Nomo1 exon 6 Nomo1Ex6qPCRR. GAPDH mRNA expression was used as a control (oligos Gapdh qPCR F and R, Table 1).
Western blot
Nomo1 protein expression was assessed by SDS-PAGE and western blotting using a rabbit anti-mouse Nomo1 antibody (1:200; Thermo Fisher). Horseradish peroxidase-conjugated anti-rabbit antibody (1:1000; Cell Signaling) was used as a secondary antibody. Antibodies were detected using ECLTM Western Blotting Detection Reagents (RPN2209, GE Healthcare). As a control, β-actin expression was measured using mouse anti-β-actin (1:20,000, Sigma Aldrich).
Statistical analysis
Statistical analysis of annexin V expression was performed using GraphPad Prism version 6.00 for Mac OS X (GraphPad Software, La Jolla, California, USA, www.graphpad.com). Differences in Nomo1 expression were tested by two-way ANOVA, followed by Tukey’s post hoc multiple comparison test. Experimental results were expressed as the mean ± standard deviation (SD). Statistical significance was concluded for values of p < 0.05.
Results
CRISPR/Cas9 reagents efficiently induce mutations at the target site of the Nomo1 sgRNAs
Before injecting embryos with CRISPR/Cas9 reagents, we tested the activity of Nomo1 sgRNAs by injecting 10 C57BL/6J zygotes with a mixture of mRNA Cas9, Cas9 protein and Nomo1 sgRNAs. A nested PCR of material obtained from single blastocyst stage embryos was performed. Genomic DNA from all the embryos was analyzed by Sanger sequencing. A large number of mutations at the expected cut site for both Nomo1 guides was detected in most of the single embryos. A high proportion of mosaic embryos with different alleles was detected (Fig. 2A); in most of them, the alleles exhibited a specific deletion corresponding to the target area (43 bp) flanked by the cut sites between Nomo1 guides (Fig. 2B) and other mutated (Fig. 2C) and wild-type (wt) alleles.
Analysis of SgRNAs and CRISPR/Cas9 activity in Nomo1 locus. A Sanger sequencing of Nomo1 exon 3 amplified from a single edited embryo with both sgRNAs. A mixed sequence is noted between the expected cleavage point (blue arrowhead) of the two sgRNAs (blue box). Subcloning and Sanger sequencing revealed B a 46-bp deletion in the sequence between cleavage points of the two sgRNAs, and C some indels due to the Cas9 action of just one cleavage point
CRISPR/Cas9 technology produces Nomo1 mosaic mice with a wide variety of mutated alleles
Two pseudopregnant females were transferred, each with 28 two-cell embryos. An offspring of 5 live (#1 to #5) and 5 dead (#6 to #10) pups were born from them. The target region of Nomo1 locus was analyzed by PCR in all of them. All pups, except #3, showed the 786-bp band corresponding to the wt allele. Additionally, six of them displayed secondary bands with higher or lower molecular weights (Fig. 3A), indicating the presence of mosaic pups. Sanger sequencing of all pups confirmed the presence of mosaic animals with three or more edited alleles in most of them. Sanger sequencing revealed a high percentage of null alleles in dead pups due to frameshift mutations (Fig. 3B).
Offspring analysis. A Genotyping of F0 (ten pups) by PCR of the Nomo1 locus. Several modifications were observed. Inset: pup #1 shows three alleles, two of which correspond to the expected molecular weight of the WT and flox alleles. B Subcloning and sequencing of some alleles carried by offspring. C Subcloning and sequencing of alleles observed in pup ID #1. Two alleles showed deletions (4 bp and 47 bp), and the third allele (flox allele) corresponds to an insertion of two Loxp sites flanking the exon 3 and silent PAM mut at the expected points. D Breeding scheme and flox allele segregation. PCR analysis of F1 offspring detected 4 of 9 pups carrying the flox allele (asterisk)
CRISPR/Cas9 technology efficiently produces a Nomo1 conditional allele
Three of the pups analyzed (#1, #9 and # 10) displayed 1 higher secondary band with the expected weight (854 bp), a result that was compatible with the conditional designed allele (Fig. 3A). Sanger sequencing revealed the presence of LoxP sites in two of the pups. In silico analysis showed that pups #9 and #10 contained 1 partially recombined allele with a single LoxP site and the mutated PAM sequence. However, pup #1 had four distinct alleles: one wt, two with insertions or deletions (indels), and one allele edited with the conditional design (Fig. 3C).
Mosaic Nomo1 conditional founder F0 establishes the colony and effectively segregates the conditional allele
Mosaic male #1 was viable and was selected as the founder (F0) of the colony. The F0 was mated with a C57Bl6/J female to segregate the conditional allele into an inbred strain. F1 litter sizes varied from 4 to 9 pups per delivery, and the conditional allele was segregated in a 1/3–1/4 ratio (Fig. 3D). Heterozygous (Nomo1f/+) F1 were fertile and mated to generate homozygous mutants (Nomo1f/f) F2. The Nomo1f/f F2 mice born had a pattern Mendelian inheritance; they were viable and fertile, and displayed no obvious alterations in size or behavior.
Cre recombinase efficiently removes Nomo1 exon 3 in vitro and in vivo
To test whether Cre activity effectively removes the LoxP exon 3 flanked region, we transduced E12.5-derived MEFs from Nomo1+/+, Nomo1f/+ and Nomo1f/f mice with GFP-Cre adenovirus. Nomo1+/+, Nomo1f/+ and Nomo1f/f MEFs expressed a high level of GFP after adenoviral infection, indicating an efficient expression of Cre recombinase (Fig. 4A). Genotyping of MEFs after adenoviral infection showed no differences in Nomo1+/+ or non-transduced MEFs. However, all mutant MEFs (Nomo1f/+ and Nomo1f/f) showed a lower band corresponding to the molecular weight expected after Cre activity (482 bp) and exon 3 removal (Fig. 4B). In order to corroborate these findings, we crossed a female Nomo1f/f with a male Msi1-Cre transgenic mouse (Msi1-CREtg/tg) [22]. Intestinal and brain tissues from Nomo1+/f; Msi1-CRE+/tg double mutants were collected and genomic DNA was isolated. A specific lower band due to Cre activity at Msi1+ cells at Nomo1 floxed locus was obtained (Fig. 4C).
Functional analysis of the Nomo1 flox allele. A Fluorescent microscopy of MEFs extracted from Nomo1+/+, Nomo1+/f and Nomo1f/f 13.5 d.p.c. embryos transduced with adeno-Cre-GFP virus. B PCR amplification of the Nomo1 locus of non-transduced and transduced MEFs with adeno-Cre-GFP virus. Nomo1+/f and Nomo1f/f transduced MEFs showed 854 bp and 482 bp bands corresponding to the flox and delta alleles, respectively. Nomo1+/f also featured a wt allele (786 bp). Whereas Nomo1+/+-transduced MEFs showed a single band (786 bp) corresponding to the wt allele. C Msi1-Cretg/tg transgenic mice were crossed with Nomo1f/f mice. PCR amplification of the Nomo1 locus detected heterozygous double mutants at F1 pups (upper gel). Intestine and Brain genomic DNA were isolated from double mutant Msi1-Cre+/tgNomo1+/f mice and PCR amplification of the Nomo1 locus detected a 482 bp band due to Cre activity on Nomo1 floxed locus (lower gel)
Cre recombinase activity in Nomo1 f/f cells generates a Nomo1 null allele
Expression of Nomo1 mRNA and protein was studied in adeno-CRE transduced and non-transduced MEFs from Nomo1+/+, Nomo1f/+ and Nomo1f/f mice. After adenoviral infection, Nomo1 mRNA expression was checked by RT-PCR in all types of MEFs. All MEFs showed an mRNA of 233 bp, corresponding to the specific amplified Nomo1 cDNA region containing exon 3. Nevertheless, Nomo1f/+ and Nomo1f/f MEFs showed an additional 187 bp lower band (Fig. 5A). PCR subcloning and Sanger sequencing of this lower band demonstrated the correct excision of exon 3. In silico analysis of the Nomo1f/f allele after Cre activity revealed the presence of an early stop codon that triggered a truncated Nomo1 protein with a different translating frame due to absence of the exon 3 (Fig. 5A).
Effect of Nomo 1 exon 3 excision. A cDNA from transduced MEFs were obtained and amplified to analyze the mRNA expression of Nomo1. MEFs with the floxed allele express wild type (233 bp) and Delta (187 bp) transcripts (left). Sanger sequencing revealed that Delta transcript corresponds to mRNA without exon 3 (right). B Q-PCR to quantify Nomo1 mRNA. Nomo1f/f and Nomo1f/+ MEFs showed a lower expression of Nomo1+/+ after adeno-Cre transduction. Nomo1f/f transduced MEFs showed the lowest level of Nomo1 expression (80% lower level, ***p < 0.001). C Western blot analysis of Nomo1 protein expression. After adeno-Cre transduction, Nomo1+/f and Nomo1f/f MEFs showed a dramatic reduction of Nomo1 protein, more evident in Nomo1f/f transduced MEFs
To study the effect of Nomo1 exon 3 excision by Cre recombinase, Nomo1 expression was measured by real-time PCR and western blot. Nomo1 mRNA expression was quantified in Nomo1+/+, Nomo1f/+ and Nomo1f/f MEFs before and after adeno-Cre transduction. We observed a substantial reduction (> 80%) of Nomo1 mRNA in Nomo1f/f-transduced MEFs compared with Nomo1+/+ (Fig. 5B). These results were corroborated at the protein level by western blot assay, which showed a lower level of Nomo1 expression in Nomo1f/+ and minimal expression in Nomo1f/f MEFs due to the presence of a minimal percentage of non-transduced Nomo1f/f MEFs (Fig. 5C).
Discussion
Genetically engineered mouse models are the best tool for studying gene function and human diseases. In particular, the Cre-LoxP system is widely used to investigate gene functions in specific cellular compartments (spatial control) and/or at specific ages (temporal control). Hundreds of gene-floxed lines and constitutive or tissue-specific Cre-driver mouse lines have been generated to date and more continue to be developed. They are key to understanding the gene function in a specific cell context. In mouse models of human cancer, instead of using tissue-specific Cre mouse lines, a more real approximation might be to turn-off/turn-on the tumor suppressor gene/oncogene in a small group of specific cells using Cre-viral vectors.
Until recently, the process of generating conditional alleles in mice involved manipulating embryonic stem cells, which is a laborious, time consuming and inefficient process. Nowadays, CRISPR/Cas9 technology has improved and simplified this process, allowing us to generate a conditional model that is quicker to use with a reduction in time and number of animals.
This technical report shows that is possible to create a conditional mouse model using a small group of females as embryo donors. However, a careful study of the gene sequence is always necessary. At this point, short exons in early positions with overlapping codons should be chosen to guarantee an effective null allele when they are removed by Cre activity. Other aspects to consider are the LoxP positions flanking the key exons and the distance between them. In order not to alter the branch site, the LoxP site should not be placed within the first 100 nucleotides upstream of the 3′ end of the intron to ensure that the splicing process is not affected. The first intron of a gene should be avoided because it could contain regulatory DNA sequences and disrupting a key transcription factor binding site could affect the expression of the gene. Regarding the size of the fragment floxed, although big deletions have been achieved with the Cre-LoxP-system flanking the full length of a gene, LoxP positions could certainly affect the efficiency of recombination if they are separated by a long distance.
Despite all these general instructions, an in vitro study to test sgRNAs and Cas9 activity is always recommended before use. In this paper we have used surplus embryos with an identical genetic background. However, mouse cell lines could be used for this purpose as long as they have the same sgRNA target sequence as the mouse embryo. Thus, the target areas would have to be sequenced in order to rule out polymorphisms and mutations that could interfere with the CRISPR/Cas9 activity. Although the CRISPR/Cas9 system is a rapid, simple, and often extremely efficient gene-editing method, the main obstacle to producing gene-edited animals is mosaicism in the founders [16, 23, 33]. As our results show, most mice had three or more alleles and, fortunately, the conditional allele was segregated. However, in general, the major challenge of mosaicism is that the mutated allele will not be transmitted to the offspring. In any case, mosaicism also complicates genotype analysis and PCR cloning, and genome sequencing approaches are always required. Nevertheless, not all are disadvantages regarding mosaicism, and it can provide useful advantages, such as the rapid estimation of candidate gene functions in vivo, and the direct comparison of mutant and wt cells in the same organ of mosaic animals. Furthermore, the presence of null alleles in a small proportion of cells enables animals to survive beyond the lethal phase, thus enabling study of the null phenotype in specific groups of cells [36]. Additionally, a mosaic founder mouse can segregate different alleles, generating several mutants in one step.
Given all these challenges and strengths, CRISPR-Cas technology allows the genome mouse embryo to be modified in an easy and conditional manner. We have used this technology to generate a conditional mouse model to study Nomo1 gene function and have demonstrated that Cre activity removes exon 3 to produce a non-functional allele.
References
Austin CP, Battey JF, Bradley A et al (2004) The Knockout Mouse Project. Nat Genet 36:921–924. https://doi.org/10.1038/ng0904-921
Cao L, Li L, Li Y et al (2018) Loss of the Nodal modulator Nomo results in chondrodysplasia in zebrafish. Curr Mol Med 18:448–458. https://doi.org/10.2174/1566524019666181212095307
Chen C, Shen MM (2004) Two modes by which Lefty proteins inhibit nodal signaling. Curr Biol 14:618–624. https://doi.org/10.1016/j.cub.2004.02.042
Chen S, Lee B, Lee AY-F et al (2016) Highly efficient mouse genome editing by CRISPR ribonucleoprotein electroporation of zygotes. J Biol Chem 291:14457–14467. https://doi.org/10.1074/jbc.m116.733154
Chen Y, Schier AF (2002) Lefty proteins are long-range inhibitors of squint-mediated nodal signaling. Curr Biol 12:2124–2128. https://doi.org/10.1016/s0960-9822(02)01362-3
Cheng SK, Olale F, Brivanlou AH, Schier AF (2004) Lefty blocks a subset of TGFbeta signals by antagonizing EGF-CFC coreceptors. PLoS Biol 2:E30. https://doi.org/10.1371/journal.pbio.0020030
Clamp M, Fry B, Kamal M et al (2007) Distinguishing protein-coding and noncoding genes in the human genome. Proc Natl Acad Sci USA 104:19428–19433. https://doi.org/10.1073/pnas.0709013104
Collins FS, Rossant J, Wurst W (2007) A mouse for all reasons. Cell 128:9–13. https://doi.org/10.1016/j.cell.2006.12.018
Feldman B, Concha ML, Saude L et al (2002) Lefty antagonism of Squint is essential for normal gastrulation. Curr Biol 12:2129–2135. https://doi.org/10.1016/s0960-9822(02)01361-1
Guan C, Ye C, Yang X, Gao J (2010) A review of current large-scale mouse knockout efforts. Genesis 48:73–85. https://doi.org/10.1002/dvg.20594
Haffner C, Frauli M, Topp S et al (2004) Nicalin and its binding partner Nomo are novel Nodal signaling antagonists. EMBO J 23:3041–3050. https://doi.org/10.1038/sj.emboj.7600307
Harms DW, Quadros RM, Seruggia D et al (2014) Mouse genome editing using the CRISPR/Cas system. Curr Protoc Hum Genet. https://doi.org/10.1002/0471142905.hg1507s83
Jacobi AM, Rettig GR, Turk R et al (2017) Simplified CRISPR tools for efficient genome editing and streamlined protocols for their delivery into mammalian cells and mouse zygotes. Methods 121–122:16–28. https://doi.org/10.1016/j.ymeth.2017.03.021
Jinek M, Chylinski K, Fonfara I et al (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science (80-) 337:816–821. https://doi.org/10.1126/science.1225829
Kim H, Kim M, Im S-K, Fang S (2018) Mouse Cre-LoxP system: general principles to determine tissue-specific roles of target genes. Lab Anim Res 34:147–159. https://doi.org/10.5625/lar.2018.34.4.147
Mehravar M, Shirazi A, Nazari M, Banan M (2019) Mosaicism in CRISPR/Cas9-mediated genome editing. Dev Biol 445:156–162. https://doi.org/10.1016/j.ydbio.2018.10.008
Miura H, Gurumurthy CB, Sato T et al (2015) CRISPR/Cas9-based generation of knockdown mice by intronic insertion of artificial microRNA using longer single-stranded DNA. Sci Rep 5:12799. https://doi.org/10.1038/srep12799
Miura H, Quadros RM, Gurumurthy CB, Ohtsuka M (2018) Easi-CRISPR for creating knock-in and conditional knockout mouse models using long ssDNA donors. Nat Protoc 13:195–215. https://doi.org/10.1038/nprot.2017.153
Mojica FJM, Montoliu L (2016) On the origin of CRISPR-Cas technology: from prokaryotes to mammals. Trends Microbiol 24:811–820. https://doi.org/10.1016/j.tim.2016.06.005
Nakagawa Y, Sakuma T, Nishimichi N et al (2016) Ultra-superovulation for the CRISPR-Cas9-mediated production of gene-knockout, single-amino-acid-substituted, and floxed mice. Biol Open 5:1142–1148. https://doi.org/10.1242/bio.019349
Perea J, Garcia JL, Perez J et al (2017) NOMO-1 gene is deleted in early-onset colorectal cancer. Oncotarget 8:24429–24436. https://doi.org/10.18632/oncotarget.15478
Pérez-Caro M, Mendez-Sanchez L, Perez-Fontan J-F et al (2011) Msi1-CRE transgenic mice: driving transgene expression to the putative gut stem cell. Transgenic Res 20 Abstr 40
Quadros RM, Harms DW, Ohtsuka M, Gurumurthy CB (2015) Insertion of sequences at the original provirus integration site of mouse ROSA26 locus using the CRISPR/Cas9 system. FEBS Open Bio 5:191–197. https://doi.org/10.1016/j.fob.2015.03.003
Quadros RM, Miura H, Harms DW et al (2017) Easi-CRISPR: a robust method for one-step generation of mice carrying conditional and insertion alleles using long ssDNA donors and CRISPR ribonucleoproteins. Genome Biol 18:92. https://doi.org/10.1186/s13059-017-1220-4
Ran FA, Hsu PD, Wright J et al (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8:2281–2308. https://doi.org/10.1038/nprot.2013.143
Schier AF (2003) Nodal signaling in vertebrate development. Annu Rev Cell Dev Biol 19:589–621. https://doi.org/10.1146/annurev.cellbio.19.041603.094522
Schier AF, Shen MM (2000) Nodal signalling in vertebrate development. Nature 403:385–389. https://doi.org/10.1038/35000126
Shen B, Zhang J, Wu H et al (2013) Generation of gene-modified mice via Cas9/RNA-mediated gene targeting. Cell Res 23:720–723
Solnica-Krezel L (2003) Vertebrate development: taming the nodal waves. Curr Biol 13:R7–R9. https://doi.org/10.1016/s0960-9822(02)01378-7
Soriano P (1999) Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet 21:70–71
Sternberg N, Hamilton D (1981) Bacteriophage P1 site-specific recombination. I. Recombination between loxP sites. J Mol Biol 150:467–486. https://doi.org/10.1016/0022-2836(81)90375-2
Wassef M, Luscan A, Battistella A et al (2017) Versatile and precise gene-targeting strategies for functional studies in mammalian cell lines. Methods 121–122:45–54. https://doi.org/10.1016/j.ymeth.2017.05.003
Yang H, Wang H, Shivalila CS et al (2013) One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell 154:1370–1379. https://doi.org/10.1016/j.cell.2013.08.022
Zhang H, Xu C, Yang R et al (2015) Silencing of nodal modulator 1 inhibits the differentiation of P19 cells into cardiomyocytes. Exp Cell Res 331:369–376. https://doi.org/10.1016/j.yexcr.2014.12.016
Zhang J, Zhao J, Jiang W et al (2012) Conditional gene manipulation: cre-ating a new biological era. J Zhejiang Univ Sci B 13:511–524. https://doi.org/10.1631/jzus.b1200042
Zhong H, Chen Y, Li Y et al (2015) CRISPR-engineered mosaicism rapidly reveals that loss of Kcnj13 function in mice mimics human disease phenotypes. Sci Rep 5:8366. https://doi.org/10.1038/srep08366
Acknowledgements
The authors wish to express their sincere thanks all Members of the Transgenic Facility (University of Salamanca) for their technical help and contributions to our CRISPR/Cas9 studies, and the staff of the Servicio de Experimentación Animal (University of Salamanca) and the Servicio de Secuenciación (IBMCC) for their technical assistance.
Funding
This work was supported by the Research Support Platform of the University of Salamanca, Spain (Nucleus) and PI16/01920 (ISCIII-FEDER).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have not conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the Ethical Standards of the institution or practice at which the studies were conducted (Conserjería de Agricultura y Ganadería de la Junta de Castilla y León ref. 338).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11033_2019_5214_MOESM1_ESM.tiff
Schematic representation of donor long ssDNA template. A Single strand DNA molecule containing LoxP sites flanking Nomo1 exon 3 sequence was used as donor template. Blue boxes represent homology arms and red triangles the LoxP sequences. The Pam mutated sequences are marked in yellow (TIFF 10262 kb)
11033_2019_5214_MOESM2_ESM.tiff
Msi-1 transgenic mice tissue specific Cre expression. A RT-PCR to detect Cre expression. Intestine and brain tissue showed Cre recombinase RNA expression. B X-Gal staining in small intestine collected from Msi1Cre/+ Rosa26 mouse (Perez-Caro et al. 2011). Left: Visualization of X-Gal staining of whole mount tissue. Right: X-Gal and eosin staining showing specific b-galactosidase areas: Cr (crypt bottom), pc (proliferative compartment). C B-galactosidase activity in specific brain areas from Msi1Cre/+ Rosa26 transgenic mice. Upper images have been adapted from Allen Developing Mouse Brain Atlas. Pyramidal layer (CA1, CA2, C3) and Dentate Gyrus (DG) granular cell layer (sg) showed specific B-Galactosidase activity. Lower images corresponding with whole mount X-gal brain staining (TIFF 10262 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
García-Tuñón, I., Vuelta, E., Lozano, L. et al. Establishment of a conditional Nomo1 mouse model by CRISPR/Cas9 technology. Mol Biol Rep 47, 1381–1391 (2020). https://doi.org/10.1007/s11033-019-05214-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-019-05214-7