Abstract
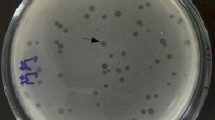
Mastitis in dairy cattle continues to be an economically important disease. However, control is complicated by a high prevalence of resistance to antibiotics. Phage therapy, therefore, is considered as an alternative way of controlling bacterial infections and contaminations. In this study, we have described isolation and characterization of a highly virulent phage SPW from wastewater of dairy farm, which possesses a strong lytic capability against mastitis-associated Staphylococcus aureus, the most important pathogen in bovine clinical and subclinical mastitis. The phage SPW produced large, round and clear plaques on bacterial culture plates. TEM showed phage SPW has an icosahedral head 62.5 nm in diameter and long tail of 106 nm, head and tail were held together by a connector of 18 ± 1.5 nm long and can be classified as a member of the Myoviridae family. Restriction analysis indicated that phage SPW was a dsDNA virus with an approximate genome size of 65–69 kb. One-step growth kinetics showed a short latency period of about 10–15 min and a rise period of 50 min and a relatively small burst size was 44 ± 3 phages particles/infected cell. Moreover, adsorption rates were not influenced by calcium ions and phage SPW was relatively stable in a wide range of temperature and pH values, and resistant to chloroform and isopropanol. The optimal multiplicity of infection (MOI) was 0.01. When phage SPW was used to infect five other clinically isolated pathogenic isolates, it showed relatively wide spectrum host range. Phage SPW was capable of eliciting efficient lysis of S. aureus, revealing it potentially as an effective approach to prophylaxis or treatment of S. aureus-associated mastitis in dairy cows.








Similar content being viewed by others
References
Levy SB, Marshall B (2004) Antibacterial resistance worldwide: causes, challenges and responses. Nat Med 10(12 Suppl):S122–S129
Gill JJ, Pacan JC, Carson ME, Leslie KE, Griffiths MW, Sabour PM (2006) Efficacy and pharmacokinetics of bacteriophage therapy in treatment of subclinical Staphylococcus aureus mastitis in lactating dairy cattle. Antimicrob Agents Chemother 50(9):2912–2918
Merril CR, Scholl D, Adhya SL (2003) The prospect for bacteriophage therapy in western medicine. Nat Rev Drug Discov 2(6):489–497
Summers WC (2001) Bacteriophage therapy. Annu Rev Microbiol 55:437–451
Sulakvelidze A, Alavidze Z, Morris JG Jr (2001) Bacteriophage therapy. Antimicrob Agents Chemother 45(3):649–659
Capparelli R, Parlato M, Borriello G, Salvatore P, Iannelli D (2007) Experimental phage therapy against Staphylococcus aureus in mice. Antimicrob Agents Chemother 51(8):2765–2773
Atterbury RJ, Van Bergen MA, Ortiz F, Lovell MA, Harris JA, De Boer A, Wagenaar JA, Allen VM, Barrow PA (2007) Bacteriophage therapy to reduce salmonella colonization of broiler chickens. Appl Environ Microbiol 73(14):4543–4549
Wills QF, Kerrigan C, Soothill JS (2005) Experimental bacteriophage protection against Staphylococcus aureus abscesses in a rabbit model. Antimicrob Agents Chemother 49(3):1220–1221
O’Flaherty S, Ross RP, Meaney W, Fitzgerald GF, Elbreki MF, Coffey A (2005) Potential of the polyvalent anti-Staphylococcus bacteriophage K for control of antibiotic-resistant staphylococci from hospitals. Appl Environ Microbiol 71(4):1836–1842
Son JS, Kim EB, Lee SJ, Jun SY, Yoon SJ, Kang SH, Choi YJ (2010) Characterization of Staphylococcus aureus derived from bovine mastitis and isolation of two lytic bacteriophages. J Gen Appl Microbiol 56(4):347–353
Han JE, Kim JH, Hwang SY, Choresca CH, Shin SP, Jun JW, Chai JY, Park YH, Park SC (2013) Isolation and characterization of a Myoviridae bacteriophage against Staphylococcus aureus isolated from dairy cows with mastitis. Res Vet Sci 95(2):758–763
Kwiatek M, Parasion S, Mizak L, Gryko R, Bartoszcze M, Kocik J (2012) Characterization of a bacteriophage, isolated from a cow with mastitis, that is lytic against Staphylococcus aureus strains. Arch Virol 157(2):225–234
Lovseth A, Loncarevic S, Berdal KG (2004) Modified multiplex PCR method for detection of pyrogenic exotoxin genes in staphylococcal isolates. J Clin Microbiol 42(8):3869–3872
Cerveny KE, DePaola A, Duckworth DH, Gulig PA (2002) Phage therapy of local and systemic disease caused by Vibrio vulnificus in iron-dextran-treated mice. Infect Immun 70(11):6251–6262
Chang HC, Chen CR, Lin JW, Shen GH, Chang KM, Tseng YH, Weng SF (2005) Isolation and characterization of novel giant Stenotrophomonas maltophilia phage phiSMA5. Appl Environ Microbiol 71(3):1387–1393
Fournier C, Kuhnert P, Frey J, Miserez R, Kirchhofer M, Kaufmann T, Steiner A, Graber HU (2008) Bovine Staphylococcus aureus: association of virulence genes, genotypes and clinical outcome. Res Vet Sci 85(3):439–448
Lu Z, Breidt F Jr, Fleming HP, Altermann E, Klaenhammer TR (2003) Isolation and characterization of a Lactobacillus plantarum bacteriophage, phiJL-1, from a cucumber fermentation. Int J Food Microbiol 84(2):225–235
Chaudhry WN, Haq IU, Andleeb S, Qadri I (2013) Characterization of a virulent bacteriophage LK1 specific for Citrobacter freundii isolated from sewage water. J Basic Microbiol 00:1–11
Ackermann HW (2007) 5500 Phages examined in the electron microscope. Arch Virol 152(2):227–243
Ackermann HW (2001) Frequency of morphological phage descriptions in the year 2000. Brief review. Arch virol 146(5):843–857
Garcia P, Madera C, Martinez B, Rodriguez A, Suarez JE (2009) Prevalence of bacteriophages infecting Staphylococcus aureus in dairy samples and their potential as biocontrol agents. J Dairy Sci 92(7):3019–3026
Dabrowska K, Skaradzinski G, Kurzepa A, Owczarek B, Zaczek M, Weber-Dabrowska B, Wietrzyk J, Maciejewska M, Budynek P, Gorski A (2010) The effects of staphylococcal bacteriophage lysates on cancer cells in vitro. Clin Exp Med 10(1):81–85
Synnott AJ, Kuang Y, Kurimoto M, Yamamichi K, Iwano H, Tanji Y (2009) Isolation from sewage influent and characterization of novel Staphylococcus aureus bacteriophages with wide host ranges and potent lytic capabilities. Appl Environ Microbiol 75(13):4483–4490
Matsuzaki S, Yasuda M, Nishikawa H, Kuroda M, Ujihara T, Shuin T, Shen Y, Jin Z, Fujimoto S, Nasimuzzaman MD et al (2003) Experimental protection of mice against lethal Staphylococcus aureus infection by novel bacteriophage phi MR11. J Infect Dis 187(4):613–624
Moldovan R, Chapman-McQuiston E, Wu XL (2007) On kinetics of phage adsorption. Biophys J 93(1):303–315
Acknowledgments
This study was supported by a grant from Shaanxi “13115” S&T Innovation Engineering Program (No. 2009ZDKG-18). We would like to thank Cheng Mingxia at College of Medicine, Xi’an Jiaotong University for the TEM study.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, L., Zhang, Z. Isolation and characterization of a virulent bacteriophage SPW specific for Staphylococcus aureus isolated from bovine mastitis of lactating dairy cattle. Mol Biol Rep 41, 5829–5838 (2014). https://doi.org/10.1007/s11033-014-3457-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-014-3457-2