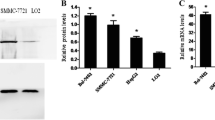
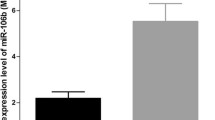
Abstract
Tumor angiogenesis, a major requirement for tumor growth and metastasis, is regulated by pro- and anti-angiogenic factors. The aim of this study was to quantify the expression of angiogenic (VEGF, HIF-1α, Angiopiotein-2) and anti-angiogenic (endostatin, angiostatin and Thrombospondin-1) factors and to discern their clinical relevance. A total 90 patients (67 HCC, 9 cirrhosis and 14 chronic hepatitis) were enrolled in the study. Tissue transcript levels of angiogenic (VEGF, HIF-1α, Ang-2) and anti-angiogenic (endostatin, angiostatin and TSP-1) factors were analyzed by quantitative real time-polymerase chain reaction (qRT-PCR) in the tissue samples. The tissue transcript levels of VEGF, HIF-1α and endostatin were found to be significantly higher in HCC in comparison to cirrhosis and chronic hepatitis. Although Ang-2, angiostatin and TSP-1 tissue transcript levels were higher in HCC group than the others groups but the difference was not statistically significant. In univariate analysis both VEGF and HIF-1α were found to be associated with poor survival of HCC patients. Multivariate analysis by the cox proportional hazard model revealed only VEGF as an independent factor predicting poor survival of the HCC patients. Angiogenic and anti-angiogenic factors are all highly expressed in HCC patients. Upregulation of tissue anti-angiogenic factors indicates the urgency for the alternative of anti-angiogenic therapies.



Similar content being viewed by others
References
Llovet JM, Burroughs A, Bruix J (2003) Hepatocellular carcinoma. Lancet 362:1907–1917
Roxburgh P, Evans TR (2008) Systemic therapy of hepatocellular carcinoma: are we making progress? Adv Ther 25:1089–1104
Llovet JM, Bruix J (2008) Novel advancements in the management of hepatocellular carcinoma in 2008. J Hepatol 48:S20–S37
Tanigawa N, Lu C, Mitsui T, Miura S (1997) Quantitation of sinusoid-like vessels in hepatocellular carcinoma: its clinical and prognostic significance. Hepatology 26:1216–1223
El-Assal ON, Yamanoi A, Soda Y et al (1998) Clinical significance of microvessel density and vascular endothelial growth factor expression in hepatocellular carcinoma and surrounding liver: possible involvement of vascular endothelial growth factor in the angiogenesis of cirrhotic liver. Hepatology 27:1554–1562
Yamamoto A, Dhar DK, El-Assal ON, Igarashi M, Tabara H, Nagasue N (1998) Thymidine phosphorylase (platelet-derived endothelial cell growth factor), microvessel density and clinical outcome in hepatocellular carcinoma. J Hepatol 29:290–299
Ramanujan S, Koenig GC, Padera TP, Stoll BR, Jain RK (2000) Local imbalance of proangiogenic and antiangiogenic factors: a potential mechanism of focal necrosis and dormancy in tumors. Cancer Res 60:1442–1448
Westphal JR, Van’t Hullenaar R, Peek R et al (2000) Angiogenic balance in human melanoma: expression of VEGF, bFGF, IL-8, PDGF and angiostatin in relation to vascular density of xenografts in vivo. Int J Cancer 86:768–776
An FQ, Matsuda M, Fujii H, Matsumoto Y (2000) Expression of vascular endothelial growth factor in surgical specimens of hepatocellular carcinoma. J Cancer Res Clin Oncol 126:153–160
Nör JE, Christensen J, Mooney DJ, Polverini PJ (1999) Vascular endothelial growth factor (VEGF)-mediated angiogenesis is associated with enhanced endothelial cell survival and induction of Bcl-2 expression. Am J Pathol 154:375–384
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N (1989) Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246:1306–1309
Ellis LM, Takahashi Y, Liu W, Shaheen RM (2000) Vascular endothelial growth factor in human colon cancer: biology and therapeutic implications. Oncologist 5(Suppl 1):11–15
Harris AL (2002) Hypoxia—a key regulatory factor in tumour growth. Nat Rev Cancer 2:38–47
Poellinger L, Johnson RS (2004) HIF-1 and hypoxic response: the plot thickens. Curr Opin Genet Dev 14:81–85
Wang GL, Semenza GL (1993) General involvement of hypoxia inducible factor 1 in transcriptional response to hypoxia. Proc Natl Acad Sci USA 9:4304–4308
Wang GL, Jiang BH, Rue EA, Semenza GL (1995) Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA 92:5510–5514
Weidemann A, Johnson RS (2008) Biology of HIF-1alpha. Cell Death Differ 15:621–627
Semenza GL (2003) Targeting HIF-1 for cancer therapy. Nat Rev Cancer 3:721–732
Asahara T, Chen D, Takahashi T et al (1998) Tie2 receptor ligands, angiopoietin-1 and angiopoietin-2, modulate VEGF-induced postnatal neovascularization. Circ Res 83:233–240
Sfiligoi C, de Luca A, Cascone I et al (2003) Angiopoietin-2 expression in breast cancer correlates with lymph node invasion and short survival. Int J Cancer 103:466–474
Tanaka F, Ishikawa S, Yanagihara K et al (2002) Expression of angiopoietins and its clinical significance in non-small cell lung cancer. Cancer Res 62:7124–7129
Ochiumi T, Tanaka S, Oka S et al (2004) Clinical significance of angiopoietin-2 expression at the deepest invasive tumor site of advanced colorectal carcinoma. Int J Oncol 24:539–547
Eggert A, Ikegaki N, Kwiatkowski J, Zhao H, Brodeur GM, Himelstein BP (2000) High-level expression of angiogenic factors is associated with advanced tumor stage in human neuroblastomas. Clin Cancer Res 6:1900–1908
Etoh T, Inoue H, Tanaka S, Barnard GF, Kitano S, Mori M (2001) Angiopoietin-2 is related to tumor angiogenesis in gastric carcinoma: possible in vivo regulation via induction of proteases. Cancer Res 61:2145–2153
Mitsuhashi N, Shimizu H, Ohtsuka M et al (2003) Angiopoietins and Tie-2 expression in angiogenesis and proliferation of human hepatocellular carcinoma. Hepatology 37:1105–1113
O’Reilly MS, Boehm T, Shing Y, Fukai N, Vasios G, Lane WS et al (1997) Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88:277–285
O’Reilly MS, Holmgren L, Shing Y, Chen C, Rosenthal RA, Moses M et al (1994) Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell 79:315–328
O’Reilly MS, Holmgren L, Chen C, Folkman J (1996) Angiostatin induces and sustains dormancy of human primary tumors in mice. Nat Med 2:689–692
Lannutti BJ, Gately ST, Quevedo ME, Soff GA, Paller AS (1997) Human angiostatin inhibits murine hemangioendothelioma tumor growth in vivo. Cancer Res 57:5277–5280
Bruix J, Sherman M, Llovet JM et al (2001) EASL panel of experts on HCC. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the study of the liver. J Hepatol 35:421–430
Gentilini P, Laffi G, La Villa G et al (1997) Long course and prognostic factors of virus-induced cirrhosis of the liver. Am J Gastroenterol 92:66–72
Lok AS, Seeff LB, Morgan TR, HALT-C Trial Group et al (2009) Incidence of hepatocellular carcinoma and associated risk factors in hepatitis C-related advanced liver disease. Gastroenterology 136:138–148
Leong DT, Gupta A, Bai HF et al (2007) Absolute quantification of gene expression in biomaterials research using real-time PCR. Biomaterials 28:203–210
Yin JL, Shackel NA, Zekry A et al (2001) Real-time reverse transcriptase-polymerase chain reaction (RT-PCR) for measurement of cytokine and growth factor mRNA expression with fluorogenic probes or SYBR Green I. Immunol Cell Biol 79:213–221
Kaido T, Uemoto S (2008) Recent evidence in the treatment of small hepatocellular carcinoma. Hepatogastroenterology 55:1460–1462
Wada H, Nagano H, Yamamoto H et al (2006) Expression pattern of angiogenic factors and prognosis after hepatic resection in hepatocellular carcinoma: importance of angiopoietin-2 and hypoxia-induced factor-1 alpha. Liver Int 26:414–423
Mabjeesh NJ, Amir S (2007) Hypoxia-inducible factor (HIF) in human tumorigenesis. Histol Histopathol 22:559–572
Hirota K, Semenza GL (2006) Regulation of angiogenesis by hypoxia-inducible factor 1. Crit Rev Oncol Hematol 59:15–26
Berra E, Ginouvès A, Pouysségur J (2006) The hypoxia-inducible-factor hydroxylases bring fresh air into hypoxia signalling. EMBO Rep 7:41–45
Uematsu S, Higashi T, Nouso K et al (2005) Altered expression of vascular endothelial growth factor, fibroblast growth factor-2 and endostatin in patients with hepatocellular carcinoma. J Gastroenterol Hepatol 20:583–588
Zeng W, Gouw AS, van den Heuvel MC et al (2008) The angiogenic makeup of human hepatocellular carcinoma does not favor vascular endothelial growth factor/angiopoietin-driven sprouting neovascularization. Hepatology 48(5):1517–1527
Huang GW, Yang LY, Lu WQ (2005) Expression of hypoxia-inducible factor 1alpha and vascular endothelial growth factor in hepatocellular carcinoma: impact on neovascularization and survival. World J Gastroenterol 11:1705–1708
Dai CX, Gao Q, Qiu SJ et al (2009) Hypoxia-inducible factor-1 alpha, in association with inflammation, angiogenesis and MYC, is a critical prognostic factor in patients with HCC after surgery. BMC Cancer 9:418
Poon RT, Chung KK, Cheung ST et al (2004) Clinical significance of Thrombospondin-1 expression in hepatocellular carcinoma. Clin Cancer Res 10:4150–4157
Gorrin Rivas MJ, Arii S, Furutani M et al (1998) Expression of human macrophage metalloelastase gene in hepatocellular carcinoma: correlation with angiostatin generation and its clinical significance. Hepatology 28:986–993
Kawahara N, Ono M, Taguchi K et al (1998) Enhanced expression of thrombospondin-1 and hypovascularity in human cholangiocarcinoma. Hepatology 28:1512–1517
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, B.K., Srinivasan, R., Kapil, S. et al. Angiogenic and anti-angiogenic factor gene transcript level quantitation by quantitative real time PCR in patients with hepatocellular carcinoma. Mol Biol Rep 40, 5843–5852 (2013). https://doi.org/10.1007/s11033-013-2690-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2690-4