Abstract
The emergence and spread of new crop diseases threatens the global food security situation. Phyllachora maydis, one of the three fungal pathogens involved in Tar Spot Complex (TSC) of maize, a disease native to Latin American countries, was detected for the first time in the United States of America (USA) in 2015. Although TSC has been previously reported to cause up to 50% of yield losses in maize in Latin America, the impact of P. maydis alone on maize yield is not known yet. However, there is a possibility that Monographella maydis, the second most important pathogen involved in TSC, would be introduced to the USA and would become associated with P. maydis and both pathogens could form the devastating complex disease in the country. The first objective of this study was to identify the TSC-vulnerable maize-producing regions across the USA by applying a climate homologue modeling procedure. The second objective was to quantify the potential economic impact of the disease on the maize industry in the USA. This study showed that even a 1% loss in maize production caused by the disease could potentially lead to a reduction in maize production by 1.5 million metric tons of grain worth US$231.6 million. Such production losses will affect not only the maize-related industries in the USA but also the food security in a number of low-income countries that are heavily dependent on US maize imports. This, in turn, may lead to increased poverty and starvation and, in some cases, to social unrest due to increased prices of maize-based staple foods. The study is intended to raise public awareness regarding potential TSC outbreaks and to develop strategies and action plans for such scenarios.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
By 2050, the world’s population is projected to increase in a range between 8.71 and 10.8 billion (assuming low and high fertility rates, respectively) compared to 7.35 billion in 2015 (The United Nations 2015). With the increase in population, ensuring food security is becoming a prime global concern as a 70% increase of food supply will be required by 2050 to ensure food supply of burgeoning population (Dubois 2011). Furthermore, the increased food resources have to originate from less land using less water as the global per capita arable land and internally renewable fresh water has been declining due to population growth (World Bank 2018).
In addition to the declining per capita arable land and other resources, the emergence and spread of devastating crop diseases has been challenging sustainable crop production (Bueno-Sancho et al. 2017). For example, the first introduction of wheat stripe rust pathogen (Puccinia striiformis) into Australia in 1978 and a secondary significant invasion of a different aggressive race of the same pathogen in 2002 (Wellings 2007; Loladze et al. 2014) have led to a number of commercially grown wheat cultivars to succumb to the disease resulting in significant economic losses. Another important wheat disease called stem or black rust of wheat, caused by Puccinia graminis, was successfully controlled worldwide. However, a virulent race of this disease, referred to as Ug99, has emerged in Africa, the Arabian Peninsula, the Middle East, and parts of Asia (Singh et al. 2006). Officially identified in 1985 in Parana State of Brazil (Igarashi et al. 1986), the wheat blast disease was limited only to several South American countries, but in 2015–2016, the disease has emerged in Bangladesh for the first time (Malaker et al. 2016), which has generated a real-time threat to the food security of entire South Asia. In case of maize, the maize lethal necrosis (MLN) disease emerged in Kenya in 2011 (Wangai et al. 2012), which later spread across large territories into surrounding countries often causing total loses of the maize crops (Mahuku et al. 2015).
Tar spot complex (TSC) is one of the major foliar diseases of maize in a number of Latin American countries (Fig. 1). An association of two fungal pathogens Phyllachora maydis and Monographella maydis and, presumably, a hyperparasitic fungus (i.e., a parasite of a parasite) Coniothyrium phyllachorae cause the disease. Although the exact mode of interaction between susceptible maize genotypes and the pathogens is not well understood yet, the combined effect of the interaction between P. maydis and M. maydis in the complex seems to be of critical importance, determining the end economic impact on grain yield (Hock et al. 1995). The grain yield losses associated with TSC have been reported to reach up to 51%, depending on the degree of susceptibility of maize genotypes and the presence of specific environmental factors favoring the development of the disease and the severity of the epidemics (Hock et al. 1989; Pereyda-Hernández et al. 2009).
The disease complex has been historically associated with tropical maize-growing areas (Fig. 2), and it was first officially documented in Mexico in 1904 and later in Bolivia, Colombia, Costa Rica, the Dominican Republic, Ecuador, El Salvador, Guatemala, Haiti, Panama, Peru, Puerto Rico, and Venezuela (Maublanc 1904; Abbott 1931; Bell and Alandia 1957; Schieber 1968; Castaño 1969; Malaguti and Subero 1972; Liu 1973; Bajet et al. 1994). The disease is more prevalent in the cooler tropical climates (average 16–23 °C) with higher humidity (Hock et al. 1989; Bajet et al. 1994). With extended leaf wetness periods at night and in the morning, monoculture and continuous production of maize over large territories favor TSC incidence, spread, and development (Hock et al. 1995). The complex is also becoming a newly emerging disease in non-tropical maize-growing regions with temperate or even colder climates, such as the high elevation plateaus of Central Mexico at over 2600 masl (Ríos-Herrera et al. 2016).
Countries in Latin America where the presence of tar spot complex has been reported. Source: authors’ development CABI (2017) Phyllachora maydis (black spot of maize). Invasive Species Compendium. Wallingford, UK: CAB International. http://www.cabi.org/isc/datasheet/40876. Accessed 25 April, 2018. based on CABI (2017)
P. maydis, one of the pathogens involved in TSC, is an obligate fungal parasite (i.e., an organism that must exploit its host plant in order to complete its life cycle) involved in TSC. This pathogen has been detected in Illinois and Indiana, the United States of America (USA), for the first time in 2015 and was reconfirmed in 2016 (Bissonnette 2015; Ruhl et al. 2016; Wise et al. 2016) and 2017 (Dr. M. Chilvers, personal communication). In addition, the presence of P. maydis has been confirmed in the state of Florida, USA, in 2016 (Bradley 2016; Hansen et al. 2016; Miller 2016).
The impact of P. maydis alone on maize yields and production in the USA is yet to be determined. However, its potential association with the other facultative saprophytes (i.e., an organism that can survive, live, and reproduce on both live and dead plants and plant residues), M. maydis, the second most important pathogen involved in TSC, can have a devastating impact on maize production in the USA. In case of such, a hypothetical invasion of M. maydis and the association of the two fungal pathogens forming the disease complex on the US territory, and its subsequent spread to the major maize-producing regions (the Corn Belt), could have a severe and devastating effect on maize production in the USA. The impact of the potential formation of TSC in the USA would be felt directly through the reduction of harvested grain or indirectly through the added costs associated with the purchase and application of disease-controlling fungicides. This can also have indirect but significant negative impacts on food and feed securities globally, especially in the most vulnerable maize import-dependent developing countries in Latin America, Africa, and Asia from rising grain prices. Food price hikes may also lead to social and political unrest and violence as these have been observed previously in Bangladesh, Egypt, and India (Kliger 2008; Clapp and Cohen 2009; Zerbe 2009). Another significant secondary impact the disease can potentially generate is the reduction of bio-ethanol production due to the losses caused by the emergence of TSC in the USA. This may potentially lead to increases in CO2 emissions due to the reduction in the use of ethanol-added fuel, which can further contribute to global climate change.
The objective of this study was to examine and quantify the potential negative effects and impacts of the TSC in the USA and its impacts at the global scale. Furthermore, the current study was intended to inform potential stakeholders regarding the threats associated with TSC and the global impacts that this disease may hypothetically bring on an international socioeconomic and political scale.
2 Tar spot complex disease of maize
The exact survival and over-wintering mechanisms of the pathogens involved in the complex are not known even in the regions where the disease is endemic. However, the reappearance of P. maydis in the USA in 2016 and 2017 after initial detection in 2015 suggests that the pathogen has survived the sub-zero temperatures and snow during the winters of 2015–2016 and 2016–2017, which does not represent the typical ecosystems to which the pathogen is naturally adapted (i.e., warm and humid tropical environments). It is unlikely that the pathogen has stayed alive through the so-called “green bridge effect” when it would have to survive on live host plants and provide the primary inoculum source for the major cropping season since most of the maize-growing regions where the pathogen has been detected in the USA are free of maize plants during the winter cycle. Although Ruhl et al. (2016) suggested that the pathogen might have been introduced to the USA by means of air movement through hurricanes, it is an unlikely scenario mostly because the ascospores of the pathogen are not as aerodynamic as, for example, the urediniospores of some cereal rusts (Puccinia spp.) and have not evolved in a way that would facilitate their movement through large territories by air.
Furthermore, P. maydis has likely coevolved with maize for hundreds of years in the tropical zones of Latin America, including those of Mexico, and there have been numerous hurricane events that could have facilitated the spread of the pathogen to North America prior to 2015. While the acreage of maize production in the USA has increased enormously during the last century, the pathogen has not been documented in the USA before 2015. Another, a more likely hypothesis of the pathogen introduction to the country could be the physical movement of people and goods from Latin America, including Mexico, to the USA. It is known that P. maydis can survive on dry leaves and parts of maize plants, and if these infected plants were imported to the maize-growing regions of the USA at some point, they could have served as a source of inoculum for the disease. An example of the infected plant parts brought to the USA could have been the husks of maize ears, often referred to in several Central American countries as “totomochtli” or “totomoxtle,” which are used extensively in some Latin American cultures for cooking purposes (e.g., making a wrapper for a dish called tamale), and taking into account the large number of people currently traveling between North, Central, and South America, the infected maize husks could have been unintentionally brought in luggage at some point. Alternatively, the spores of the disease could have been brought into the country on human clothing similar to the case of stripe rust, a devastating disease of wheat introduced to Australia in 1978 (Wellings 2007).
Because of the historical confinement of the disease mostly to the tropical environments of parts of Latin America, little attention has been paid to the research of TSC biology and to the development of resistant maize germplasm from outside of the region. The International Maize and Wheat Improvement Center (CIMMYT) has been the only international research and development organization involved in the improvement of resistance to TSC in advanced maize germplasm for approximately the last 30 years. However, the major objectives of CIMMYT principally concentrated on the improvement and distribution of enhanced and disease-resistant maize and wheat germplasm. Unfortunately, not enough attention has been paid to the research of the biology of the disease itself, which would have helped develop better-integrated disease management procedures, prevent its large outbreaks and epidemics, and reduce the inoculum load in the fields. Currently, the mechanisms of over-wintering/over-summering as well as the existence of alternative plant hosts of either P. maydis and/or M. maydis are not known.
3 The global role of the USA in maize production
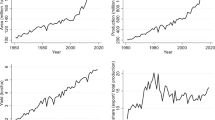
The USA is the single largest maize producer and exporter in the world. In 1964, the total world maize production reached 215.2 million metric tons (MMT), grown on 107.9 million ha and more than 22 MMT of maize were internationally traded (Tables 1 and 2). In the same year, the USA alone produced 88.5 MMT of maize, which was more than 41% of the total maize produced in the world on a total of 22.4 million ha. In 2014, the total world maize production reached nearly 1038 MMT produced from 184.8 million ha. Of these, the USA alone has produced 361 MMT of maize, reaching 34.8% of the total global maize production from 35.6 million ha, covering 18.2% of the total world maize acreage. The share of Asia and South America in the total global maize production has been increasing over the years as these regions now produce nearly 30 and 12% of the world’s maize, respectively. In contrast, the share of Africa, Europe, and the other regions in the world in global maize production has been either stagnant or decreasing (Table 1). The average maize yields have been historically low across Africa compared to the world average.
In 1964, the average global maize yield was 1.99 tons/ha, while the average maize yield in the USA and Africa was 3.95 and 0.98 tons/ha, respectively (Table 2). In 2014, the average global and the US maize yields have reached 5.62 and 10.73 tons/ha, respectively. In contrast, the average maize yield across the African continent remained low at 2.1 tons/ha. Exports by the USA have increased significantly from 12.2 MMT in 1964 to 24.2 MMT in 2014 (Table 2), making the USA the largest global exporter of maize.
The countries that import the most maize from the USA are Japan, Mexico, South Korea, and Colombia (Table 3), although the USA has also imported maize from Chile, Canada, Argentina, and Romania. On triennium average (2013–2015), the USA has exported more than US$1 billion worth of maize every year during 2013–2015, and at the same time, the country has imported US$572.2 million worth of maize from its trading partners. Importantly, a number of developing countries depend largely on maize imports from the USA. For example, Bangladesh, Costa Rica, Egypt, Indonesia, Pakistan, Panama, Peru, Turkey, and Venezuela import a significant amount of maize from the USA. These figures indicate that the supply of maize from the USA through international trade is critical to ensure the food and feed securities of many developing countries.
Despite the global leadership of the USA in maize production and trade, the share of the USA in global maize exports has been declining over the years, while the yearly per capita maize consumption around the world has increased steadily over the years (Fig. 3). In 1961, the global average annual maize consumption was less than 12 kg per capita, but in 2013, it had increased to nearly 18 kg per capita (FAOSTAT 2017). Furthermore, over the years, global maize exports have increased dramatically from less than 24 MMT in 1964 to approximately 129 MMT in 2014 (Table 2). However, the percentage share of the USA in the global maize export market has been declining since 1997; in 1964, the share of the USA in the total global maize export market was 52%, but in 2014, it had decreased to less than 19%. A further reduction in maize yield and maize production in the USA due to a possible spread of TSC may cause an additional decline in US maize exports. This will not only significantly affect the US$51.5 billion-worth maize economy of the USA (NCGA 2017) but will also tremendously affect the food and feed securities of many of the maize import-dependent developing countries through rising international maize prices. The so-called “tortilla crisis” is an example of how a grain price spike in the USA has caused the increase of the price of tortilla, an unleavened flat maize bread, by 72% in Mexico in 2006–2007 (Keleman and Rañó 2011).
World export of maize (million tons), percentage share of the USA in export, and maize consumption in the world (per capita/yearly/kg). Source: authors’ calculation based on FAOSTAT (2018)
Likewise, the US feed industry as well as the domestic supply and the exports of beef, poultry, and pork and dairy products may be significantly negatively affected by a reduction in maize yield and production. In 1996, the USA exported a little less than 500,000 metric tons of pork, nearly 900,000 metric tons of beef, and more than 2 million and 200,000 metric tons of poultry. In 2016, the exports of pork, beef, and poultry stood at nearly 2500, 1100, and nearly 3900 metric tons, respectively. According to the latest report (NCGA 2017), a total of 370 MMT of maize was used in the domestic market of the USA in 2016, of which 142.2 MMT (38.4%) was used as feed and residual production, 56.7 MMT (15.3%) was exported, and 107.1 MMT (28.9%) was used to produce bio-ethanol, an environmentally friendly fuel that is mixed with gasoline to reduce CO2 emissions. When bio-ethanol is mixed with ordinary fuel such as gasoline (currently at 10%), it is found that the net emission of CO2 is reduced by approximately 2.29 to 2.21 kg/l, which is close to 3.49% (The Government of Canada 2016). In 2016, more than 58 billion l of ethanol was used in the USA (Batres-Marquez 2017). Assuming the ethanol was mixed at the 10% level with gasoline, the CO2 emissions were reduced by 4.64 MMT only in 2016. The rest of maize was used for seed, beverages and alcohol, starch, and sweeteners and for high-fructose corn syrup preparation.
A reduction of maize production in the USA due to a possible outbreak of TSC therefore can have severe negative impacts on the animal and poultry feed industries as well as on the exports and domestic consumption of animal and poultry products. A reduction in maize yield and production may reduce ethanol production and increase ethanol prices, which can reduce the use of ethanol as an additive to gasoline and increase CO2 emissions and thus have a significant negative impact on the status quo of global climate change.
4 Materials and methods
4.1 Identifying maize-growing regions that are vulnerable to tar spot complex
The following environmental conditions have been considered the optimal for the development and outbreak of TSC: monthly mean temperatures of 17–22 °C, approximately 7 h of leaf wetness per night (dew formation, rain, fog), relative humidity above 75%, a minimum of 10 to 20 foggy days in a month, a minimum monthly rainfall of 150 mm, and 1800–1900 sunlight hours per year (Hock et al. 1995). The long-term data on temperature and rainfall patterns in the 13 counties in the states of Indiana, Illinois, Iowa, and Wisconsin, where the presence of P. maydis has been confirmed, were used to identify the homologue areas in the rest of the USA that match the temperature and rainfall patterns of these 13 counties during the maize growing season (Fig. 4). To identify locations similar in agro-climatic conditions relative to those where P. maydis has been detected, this study considered 30 years of historical weather data on temperature and rainfall. Since no precise geo-referenced location data was available for the locations where P. maydis has been reported, the centroids, being the geometric centers, of each county where P. maydis was reported were used to identify the outbreak geographic epicenters (Table 4).
The county-level information on the presence of P. maydis in the four Corn Belt states (Bissonnette 2015; Wise et al. 2016) was used to generate an analogue climate map utilizing the analogue tool. The analogue climate approach has been utilized for a number of studies mainly focused on climate change analysis (Hallegatte et al. 2007; Burke et al. 2009; Hayman et al. 2010; Thornton et al. 2011; Webb et al. 2013; Leibing et al. 2013; Berry et al. 2014; Kellett et al. 2015; Pugh et al. 2016; Dahinden et al. 2017; Flückiger et al. 2017). The analogue tool used here was based on R code, which was developed by the CGIAR Research Program on Climate Change, Agriculture and Food Security (CCAFS) and the University of Reading, UK, for climate change studies (Ramírez-Villegas et al. 2011). The R code is available in the online archive (Google Code Archive 2017). The analog tool was used to analyze climate parameters at a given location, taking into account rainfall and average temperature as variables (in weighted combination of each other or each factor independently). The tool was also used for spatial analysis to identify areas in the USA that had statistically similar (analogue) climates, based on a weighted similarity index, ranging from zero to 100. Notably, both variables can be weighted depending on the specific nature of the analysis needed (e.g., disease risk, crop aptitude, biotic and abiotic stress-adapted landraces, and representativeness of trial sites). After generating the analogue area grid for each county where P. maydis was detected, the individual grids were merged into one single analogue grid using the highest value for overlapping cells. Zonal statistics was used in ArcGIS software (ESRI 2015) to calculate the average similarity value per county in order to match with the county-level production data. The gridded outputs for all locations with similarity indices of at least 0.5 (50%), 0.6 (60%), and 0.7 (70%) were merged in a GIS and used to identify maize-producing counties in the USA at risk of TSC appearance. The data from the state of Minnesota, USA, was not considered in the current study since by the time of the analysis, the disease had not yet been officially confirmed in that state.
5 Results and discussion: The analysis of different scenarios of losses caused by potential tar spot complex outbreaks
The climate data (from one county in Iowa, two counties in Wisconsin, three counties in Illinois and seven counties in Indiana; Table 4; Fig. 4) was matched with the production, acreage and yield information of the United States Department of Agriculture (USDA 2016). In 2016, the USA produced 382.4 MMT of maize from 34.9 million ha (Table 5). The climate analogue analysis shows that in a potential situation of a climate similarity levels of 50% and above with the regions where P. maydis has emerged, a total of 24.4 million ha of maize cropland distributed over 782 counties within 21 states (producing 282.1 MMT of maize in 2016) would be identified as vulnerable to a potential TSC outbreak (Table 5). In case of a 60% and above levels of similarity in climatic patterns, a total of 19.6 million ha of maize cropland distributed between 503 counties across 13 states (producing estimated 233.6 MMT of maize in 2016) would be identified as prone to potential TSC epidemics (Table 5). Finally, under the scenario of a 70% and above level of climate similarity, a total of 11.97 million ha of maize cropland spread across 274 counties over nine states (producing 146.6 MMT of maize in 2016) would be recognized as vulnerable to possible TSC epidemics (Table 5). The vulnerable area, therefore, stretches into a broad territory across the Corn Belt of the USA (Fig. 4). This area represents a vast zone of maize-producing land, although most of the maize-producing regions across the Corn Belt remained relatively spared due to their different agro-climatic conditions.
The present study used three TSC-induced grain production shocks: a loss in maize production of 1, 3, and 5% and applied them across the vulnerable areas that may be prone to TSC epidemics with at least 50, 60, and 70% climate similarity indices compared to the disease epicenter, where P. maydis has been detected. The situation can be interpreted with an average scenario; for instance, 50% of the vulnerable area could be affected by a 1% yield loss, which is equivalent to the total of 0.5% production loss. Alternatively, 1% of the vulnerable area could be affected by a devastating 100% yield loss, which is equivalent to 1% loss of the total production, or variations thereof. As the impact of P. maydis alone (i.e., not the entire disease complex) on maize yield is not yet clearly understood, we assume the 1, 3, and 5% production shocks to be reasonable (albeit very conservative) scenarios within this ex ante impact assessment framework, which could also be considered a minimal level of reduction that TSC can potentially cause on susceptible maize hybrids (i.e., up to 50%).
An additional but important caveat is that these are potential TSC-induced production losses in the vulnerable areas during the years with high TSC incidence and severity. These scenarios depict specific situations where the season’s favorable environmental factors are conducive to an outbreak of TSC in the vulnerable areas (i.e., the simultaneous combination of cool and humid temperature, foggy night during the maize-growing cycles, and the presence of a high inoculum pressure). Such highly conducive conditions for TSC do not occur in all years, even in the most vulnerable tropical areas of Latin America. The scenarios thereby represent occurrences that may happen in one particular year simultaneously across the USA—but not necessarily every year in the vulnerable areas. Even if TSC occurs, the disease may manifest in a minimal incidence and/or severity. Likewise, if the disease appears late in the growing season when the grain filling process cannot be affected significantly, the damages will remain minimal. However, the appearance of P. maydis from 2015 through 2017 in various locations of the USA indicates that this pathogen is already well established in the country and will be very likely present in the country in the future.
Considering that preventive improvement of resistance to TSC has never been deliberately conducted in the maize breeding programs in the USA, the majority of maize germplasm is expected to be highly to moderately susceptible to TSC. Therefore, assuming 1, 3, and 5% loss scenarios only in the regions with at least 50, 60, and 70% similarity of climate indices would be a highly conservative approach to quantify the economic impacts of TSC on maize production in the USA.
An assumed 1% reduction in maize production caused by a possible TSC outbreak in the vulnerable regions under the 50, 60, and 70% and above levels of climatic similarities can reduce maize production in the USA by 2.8 MMT, 2.3 MMT, and 1.5 MMT, respectively (Table 5). In such a case, considering the price of maize is US$158/ton,Footnote 1 the simulated production losses would be worth of US$445.7 million, US$369.1 million, and US$231.6 million, respectively. Assuming a 3% loss scenario under the three levels of climatic similarities, the reduction of maize production in the USA can potentially reach 8.5 MMT, 7.0 MMT, and 4.4 MMT, respectively, worth US$1337.2 million, US$1107.2 million, and US$694.9 million, respectively (Table 5). Finally, a scenario of a 5% loss in maize production under the three levels of climatic similarities can reduce maize production in the USA by 14.1 MMT, 11.7 MMT, and 7.3 MMT, respectively, worth US$2228.7 million, US$1845.3 million, and US$1158.1 million, respectively (Table 5).
The likely proportional reduction in maize usage for export, ethanol production, consumption, and feed, due to the potential losses in maize production caused by TSC is summarized in Table 6. First, the USA exports of maize reached nearly 57 MMT in 2016, which comprised 15.3% of the total maize used in the USA (approximately 370 MMT). In the same year, 28.9% (107 MMT) of maize was used for ethanol production; 9.5% (36 MMT) was consumed as cereal, starch, beverage and alcohol, and sweeteners; 38.4% (142 MMT) was used as feed and residuals; and 7.6% (28 MMT) was used as distillers dried grains with soluble (DDGS, a by-product of the ethyl alcohol distillation process from grain after a yeast fermentation used as a feed additive).
Table 6 presents the impacts of a potential TSC outbreak-induced 1, 3, and 5% levels of maize production losses of the USA. In the analysis, we assume a single-sector adjustment method at a time. This implies that the loss in maize production due to a potential TSC outbreak will be adjusted only by a shift in a single sector, while the other sectors will remain unchanged. As we have already demonstrated earlier, under a scenario of a 50% climate similarity index, 24.4 million ha was identified as vulnerable to TSC (Table 5). A reduction of maize production by 1, 3, and 5% in these 24.4 million-ha areas could reduce the total maize production by 2.8 MMT, 8.5 MMT, and 14.1 MMT, respectively (Table 6). In the event that the USA compensates for the production losses only by reducing maize exports while keeping the other maize sectors unaffected, the country would have to reduce maize exports by 5–24.9% (Table 6). Similarly, if the country compensates for the losses by adjusting the ethanol production, the use of maize for this purpose would have to reduce by 2.6–13.2% (Table 6). Likewise, the USA would have to reduce domestic maize consumption by 7.8–38.9% to compensate for the production losses. In a similar manner, the reductions would be 2–10% in the sectors of feed and residual production and 10–50% in DDGS production.
Considering a 60% climate similarity index scenario, 19.6 million ha of maize land would be identified as vulnerable to potential TSC outbreaks. A reduction of maize production by 1, 3, and 5% in these 19.6 million-ha areas could reduce the total maize production by 2.3 MMT, 7 MMT, and 11.7 MMT, respectively (Table 6). To compensate for the production losses only by minimizing the exports, the USA would have to reduce the exports by 4–21% (Table 6). Similarly, if the country opts to adjust the production losses by reducing ethanol production, the use of maize for this purpose would have to decrease by 2–11% (Table 6). If, however, the USA decides to reduce maize consumption to adjust for the losses, the consumption has to be reduced by 6.5–32%, and finally, in case of adjustment in feed and DDGS production, they have to be reduced by 1.6–8 and 8–41%, respectively.
Considering a more conservative and restrictive approach, in a scenario of a 70% climate similarity index, nearly 12 million ha under maize would be found vulnerable to TSC (Table 5). A reduction of maize production by 1, 3, and 5% on this land could reduce total maize production by 1.5 MMT, 4.4 MMT, and 7.3 MMT, respectively (Table 6). In these cases, maize exports would have to reduce by 2.6–13% to compensate for the potential production losses. Alternatively, maize use for ethanol production, consumption, and feed and residual production would have to be reduced by 1.4–6.8, 4–20, and 1–5%, respectively (Table 6). Table 7 presents the impacts of TSC-induced maize production loss assuming a simultaneous adjustment in export, consumption, and other usage of maize. It also confirms that the USA would have to reduce export, consumption, and other usage of maize to adjust to the reduction in production due to a potential outbreak of TSC.
It has to be noted that the present study applied a partial equilibrium modeling approach in quantifying the impacts of TSC-induced potential maize production losses in the USA, assuming the impacts in other crop sector remain the same. In reality, however, any change in the maize production sector due to a potential outbreak of TSC may also affect the land allocation and production dynamics of other crops. However, the interpretation of these complex relationships is beyond the scope of the present study. Furthermore, the present study is not aiming at the prediction of the potential negative socioeconomic impacts of TSC outbreaks on the welfare at the household level in developing countries.
6 Conclusion and policy implications
The emergence of new crop diseases have previously threatened food security in several regions, and with the increased global human movement, this trend is expected to rise further: the emergence of the so-called Ug99 race of wheat stem rust in Africa, the Middle East, and the Arabian Peninsula, the emergence of wheat blast in Bangladesh, the spread of MLN in Africa, and lastly, the appearance of a P. maydis in the USA are examples of such real-time threats to the global food security.
One major component of TSC, P. maydis, has already been detected in the USA. Due to the geographic proximity of the USA to the tropical areas of Central America and increased human movement between the regions, there is a great risk of invasion of M. maydis, the second most important pathogen involved in TSC, into the country. This may cause the two pathogens to form the complex disease, which will result in severe outbreaks of TSC in the country since most of the US maize germplasm is expected to be susceptible.
The USA is the single largest maize producer and exporter in the world. The food and feed security of many low-income developing countries relies on US maize imports. Since TSC has been reported to cause up to 50% of yield loss, the emergence of the full-scale disease complex in the USA can have catastrophic impacts not only on more than US$51 billion-worth maize economy of the country but also on the food and feed securities at the global scale.
The present study identified 12 to 24.4 million ha under maize in the USA as vulnerable to potential TSC outbreaks. Taking into account the most conservative estimations of the potential yield loses that we have used in the current study, only 1% of maize yield decrease in 12 million ha under maize would result in the loss of 1.5 MMT of grain. This would translate to a monetary loss of US$231.6 million and will result in the shift of the dynamics of the use of grain in the country. Therefore, by using this most conservative estimation approach, we could demonstrate the deep impact the disease can have on the entire industry.
Furthermore, a potential reduction in the US maize exports may have an indirect yet very significant negative impact on food and feed securities globally, especially in the most vulnerable import-dependent developing countries in Latin America, Africa, and Asia. An increase in domestic food prices due to an increase in the price of maize in international markets may lead to social and political unrest and violence as this has been observed previously in Bangladesh, Egypt, and India. In addition, a potential reduction of bio-ethanol production due to the losses caused by TSC in the USA can lead to increases in CO2 emissions due to the reduction in the use of ethanol-added fuel, which can worsen the already grim situation with the global climate change.
The current study was envisioned to raise awareness regarding this devastating foliar disease of maize, as well as to call for action to tackle the potential impact the disease may have on the maize industry and to suggest developing both short-term and long-term action plans to prevent or minimize the potential economic losses. Significant investments are needed in research and development to understand better the biology of the pathogens involved in the complex, including the mechanisms of over-wintering and survival, the dissemination and spreading of the pathogens over large distances, the development of locally adapted resistant maize germplasm, and the establishment of integrated disease control practices. In the short run, it is also crucial to create an action plan to control outbreaks of the disease with fungicide applications in case of severe epidemics. This will include testing fungicides with different modes of action and application dosages and later certifying the fungicides for their use to control TSC. However, sole dependence on fungicide application to control TSC cannot be a viable option, considering the costs of the products and their application and potential impact they can have on the local ecology. Since TSC is a very likely biotic threat to the USA, raising awareness of the disease among maize growers and all major stakeholders through extension and information dissemination is critically important. For the longer-term solutions for the disease management, significant investments will need to be made to identify new and diverse sources and donors of resistance to the disease, which can be incorporated into the established elite and high-yielding maize germplasm. Furthermore, to prepare for the long-term solutions, a concerted effort and assured long-standing investments are needed immediately to develop and disseminate TSC-resistant maize germplasm as well as to undertake comprehensive disease surveillance and forecasting measures to ensure global maize food and feed securities.
Notes
As of June 2017, the international price of maize was US$157.96/ton (IndexMundi 2017). Available from URL: http://www.indexmundi.com/commodities/?commodity=corn (accessed on July 9, 2017).
References
Abbott E (1931) Further notes on plant diseases in Peru. Phytopathology 21(11):1061–1071
Bajet NB, Renfro BL, Carrasco JMV (1994) Control of tar spot of maize and its effect on yield. Int J Pest Manag 40(2):121–125
Batres-Marquez P (2017) U.S. ethanol production, consumption, and relevance of increasing exports. Agricultural Marketing Resource Center, Urbandale, Iowa, USA. https://www.agmrc.org/renewable-energy/renewable-energy-climate-change-report/renewable-energy-climate-change-report/april-2017-report/us-ethanol-production-consumption-and-the-relevance-of-increasing-exports/. Accessed 16 Mar 2018
Bell FH, Alandia BS (1957) Diseases of temperate climate crops in Bolivia. Plant Dis Rep 41:646–649
Berry P, Ramirez-Villegas J, Bramley H et al (2014) Regional impacts of climate change on agriculture and the role of adaptation. In: Jackson M, Ford-Lloyd B, Parry M (eds) Plant genetic resources and climate change. CABI, Wallingford, p 78
Bissonnette S (2015) Corn disease alert: new fungal leaf disease “tar spot” Phyllachora maydist identified in 3 northern Illinois counties. In: Bull. Pest Manag. Crop Dev. Inf. Illinois. Dep. Crop Sci. Univ. http://bulletin.ipm.illinois.edu/?p=3423. Accessed 16 Nov 2017
Bradley CA (2016) Corn disease to watch for in 2016: southern rust and tar spot. In: Kentucky Pest News. https://kentuckypestnews.wordpress.com/2016/06/14/corn-diseases-to-watch-for-in-2016-southern-rust-and-tar-spot/. Accessed 15 Nov 2017
Bueno-Sancho V, Persoons A, Hubbard A et al (2017) Pathogenomic analysis of wheat yellow rust lineages detects seasonal variation and host specificity. Genome Biol Evol 9(12):3282–3296
Burke MB, Lobell DB, Guarino L (2009) Shifts in African crop climates by 2050, and the implications for crop improvement and genetic resources conservation. Glob Environ Chang 19(3):317–325
Castaño A (1969) Mancha de asfalto (Tar Spot) de la hoja del maíz. Agric Trop 25:332
Clapp J, Cohen MJ (2009) The global food crisis: governance challenges and opportunities. Wilfrid Laurier University Press, Waterloo, Canada
Dahinden F, Fischer EM, Knutti R (2017) Future local climate unlike currently observed anywhere. Environ Res Lett 12(8):1–9
Dubois O (2011) The state of the world’s land and water resources for food and agriculture: managing systems at risk. Food and Agriculture Organization of the United Nations (FAO), Rome, Italy. http://www.fao.org/docrep/015/i1688e/i1688e00.pdf. Accessed 1 Apr 2018
ESRI (Environmental Systems Research Institute) (2015) ArcGIS release 10.3.1. Redlands, California, USA
FAOSTAT (2017) FAOSTAT: food balance sheets. http://www.fao.org/faostat/en/#data/FBS. Accessed 8 Jan 2018
FAOSTAT (2018) FAOSTAT: Crops and livestock products. http://www.fao.org/faostat/en/#data/TP. Accessed 25 April 2018
Flückiger S, Brönnimann S, Holzkämper A, Fuhrer J, Krämer D, Pfister C, Rohr C (2017) Simulating crop yield losses in Switzerland for historical and present Tambora climate scenarios. Environ Res Lett 12(7):074026
Google Code Archive (2017) CCAFS-analogues. Climate Change Agriculture and Food Security (CCAFS) https://code.google.com/archive/p/ccafs-analogues/downloads?page=1wnloads?page=1. Accessed 1 Apr 2018
Government of Canada (2016) Learn the facts: fuel consumption and CO2. Ontario: Ministry of Natural Resources, Government of Canada. http://www.nrcan.gc.ca/energy/efficiency/transportation/cars-light-trucks/buying/16770. Accessed 1 Apr. 2018
Hallegatte S b, Hourcade J-C, Ambrosi P (2007) Using climate analogues for assessing climate change economic impacts in urban areas. Clim Chang 82(1–2):47–60
Hansen J, Davison D, Jones D, Sun X (2016) Phyllachora maydis, corn tar spot. In: Pest Alert, Bur. Entomol. Nematol. Plant Pathol. http://www.freshfromflorida.com/content/download/69885/1624208/Pest Alert Phyllachora maydis, Corn Tar Spot.pdf. Accessed 15 Nov 2017
Hayman P, Wilhelm N, Alexander B, Nidumolu U (2010) Using temporal and spatial analogues to consider impacts and adaptation to climate change in the South Australian grain belt. In: Proceedings of 15th Agronomy Conference, Nov 15–18, 2010, Lincoln, New Zealand. pp 15–18. http://agronomyaustraliaproceedings.org/index.php/2-uncategorised/558-2010-index. Accessed 1 Apr 2018
Hock J, Kranz J, Renfro B (1989) El complejo “mancha de asfalto” de maíz: Su distribucción geográfica, requisitos ambientales e importancia económica en México. Rev Mex Fitopatol 7(2):129–135
Hock J, Kranz J, Renfo BL (1995) Studies on the epidemiology of the tar spot disease complex of maize in Mexico. Plant Pathol 44(3):490–502
Igarashi S, Utiamada CM, Igarashi LC, Kazuma AH, Lopes RS (1986) Pyricularia em trigo. 1. Ocorrencia de Pyricularia sp noestado do Parana. Fitopatol Bras 11:351–352
Keleman A, Rañó HG (2011) The Mexican tortilla crisis of 2007: the impacts of grain-price increases on food-production chains. Dev Pract 21(4–5):550–565
Kellett J, Hamilton C, Ness D, Pullen S (2015) Testing the limits of regional climate analogue studies: an Australian example. Land Use Policy 44:54–61
Kliger R (2008) Cairo grappling with bread crisis. The Jerusalem Post March 17 2017. http://www.jpost.com/Middle-East/Cairo-Grappling-with-Bread-Crisis. Accessed 16 Nov 2017
Leibing C, Signer J, van Zonneveld M, Jarvis A, Dvorak W (2013) Selection of provenances to adapt tropical pine forestry to climate change on the basis of climate analogs. Forests 4:155–178
Liu L-J (1973) Incidence of tar spot disease of corn in Puerto Rico. J Agric Univ Puerto Rico 42:211–216
Loladze A, Druml T, Wellings CR (2014) Temperature adaptation in Australasian populations of Puccinia striiformis f. sp. tritici. Plant Pathol 63(3):572–580
Mahuku G, Lockhart BE, Wanjala B, Jones MW, Kimunye JN, Stewart LR, Cassone BJ, Sevgan S, Nyasani JO, Kusia E, Kumar PL, Niblett CL, Kiggundu A, Asea G, Pappu HR, Wangai A, Prasanna BM, Redinbaugh MG (2015) Maize lethal necrosis (MLN), an emerging threat to maize-based food security in Sub-Saharan Africa. Phytopathology 105(7):956–965
Malaguti G, Subero LJ (1972) Mancha de asfalto del maiz. Agron Trop 22:443–445
Malaker PK, Barma NCD, Tiwari TP, Collis WJ, Duveiller E, Singh PK, Joshi AK, Singh RP, Braun HJ, Peterson GL, Pedley KF, Farman ML, Valent B (2016) First report of wheat blast caused by Magnaporthe oryzae Pathotype triticum in Bangladesh. Plant Dis 100(11):2330
Maublanc A (1904) Espéces nouvelles de champignons inferieurs. Bull la Soc Mycol Fr 20(2):72–74
Miller C (2016) Tar spot of corn detected for the first time in Florida. In: Inst. Food Agric. Sci. Palm Beach Cty. Coop. Ext. Serv. Dep. http://discover.pbcgov.org/coextension/agriculture/pdf/Tar Spot of Corn.pdf. Accessed 15 Nov 2017
NCGA (National Corn Growers Association) (2017) The world of corn: metric version 2017. Washington D.C. Natl. Corn Grow. Assoc. http://www.worldofcorn.com/pdf/_WOC_2017_Metric.pdf. Accessed 26 Nov 2017
Pereyda-Hernández J, Hernández-Morales J, Sandoval-Islas JS et al (2009) Etiología y manejo de la mancha de asfalto (Phyllachora maydis Maubl.) del maíz en Guerrero, México. Agrociencia 43(5):511–519
Pugh TAM, Müller C, Elliott J, Deryng D, Folberth C, Olin S, Schmid E, Arneth A (2016) Climate analogues suggest limited potential for intensification of production on current croplands under climate change. Nat Commun 7:12608
Ramírez-Villegas J, Lau C, Köhler A-K, Signer J, Jarvis A, Arnel N, Osborne T, Hooker J (2011) Climate analogues: finding tomorrow’s agriculture today. Working Paper No. 2. CGIAR Research Program on Climate Change Agriculture and Food Security (CCAFS). Cali, Colombia
Ríos-Herrera EN, Partida-Martínez LP, Ochoa-Fuentes YM, Cerna-Chávez E, Hernández-Castillo FD, Flores-Olivas A, Olalde-Portugal V, Rodríguez-Guerra R (2016) First report on the presence of Phyllachora sp. in corn crops at Toluca, Estado de Mexico. Am J Plant Sci 7(5):733–739
Ruhl G, Romberg MK, Bissonnette S, Plewa D, Creswell T, Wise KA (2016) First report of tar spot on corn caused by Phyllachora maydis in the United States. Plant Dis 100(7):1496
Schieber E (1968) Preliminary studies on Phyllachora maydis affecting corn in Central America. In: Phytopathology. American Phytopathological Society, St. Paul, p 554
Singh RP, Hodson DP, Jin Y et al (2006) Current status, likely migration and strategies to mitigate the threat to wheat production from race Ug99 (TTKS) of stem rust pathogen. CAB Rev Perspect Agric Vet Sci Nutr Nat Resour 1:1–13 http://cabiblog.typepad.com/hand_picked/files/PAV054.pdf. Accessed 1 Apr 2018
The United Nations (2015) World population prospects:the 2015 revision. Department of Economic and Social Affairs, Population Division. The United Nations, New York
Thornton PK, Jones PG, Ericksen PJ, Challinor AJ (2011) Agriculture and food systems in Sub-Saharan Africa in a 4°C+ world. Philos Trans R Soc A Math Phys Eng Sci 369(1934):117–136
UNCOMTRADE (2017) UN Comtrade Database. Department of Economic and Social Affairs, Statistics Division, Trade Statistics, United Nations, New York https://comtrade.un.org/data/. Accessed 23 Jul 2017
USDA (United States Department of Agriculture) (2016) National Agricultural Statistics Service: quick stats. United States Department of Agriculture, Washington D.C. https://quickstats.nass.usda.gov/results/95D392AB-2429-30EC-9B9B-C3C9F8B1FECC. Accessed 10 Nov 2017
Wangai AW, Redinbaugh MG, Kinyua ZM, Miano DW, Leley PK, Kasina M, Mahuku G, Scheets K, Jeffers D (2012) First report of maize chlorotic mottle virus and maize lethal necrosis in Kenya. Plant Dis 96(10):1582
Webb LB, Watterson I, Bhend J, Whetton PH, Barlow EWR (2013) Global climate analogues for winegrowing regions in future periods: projections of temperature and precipitation. Aust J Grape Wine Res 19(3):331–341
Wellings CR (2007) Puccinia striiformis in Australia: a review of the incursion, evolution, and adaptation of stripe rust in the period 1979-2006. Aust J Agric Res 58(6):567–575
Wise K, Ruhl G (2015) Tar spot on corn confirmed in the United States. Pest Crop Newsletter, Purdue Coop. Ext. Serv. Issue 24. https://extension.entm.purdue.edu/pestcrop/2015/Issue24/. Accessed 16 July 2017
Wise K, Ruhl G, Creswell T (2016) Disease of corn: tar spot. In: Purdue Crop. Ext. Serv. Purdue Univ. USA. https://www.extension.purdue.edu/extmedia/BP/BP-90-W.pdf. Accessed 16 Nov 2017
World Bank (2018) World development indicators: arable land (per capita/ha) and renewable internal freshwater resources per capita (cubic meters). http://databank.worldbank.org/data/reports.aspx?source=world-development-indicators. Accessed 26 Mar 2018
Zerbe N (2009) Setting the global dinner table. Exploring the limits of the marketization of food security. Setting the Global Dinner Table: Exploring the Limits of the Marketization of Food Security. Centre for International Governance Innovation. Wilfrid Laurier University Press, Waterloo, Canada
Acknowledgements
The authors would like to thank Ms. Jennifer Johnson for reviewing the manuscript. The contents and opinions expressed herein are those of the authors’ and do not necessarily reflect the views of the authors’ institution and donors and shall not be used for advertising or product endorsement purposes. The usual disclaimer applies.
Funding
This work was supported by the CGIAR Research Program on Maize (MAIZE) funded by the Government of Australia, Belgium, Canada, China, France, India, Japan, Korea, Mexico, Netherlands, New Zealand, Norway, Sweden, Switzerland, UK, USA and the World Bank.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no conflicts of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Mottaleb, K.A., Loladze, A., Sonder, K. et al. Threats of Tar Spot Complex disease of maize in the United States of America and its global consequences. Mitig Adapt Strateg Glob Change 24, 281–300 (2019). https://doi.org/10.1007/s11027-018-9812-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11027-018-9812-1